SANTA CLARA, Calif.–(BUSINESS WIRE)–Coredio, a digital health company developing the first software-as-a-medical-device (SaMD) platform dedicated to heart failure (HF) hemodynamic assessment, today announced that the U.S. Food and Drug Administration has granted its Cardiac Performance Simulation Engine (CPSE™) Breakthrough Device Designation and accepted the platform into the FDA’s Total Product Life Cycle Advisory Program (TAP). CPSE™ is a software-only platform designed to deliver catheter
Coronary/Structural Heart
Groundbreaking therapy for advanced heart failure: Outcomes of the BIOVAT-HF clinical trial published in NEJM
Repairon Announces Publication of Clinical Trial Results for Engineered Human Heart Muscle Tissue in the New England Journal of MedicineGÖTTINGEN, Germany and PROVIDENCE, R.I., May 28, 2026 (GLOBE NEWSWIRE) — Repairon, a biotechnology company developing regenerative cardiac therapies, today announced that clinical results evaluating its engineered human heart muscle tissue for patients with advanced heart failure have been published in the New England Journal of Medicine (NEJM). The publication concludes that restoration of heart muscle function in patients with advanced heart failure is achievable, resulting in improved health and quality of life. Publication details: Zimmermann WH, Ensminger S, Kutschka I, et al. Stem-Cell-Derived Biologic Ventricular Assist Tissue in Heart Failure. N Engl J Med. May 28;394(20):1991-2001 DOI: 10.1056/NEJMoa2513525 The data were generated in the BioVAT-HF clinical trial (BiologicalVentricularAssistTissue in TerminalHeartFailure). Designed to assess safety and preliminary efficacy, this Phase 1-2 study involved implanting fully functional heart muscle patches engineered from human induced pluripotent stem cell-derived terminally differentiated heart muscle cells onto the weakened left ventricular muscle of patients with advanced heart failure with reduced ejection fraction (HFrEF), alongside guideline-directed medical therapy. The trial began with a dose-escalation phase to establish the safe maximum dose, followed by treatment at that dose to further evaluate safety and efficacy. Of the 20 patients enrolled, 16 received the safe maximum dose. At the time of reporting, the last enrolled patient had completed 3 months of follow-up, and follow-up across these patients ranged from 6 to 52 months. Key safety findings from the NEJM Publication indicate that 3 patients died during the trial from causes that the Data Safety Monitoring Board graded as unrelated to the BioVAT. Severe adverse events were mostly related to underlying heart and concurrent diseases as well as to immunosuppression requiring adaptation of the immunosuppression regimen. 3 patients experienced episodes of ventricular tachycardia which were found to be unrelated to the BioVAT transplant. No patients had ventricular fibrillation. Among the 16 safe maximal dose patients, there were heart failure hospitalizations for 2 patients. The efficacy findings from the NEJM Publication indicate that for those patients who received the safe maximal dose, from baseline: Target heart wall thickness increased 4.5 mm at 3 months and 2.9 mm at 12 months follow-up.Left ventricular ejection fraction increased 3.9% at 3 months and 6.9% at the latest timepoint.Quality of Life as measured by KCCQ-OSS increased 6.7 points at 3 months and 15 points at 12 months follow-up. The study outcomes support preclinical findings that engineered heart muscle can integrate with damaged myocardium, form a vascularized layer, and contract in synchrony with native tissue. This was further confirmed by analysis of an explanted heart from a patient in the dose-finding cohort who later underwent cardiac transplantation, providing clear evidence of human heart remuscularization and associated increases in wall thickness, ejection fraction, and quality of life. The authors concluded that further clinical investigations with longer follow-up times are warranted. Wolfram-Hubertus Zimmermann. MD, professor and director of the Institute of Pharmacology and Toxicology at the University Medical Center Goettingen, Germany, and principal author of the NEJM Publication, remarked that “heart failure therapies available today can often slow the progression of the disease, but they cannot replace destroyed heart muscle. Our goal, therefore, is to generate new, functional heart muscle tissue and thereby provide targeted support to the weakened heart.” Lothar Germeroth, Ph.D., Repairon’s CEO, stated: “We are highly encouraged by these Phase II results, which we believe validate the therapeutic potential of our regenerative cardiac patch platform. Heart failure remains one of the leading causes of morbidity and mortality worldwide, and we believe these