MINNEAPOLIS (May 20, 2026) – Peytant Solutions, the medical technology company that developed the first and only stent covered with human-derived amniotic membrane to receive FDA De Novo granted clearance, today announced the appointment of Tom Douthitt as chief executive officer. Douthitt is an experienced medical device executive with deep […]
Author: Ken Dropiewski
LivaNova Appoints Stefano Folli to Lead its Cardiopulmonary Business Unit
LONDON–(BUSINESS WIRE)—- $LIVN #cardiacsurgery–LivaNova PLC (Nasdaq: LIVN), a market-leading medical technology company, today announced that Stefano Folli will join the Company as incoming President, Cardiopulmonary (CP), effective June 1, succeeding Franco Poletti, who will retire on July 31. Folli will partner closely with Poletti over the coming months to ensure a seamless leadership transition before assuming full responsibility for the business on August 1. Folli will join the LivaNova Executive Leadership Tea
Carnegie Mellon University and Cleveland Clinic Develop AI System to Interpret Cardiac MRI Scans with Enhanced Accuracy
PITTSBURGH & CLEVELAND–(BUSINESS WIRE)– #AI–A team of researchers from Carnegie Mellon University, in collaboration with Cleveland Clinic’s Cardiovascular Innovation Research Center, has developed an artificial intelligence (AI) system capable of interpreting some of the most complex heart scans in medicine, cardiac magnetic resonance imaging (MRI), without the need for manually labeled training data. The novel system, called CMR-CLIP, is designed to interpret cardiac MRI scans by connecting movin
BioCardia Announces CardiAMP Chronic Myocardial Ischemia Trial Results Presented at EuroPCR Showed Durable Improvements in Exercise Tolerance with Reduced Angina Frequency
– Positive CardiAMP CMI Trial open-label cohort results demonstrated opportunity for locally delivered cell therapy to enhance therapeutic options for patients with severely symptomatic refractory angina and validate continued clinical development – Novel therapeutic approach targets a critically important unmet medical need for patients who have debilitated quality-of-life and have exhausted all available treatment options SUNNYVALE, Calif., May 21, 2026 (GLOBE NEWSWIRE) — BioCardia, Inc. (NASDAQ: BCDA), a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today announced the oral presentation of the results of the CardiAMP® Cell Therapy in Chronic Myocardial Ischemia Trial (CardiAMP CMI) preliminary open label cohort at EuroPCR, a leading interventional cardiovascular medicine conference held in Paris. The presentation was made by Dr. Amish Raval, M.D., Professor of Medicine at University of Wisconsin School of Medicine and Public Health. Results presented by Dr. Raval, on behalf of the CardiAMP CMI Investigators, showed that the minimally invasive CardiAMP Cell Therapy procedure was well tolerated with no treatment emergent major adverse cardiac events and patients demonstrated positive clinical outcomes including increased exercise tolerance and reduced frequency of angina episodes with the autologous cell therapy up through 2 years follow-up. Prior to treatment, all patients were on guideline-directed medical therapy (GDMT) and had received all available percutaneous and/or surgical options as appropriate for their medical condition. Patients experienced increased exercise tolerance, improving by an average of 179 seconds, which persisted for the two-year study follow-up. Angina episodes were reduced by an average of 82% by six-months after treatment. Dr. Raval’s presentation is here: EuroPCR2026. “Chronic ischemic heart disease results in considerable limitations of daily life activities due to chest discomfort, shortness of breath, and related disabling symptoms despite optimal medical therapy,” said Carl Pepine, MD, MACC, Professor of Medicine, Division of Cardiovascular Medicine, University of Florida at Gainesville. “A cell-based approach, added to this medical therapy, has potential to better manage these symptoms and improve the quality of life.” “We are thankful for the physician scientists who contributed their great experience to this program as well as the patients who participated,” said Peter Altman, PhD, BioCardia President and CEO. “These results support this CardiAMP cell therapy approach which has potential to help patients suffering from refractory angina. To our knowledge, only cell therapy has had a positive impact on both exercise tolerance and angina episodes in these patients.” About Chronic Myocardial Ischemia with Refractory Angina Chronic myocardial ischemia occurs in the setting of coronary artery disease when there is reduced blood flow to the heart. This causes angina, a type of chest pain which is characterized as refractory angina when this pain cannot be controlled by a combination of optimal medical therapy, angioplasty or bypass surgery, and is estimated to impact 600,000 to 1.8 million patients in the United States. Up to 15% of patients who have ischemia or angina and undergo cardiac catheterization are suboptimal candidates for conventional revascularization. Although prognosis of refractory angina has improved in recent years, patients with refractory angina experience a significantly impaired quality of life with disproportionately high utilization of healthcare services. These observations reflect the great need for new therapies for these patients. About the CardiAMP Cell Therapy Program