findings may open a new chapter in myocardial regeneration and restorative cardiovascular medicine.” High medical need in advanced heart failure: Approximately 5% of the global population suffers from chronic heart failure of any severity, and it remains one of the most common causes of death. In the US, heart failure represents the most common cause of hospitalization and mortality in the senior population, and over 6 million people are affected. As heart failure progresses to advanced stages, patients experience weakness with discomfort during all physical activities and at rest, sometimes even requiring constant bed rest. For these severely ill patients, the only treatment options currently available are mechanical pump devices or heart transplantation. About Repairon: Repairon GmbH is a German biotech company based in Göttingen, Germany, focused on developing regenerative cell therapies for cardiac medicine. The company was founded in 2014 based on research by Wolfram-Hubertus Zimmermann, MD and his team at the University Medical Center Goettingen, who have developed several tissue engineering technologies with proven applicability for organ repair and drug development. Repairon’s lead therapeutic candidate, the human engineered heart muscle patch, is currently being evaluated in the BioVAT-HF Phase 2 clinical trial as a biological ventricular assist tissue (BioVAT) for end-stage heart failure. Company contact:Dr. Lothar GermerothRepairon GmbH37079 Göttingen – GermanyEmail: l.germeroth@repairon.comUS contact:Frank AhmannRepairon USAProvidence, RI Email: f.ahmann@repairon.com
Cardiosense Receives FDA De Novo Classification for Novel Cardiac Technology Designed to Improve Care for Patients with Heart Failure
CHICAGO–(BUSINESS WIRE)–Cardiosense, a healthcare technology company focused on heart failure care, today announced that the U.S. Food & Drug Administration (FDA) has granted De Novo classification for its PCWP Analysis Software™, a first-in-class technology to estimate a key indicator of heart health noninvasively. With this authorization, Cardiosense has a clear path forward in supporting clinicians who manage heart failure by providing critical data to improve therapy and avoid prolong
Editas Medicine Presents EDIT-401 Preclinical Data Demonstrating Robust Reductions in LDL-C, Lp(a), and ApoB in Non-Human Primates at the 94th European Atherosclerosis Society Congress
Single dose of EDIT-401 achieved ~90% or greater mean reductions in LDL-C, Lp(a), and ApoB in non-human primates Data reinforce differentiated LDLR upregulation approach with rapid, dose-dependent effects on multiple atherogenic lipoproteins Company on track to submit CTN by mid-2026 for EDIT-401 and achieve early in vivo human proof-of-concept data by the end of 2026 CAMBRIDGE, Mass., May 26, 2026 (GLOBE NEWSWIRE) — Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company focused on developing transformative medicines for serious diseases, presented new preclinical data for EDIT-401, its lead in vivo development candidate, in an oral presentation at the 94th European Atherosclerosis Society (EAS) Congress in Athens, Greece on May 25, 2026. In the data presented, EDIT-401 achieved robust reductions in LDL-cholesterol (LDL-C), lipoprotein(a) (Lp(a)), and apolipoprotein B (ApoB) in non-human primates (NHPs), supporting its potential as a best-in-class medicine for hyperlipidemia. Key EDIT-401 preclinical data in NHPs presented include: A single dose of EDIT-401 achieved ≥90% mean reduction in LDL-C, with rapid and dose-dependent effect.EDIT-401 achieved rapid, dose dependent ~90% mean reduction in Lp(a), an independent risk factor for atherosclerotic cardiovascular disease (ASCVD).EDIT-401 achieved rapid, dose-dependent ~90% mean reduction in ApoB, a key measure of total plaque-causing cholesterol particles and predictive measure for ASCVD.Reductions in LDL-C, Lp(a), and ApoB were highly correlated, supporting a unified mechanism facilitated by LDLR upregulation. “The consistent reductions of ~≥90 percent with EDIT-401 in LDL-C, Lp(a), and ApoB observed in these preclinical studies highlight the transformative potential of our LDLR upregulation approach to address multiple drivers of cardiovascular risk, including residual risk beyond LDL-C alone,” said Linda C. Burkly, Ph.D., Executive Vice President and Chief Scientific Officer, Editas Medicine. “These robust and consistent reductions across multiple atherogenic lipoproteins with a single dose further support EDIT-401 as a potentially best-in-class in vivo gene editing medicine for people living with hyperlipidemia.” The abstract can be accessed on the EAS website, and the presentation is available on the Editas Medicine website. Editas continues to advance preclinical studies for EDIT-401, including an ongoing Good Laboratory Practice (GLP) toxicology study in NHPs. Interim results from this study demonstrated EDIT-401 was well-tolerated with no adverse clinical observations, no notable treatment-related liver enzyme elevations, and no liver histopathology findings in non-GLP toxicology at the therapeutically relevant dose of 1.5 mg/kg. The Company also received positive pre-IND feedback from the U.S. Food and Drug Administration (FDA) on its nonclinical package, CMC plans, and study design to support an Investigational New Drug Application (IND). The Company plans to submit a Clinical Trial Notification (CTN) in Australia to the Therapeutic Goods Administration (TGA) by mid-2026 to initiate a first-in-human clinical trial of EDIT-401 in patients with Heterozygous Familial Hypercholesterolemia (HeFH) later this year, and is on track to have early in vivo human proof-of-concept data for EDIT-401 by the end of 2026. About Editas Medicine As a pioneering gene editing company, Editas Medicine is focused on translating the power and potential of CRISPR genome editing systems into a robust pipeline of transformative in vivo medicines for people living with serious diseases around the world. Editas Medicine aims to discover, develop, manufacture, and commercialize durable, precision in vivo gene editing medicines for a broad class of diseases. Editas Medicine is the exclusive licensee of Broad Institute’s Cas12a patent estate and Broad Institute and Harvard University’s Cas9 patent estates for human medicines. For the latest information and scientific presentations, please visit www.editasmedicine.com. Forward-Looking StatementsThis press release contains forward-looking statements and information within the meaning of The Private Securities Litigation Reform Act of 1995. The words ‘‘anticipate,’’ ‘‘believe,’’ ‘‘continue,’’ ‘‘could,’’ ‘‘estimate,’’ ‘‘expect,’’ ‘‘intend,’’ ‘‘may,’’ ‘‘plan,’’ ‘‘potential,’’ ‘‘predict,’’ ‘‘project,’’ ‘‘target,’’ ‘‘should,’’ ‘‘would,’’ and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Forward-looking statements in this press release include statements regarding the initiation, timing, progress and results of the Company’s preclinical studies and its research and development programs, including initiating a first-in-human study for EDIT-401 in 2026 and achievement of early in vivo human proof-of-concept data for EDIT-401 by the end of 2026; the potential of, and expectations for, EDIT-401; and the timing or likelihood of regulatory filings and approvals, including submitting a CTN in Australia by mid-2026 for EDIT-401. The Company may not actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements, and you should not place undue reliance on these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in these forward-looking statements as a result of various important factors, including: uncertainties inherent in the initiation and completion of preclinical studies and clinical trials; availability and timing of results from preclinical studies and clinical trials; uncertainties relating to planned regulatory submissions to initiate clinical trials, including that results of preclinical studies will warrant such submissions or that regulatory agencies may require additional preclinical studies, that regulatory submissions shall occur on the expected timelines and that regulatory authorities will provide clearance for trials to be initiated; that the results and outcome of preclinical studies may not be predictive of the results of clinical trials; and the availability of funding sufficient for the Company’s foreseeable and unforeseeable operating expenses and capital expenditure requirements. These and other risks are described in greater detail under the caption “Risk Factors” included in the Company’s most recent Annual Report on Form 10-K, which is on file with the Securities and Exchange Commission, as updated by the Company’s subsequent filings with the Securities and Exchange Commission, and in other filings that the Company may make with the Securities and Exchange Commission in the future. Any forward-looking statements contained in this press release represent the Company’s views only as of the date hereof and should not be relied upon as representing its views as of any subsequent date. Except as required by law, the Company explicitly disclaims any obligation to update any forward-looking statements. CONTACT: Investor and Media Contacts:
ir@editasmed.com
media@editasmed.com
Novo Nordisk global observational study reveals 2 in 5 people with cardiovascular disease have cardiovascular inflammation, increasing their risk of heart attack and stroke