Designated by the FDA as a Breakthrough Therapy for Ischemic Heart Failure, CardiAMP Cell Therapy uses a patient’s own bone marrow cells delivered to the heart in a minimally invasive, catheter-based procedure to potentially stimulate the body’s natural healing response. CardiAMP Cell Therapy incorporates three proprietary elements not previously utilized in investigational cardiac cell therapy: a pre-procedural cell analysis for patient selection, a high target dosage of cells, and a proprietary delivery system that has been shown to be safer than other intramyocardial delivery systems and exponentially more successful in cell retention. The CardiAMP cell therapy trials for the indications of both chronic myocardial ischemia and ischemic heart failure are covered by the Center for Medicare and Medicaid for both treatment and control procedures. CAUTION – Limited by United States law to investigational use. About BioCardia® BioCardia, Inc., headquartered in Sunnyvale, California, is a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary disease. CardiAMP® autologous and CardiALLO™ allogeneic cell therapies are the Company’s biotherapeutic platforms with three cardiac clinical stage product candidates in development. These therapies are enabled by its Helix™ biotherapeutic delivery and Morph® vascular navigation product platforms, and soon the Heart3D™ fusion imaging platform. BioCardia selectively partners on biotherapeutic delivery with peers developing important biologic therapies. For more information visit https://www.biocardia.com. Forward Looking Statements: This press release contains forward-looking statements that are subject to many risks and uncertainties. Forward-looking statements may include, among other things, statements relating to the continued development, ability to offset clinical costs utilizing Medicare reimbursement and the ultimate success of our clinical cell therapy programs. These forward-looking statements are made as of the date of this press release. We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. Factors that could cause or contribute to such differences include, but are not limited to, the Company’s liquidity position and its ability to raise additional funds, as well as the Company’s ability to successfully advance its clinical trials. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Form 10-K filed with the Securities and Exchange Commission on March 24, 2026, under the caption titled “Risk Factors,” and in our subsequently filed Quarterly Reports on Form 10-Q. BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law. Media Contact: Miranda Peto, Investor RelationsEmail: mpeto@BioCardia.com Phone: 650-226-0120 Investor Contact: David McClung, Chief Financial OfficerEmail: investors@BioCardia.comPhone: 650-226-0120
Adagio Medical Submits Premarket Approval Application to FDA for vCLAS® Ventricular Ablation System for the Treatment of Ventricular Tachycardia
LAGUNA HILLS, Calif.–(BUSINESS WIRE)—- $ADGM #ARRHYTHMIA–Adagio Medical Holdings, Inc (Nasdaq: ADGM) (“Adagio” or “the Company”), a leading innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, today announced the submission of its Premarket Approval (“PMA”) application to the U.S. Food and Drug Administration (“FDA”) for the vCLAS Ventricular Ablation System for the treatment of drug-refractory, recurrent, sustained monomorphic ventricular tachycardia (“VT”) in patients with isch
Reflow Medical Reports Six-Month Results from DEEPER CORONARY Study of Spur® Elute Stent
SAN CLEMENTE, Calif.–(BUSINESS WIRE)– #EuroPCR–Reflow Medical announces six-month DEEPER CORONARY study results for the Spur® Elute Sirolimus-Eluting Retrievable Stent System at EuroPCR 2026.
MemorialCare Heart & Vascular Institute Becomes First Hospital in Los Angeles County to Offer Cleerly AI-Powered Heart Disease Analysis, Quantifying Total Plaque Volume and Soft Plaque
Systemwide launch brings AI powered assessment to Coronary CTA’s to MemorialCare’s Long Beach Medical Center, Orange Coast Medical Center, and Saddleback Medical Center. FOUNTAIN VALLEY, Calif., May 20, 2026 /PRNewswire/ — MemorialCare today announced the systemwide implementation of…
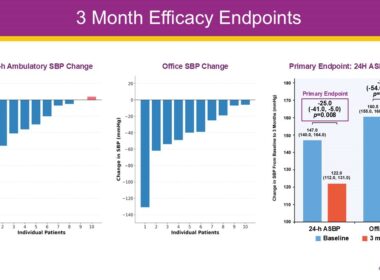
DeepQure Announces Robust 3-Month Results from HERO-HTN Clinical Study at EuroPCR 2026
PARIS, May 20, 2026 /PRNewswire/ — DeepQure, a clinical-stage medical device company, today announced robust 3-month clinical data from its HERO-HTN-FIH study during a featured presentation at EuroPCR 2026. The interim results demonstrate that the HyperQure™ extravascular renal…
Jupiter Endovascular Announces Positive Results From SPIRARE II Pivotal Trial at EuroPCR 2026
MENLO PARK, Calif.–(BUSINESS WIRE)– #TFX–Jupiter Endovascular announced positive results from its SPIRARE II pivotal trial at EuroPCR showing the trial met both of its primary endpoints.
JenaValve Appoints Tonya Porter as Vice President of Regulatory Affairs
IRVINE, Calif., May 20, 2026 (GLOBE NEWSWIRE) — JenaValve Technology, Inc., developer and manufacturer of the Trilogy® Transcatheter Heart Valve (THV) System, today announced the appointment of Tonya Porter as Vice President of Regulatory Affairs. Tonya will lead the company’s regulatory affairs function as JenaValve advances its programs through clinical development and ongoing commercialization efforts. She succeeds Jane Metcalf, who is retiring following a distinguished tenure at JenaValve.