The Novo Nordisk study POSEIDON – including 18,904 patients across 18 countries – showed that cardiovascular (CV) inflammation remains highly prevalent among people with cardiovascular disease (CVD), despite current standard of care treatment1,2In fact, two in five people with atherosclerotic cardiovascular disease (ASCVD) and chronic kidney disease (CKD), or heart failure, have CV inflammation1,2This matters because CV inflammation is an independent risk factor for CV events, such as heart attack and stroke, in people living with CVD, and it shows a significant gap in CV care globally3 Bagsværd, Denmark, 26 May 2026 – Novo Nordisk today presented new results from the landmark POSEIDON real-world evidence study at the 94th European Atherosclerosis Society (EAS) Congress in Athens, Greece. The study showed that CV inflammation remains highly prevalent among people with CVD despite current standard-of-care treatment. The study found that 2 in 5 people with ASCVD and CKD had CV inflammation, which is associated with an increased risk of major CV events2,4. A second POSEIDON analysis recently published in the European Journal of Heart Failure showed that two in five people with heart failure also have CV inflammation1. In POSEIDON, CV inflammation was measured and defined by high-sensitivity C-reactive protein (hsCRP) levels ≥2 mg/L1. hsCRP is the most commonly used and widely available blood test for measuring CV inflammation4-6. These findings underscore a significant gap in current CV care. Even when people receive guideline-recommended treatments to control, e.g., cholesterol, blood pressure and blood sugar, inflammation-driven CV risk persists3,7. The POSEIDON study represents one of the largest contemporary global assessments of CV inflammation prevalence in this high-risk population1,2. “The POSEIDON study provides critical evidence that cardiovascular inflammation represents a significant source of persistent risk in people living with atherosclerotic cardiovascular disease and chronic kidney disease or heart failure, despite receiving standard of care treatment today,” said Filip Knop, senior vice president and chief medical officer at Novo Nordisk. “Understanding the scope of cardiovascular inflammatory risk is essential, as we continue our innovation-driven research to develop a first-in-class therapy with the potential to address this critical unmet need.” POSEIDON enrolled 18,904 patients across 18 countries spanning Europe, North America, South America and Asia-Pacific between 2023 and 20251,2. Within the study, 13,475 patients had ASCVD, of whom 5,757 (42.7%) had CKD, while 11,809 patients had heart failure spanning across all types of heart failure (preserved, mildly reduced or reduced)1,2. Cardiovascular inflammation plays a central role in the development and progression of ASCVD8,9. Multiple studies have shown that people with CV inflammation face an increased risk of major adverse cardiovascular events, including heart attack, stroke and CV death3-5. Inflammation also contributes to CKD progression, and CKD itself may promote inflammation, creating a cycle that amplifies CV risk9. It also plays a key role in heart failure, and it is common across all types of heart failure, particularly in people with obesity, kidney disease and other metabolic conditions1,10. “POSEIDON makes clear that inflammation is not a peripheral concern – it is a shared driver of risk affecting millions of patients worldwide with cardiovascular disease who remain vulnerable despite our best current therapies,” said Professor Carolyn S.P. Lam, Senior Consultant, Department of Cardiology, National Heart Centre Singapore; and Professor, Cardiovascular & Metabolic Disorders Signature Research Programme, Duke-NUS Medical School. “What is striking is the consistency of inflammatory signals across such diverse patient populations. That consistency points to a practical way forward – identifying patients most likely to benefit from therapies that directly target inflammation. This reframes how we should think about residual cardiovascular risk, and it underscores the promise of emerging anti-inflammatory therapies to address a real unmet need.” The growing recognition of the role of inflammation in cardiovascular disease is reflected in recent guidelines from the European Society of Cardiology (ESC), the American Heart Association (AHA), and the American College of Cardiology (ACC), which include elevated hsCRP as a risk-modifying biomarker to guide more intensive preventive initiatives11,12. About POSEIDONPOSEIDON is a large, multinational, cross-sectional observational real-world evidence study designed to evaluate the prevalence and characteristics of high inflammatory risk (defined as hsCRP ≥2 mg/L) in patients with ASCVD, with and without CKD and/or heart failure. The study enrolled 18,904 patients across 18 countries between 2023 and 2025. Patients with recent infections, hospitalisations or unplanned medical visits were excluded to ensure that inflammatory markers reflected CV inflammation rather than acute conditions1,2. About cardiovascular inflammationCardiovascular inflammation is increasingly recognised as a key driver of ASCVD and a major contributor to persistent cardiovascular risk in people receiving standard preventive therapies. It also plays a key role in heart failure, and it is common across all types of heart failure (preserved, mildly reduced or reduced), particularly in people with obesity, kidney disease and other metabolic conditions. hsCRP is the most widely available blood test and validated biomarker of CV inflammation. Levels of CV inflammation ≥2 mg/L are associated with increased risk of major adverse CV events, including heart attack, stroke and CV death4-6. Despite standard of care management of traditional risk factors such as cholesterol, blood pressure and blood sugar, many people with ASCVD continue to face elevated CV risk driven in part by persistent inflammation2,3,7. The same is true for people with any type of heart failure, where CV inflammation is associated with more severe symptoms and an increase in the overall CV risk1. About Novo NordiskNovo Nordisk is a leading global healthcare company founded in 1923 and headquartered in Denmark. Our purpose is to drive change to defeat serious chronic diseases built upon our heritage in diabetes. We do so by pioneering scientific breakthroughs, expanding access to our medicines, and working to prevent and ultimately cure disease. Novo Nordisk employs about 67,900 people in 80 countries and markets its products in around 170 countries. For more information, visit novonordisk.com, Facebook, Instagram, X, LinkedIn and YouTube. Contacts for further information Novo Nordisk Media: Ambre James-Brown+45 3079 9289globalmedia@novonordisk.com Liz Skrbkova (US)+1 609 917 0632usmediarelations@novonordisk.comNovo Nordisk Investors: Michael Novod+45 3075 6050nvno@novonordisk.comJacob Martin Wiborg Rode+45 3075 5956jrde@novonordisk.comSina Meyer +45 3079 6656 azey@novonordisk.comMax Ung+45 3077 6414mxun@novonordisk.comChristoffer Sho Togo Tullin+45 3079 1471cftu@novonordisk.com Alex Bruce+45 3444 2613axeu@novonordisk.com Mads Berner Bruun+45 3075 2936mbbz@novonordisk.comFrederik Taylor Pitter (US)+1 609 613 0568fptr@novonordisk.com _______________________References1. Lam CSP, Contreras J, Darwesh R, et al. Prevalence and Predictors of High Inflammatory Risk in Heart Failure Subtypes: Findings From the Global POSEIDON Study. Eur J Heart Fail. 2026.2. Navar A, Bai L, Højen J, et al. Global Prevalence of Elevated High-Sensitivity C-Reactive Protein in Patients with Atherosclerotic Cardiovascular Disease, With and Without Chronic Kidney Disease: Findings From the POSEIDON Study. Late breaker oral presentation at the European Atherosclerosis Society 2026; May 24-27 2026; Athens, Greece.3. Ridker PM. Clinician’s Guide to Reducing Inflammation to Reduce Atherothrombotic Risk: JACC Review Topic of the Week. J Am Coll Cardiol. 2018;72(25):3320-3331.4. Ridker PM, Bhatt DL, Pradhan AD, et al. Inflammation and cholesterol as predictors of cardiovascular events among patients receiving statin therapy: a collaborative analysis of three randomised trials. Lancet. 2023;401(10384):1293-1301.5. Ridker PM, Lei L, Louie MJ, et al. Inflammation and Cholesterol as Predictors of Cardiovascular Events Among 13 970 Contemporary High-Risk Patients With Statin Intolerance. Circulation. 2024;149(1):28-35.6. FDA. Review Criteria for Assessment of C-Reactive Protein (CRP), High Sensitivity C-Reactive Protein (hsCRP) and Cardiac C-Reactive Assays. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/review-criteria-assessment-c-reactive-protein-crp-high-sensitivity-c-reactive-protein-hscrp-and Last accessed: May 2026.7. Peikert A, Kaier K, Merz J, et al. Residual inflammatory risk in coronary heart disease: incidence of elevated high-sensitive CRP in a real-world cohort. Clin Res Cardiol. 2020;109(3):315-323.8. Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. New England Journal of Medicine. 2005;3521685–1695.9. Lawler PR, Bhatt DL, Godoy LC, et al. Targeting cardiovascular inflammation: next steps in clinical translation. European Heart Journal. 2021;42113–131.10. Mesquita T, Lin YN, Ibrahim A. Chronic low-grade inflammation in heart failure with preserved ejection fraction. Aging Cell. 2021;20(9):e13453.11. Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;74(10):e177-e232.12. Visseren FLJ, Mach F, Smulders YM, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies With the special contribution of the European Association of Preventive Cardiology (EAPC). European Heart Journal. 2021;42(34):3227-3337.
Attachment
PR260526_POSEIDON-EAS
A single dose of Lilly’s PCSK9 base editor, VERVE-102, reduced PCSK9 by up to 88% and LDL-C by up to 62%, with durable effects supporting its potential as a one-time treatment for hypercholesterolemia
In the Phase 1b Heart-2 trial, a single intravenous infusion of VERVE-102 produced dose-dependent lowering of PCSK9 and LDL-C, with both reductions sustained over follow-up of up to 18 months in participants at high risk for cardiovascular disease VERVE-102 is designed to mimic the…
BioCardia Announces CardiAMP Chronic Myocardial Ischemia Trial Results Presented at EuroPCR Showed Durable Improvements in Exercise Tolerance with Reduced Angina Frequency
– Positive CardiAMP CMI Trial open-label cohort results demonstrated opportunity for locally delivered cell therapy to enhance therapeutic options for patients with severely symptomatic refractory angina and validate continued clinical development – Novel therapeutic approach targets a critically important unmet medical need for patients who have debilitated quality-of-life and have exhausted all available treatment options SUNNYVALE, Calif., May 21, 2026 (GLOBE NEWSWIRE) — BioCardia, Inc. (NASDAQ: BCDA), a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today announced the oral presentation of the results of the CardiAMP® Cell Therapy in Chronic Myocardial Ischemia Trial (CardiAMP CMI) preliminary open label cohort at EuroPCR, a leading interventional cardiovascular medicine conference held in Paris. The presentation was made by Dr. Amish Raval, M.D., Professor of Medicine at University of Wisconsin School of Medicine and Public Health. Results presented by Dr. Raval, on behalf of the CardiAMP CMI Investigators, showed that the minimally invasive CardiAMP Cell Therapy procedure was well tolerated with no treatment emergent major adverse cardiac events and patients demonstrated positive clinical outcomes including increased exercise tolerance and reduced frequency of angina episodes with the autologous cell therapy up through 2 years follow-up. Prior to treatment, all patients were on guideline-directed medical therapy (GDMT) and had received all available percutaneous and/or surgical options as appropriate for their medical condition. Patients experienced increased exercise tolerance, improving by an average of 179 seconds, which persisted for the two-year study follow-up. Angina episodes were reduced by an average of 82% by six-months after treatment. Dr. Raval’s presentation is here: EuroPCR2026. “Chronic ischemic heart disease results in considerable limitations of daily life activities due to chest discomfort, shortness of breath, and related disabling symptoms despite optimal medical therapy,” said Carl Pepine, MD, MACC, Professor of Medicine, Division of Cardiovascular Medicine, University of Florida at Gainesville. “A cell-based approach, added to this medical therapy, has potential to better manage these symptoms and improve the quality of life.” “We are thankful for the physician scientists who contributed their great experience to this program as well as the patients who participated,” said Peter Altman, PhD, BioCardia President and CEO. “These results support this CardiAMP cell therapy approach which has potential to help patients suffering from refractory angina. To our knowledge, only cell therapy has had a positive impact on both exercise tolerance and angina episodes in these patients.” About Chronic Myocardial Ischemia with Refractory Angina Chronic myocardial ischemia occurs in the setting of coronary artery disease when there is reduced blood flow to the heart. This causes angina, a type of chest pain which is characterized as refractory angina when this pain cannot be controlled by a combination of optimal medical therapy, angioplasty or bypass surgery, and is estimated to impact 600,000 to 1.8 million patients in the United States. Up to 15% of patients who have ischemia or angina and undergo cardiac catheterization are suboptimal candidates for conventional revascularization. Although prognosis of refractory angina has improved in recent years, patients with refractory angina experience a significantly impaired quality of life with disproportionately high utilization of healthcare services. These observations reflect the great need for new therapies for these patients. About the CardiAMP Cell Therapy Program Designated by the FDA as a Breakthrough Therapy for Ischemic Heart Failure, CardiAMP Cell Therapy uses a patient’s own bone marrow cells delivered to the heart in a minimally invasive, catheter-based procedure to potentially stimulate the body’s natural healing response. CardiAMP Cell Therapy incorporates three proprietary elements not previously utilized in investigational cardiac cell therapy: a pre-procedural cell analysis for patient selection, a high target dosage of cells, and a proprietary delivery system that has been shown to be safer than other intramyocardial delivery systems and exponentially more successful in cell retention. The CardiAMP cell therapy trials for the indications of both chronic myocardial ischemia and ischemic heart failure are covered by the Center for Medicare and Medicaid for both treatment and control procedures. CAUTION – Limited by United States law to investigational use. About BioCardia® BioCardia, Inc., headquartered in Sunnyvale, California, is a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary disease. CardiAMP® autologous and CardiALLO™ allogeneic cell therapies are the Company’s biotherapeutic platforms with three cardiac clinical stage product candidates in development. These therapies are enabled by its Helix™ biotherapeutic delivery and Morph® vascular navigation product platforms, and soon the Heart3D™ fusion imaging platform. BioCardia selectively partners on biotherapeutic delivery with peers developing important biologic therapies. For more information visit https://www.biocardia.com. Forward Looking Statements: This press release contains forward-looking statements that are subject to many risks and uncertainties. Forward-looking statements may include, among other things, statements relating to the continued development, ability to offset clinical costs utilizing Medicare reimbursement and the ultimate success of our clinical cell therapy programs. These forward-looking statements are made as of the date of this press release. We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. Factors that could cause or contribute to such differences include, but are not limited to, the Company’s liquidity position and its ability to raise additional funds, as well as the Company’s ability to successfully advance its clinical trials. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Form 10-K filed with the Securities and Exchange Commission on March 24, 2026, under the caption titled “Risk Factors,” and in our subsequently filed Quarterly Reports on Form 10-Q. BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law. Media Contact: Miranda Peto, Investor RelationsEmail: mpeto@BioCardia.com Phone: 650-226-0120 Investor Contact: David McClung, Chief Financial OfficerEmail: investors@BioCardia.comPhone: 650-226-0120
Reflow Medical Reports Six-Month Results from DEEPER CORONARY Study of Spur® Elute Stent
SAN CLEMENTE, Calif.–(BUSINESS WIRE)– #EuroPCR–Reflow Medical announces six-month DEEPER CORONARY study results for the Spur® Elute Sirolimus-Eluting Retrievable Stent System at EuroPCR 2026.
MemorialCare Heart & Vascular Institute Becomes First Hospital in Los Angeles County to Offer Cleerly AI-Powered Heart Disease Analysis, Quantifying Total Plaque Volume and Soft Plaque
Systemwide launch brings AI powered assessment to Coronary CTA’s to MemorialCare’s Long Beach Medical Center, Orange Coast Medical Center, and Saddleback Medical Center. FOUNTAIN VALLEY, Calif., May 20, 2026 /PRNewswire/ — MemorialCare today announced the systemwide implementation of…
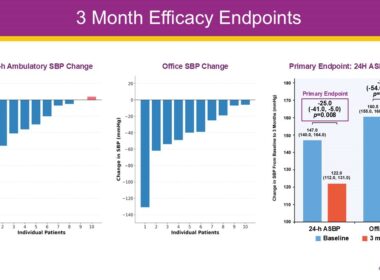
DeepQure Announces Robust 3-Month Results from HERO-HTN Clinical Study at EuroPCR 2026
PARIS, May 20, 2026 /PRNewswire/ — DeepQure, a clinical-stage medical device company, today announced robust 3-month clinical data from its HERO-HTN-FIH study during a featured presentation at EuroPCR 2026. The interim results demonstrate that the HyperQure™ extravascular renal…