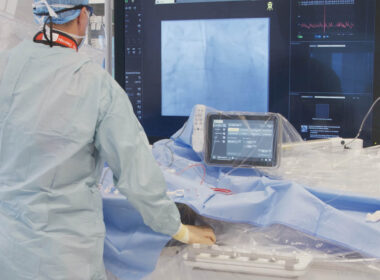
Philips Azurion with SmartIQ technology
Physicians are using Philips Azurion with SmartIQ technology during an medical procedure.
May 18, 2026 Greater differentiation of clinical details through contrast-rich images with reduced background noiseFor coronary procedures, a new ultra-low dose protocol employs over 50% less X-ray radiation dose [1]First SmartIQ clinical pilot study published in JSCAI, further building on Philips’ strong track record in evidence generation [2] Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced Philips SmartIQ, a breakthrough coronary imaging technology for its Azurion image-guided therapy. Developed in collaboration with leading cardiovascular centers, SmartIQ is designed to address one of the field’s most fundamental challenges: the trade-off between image quality and radiation exposure for patients and clinical staff. It aims to empower clinicians with exceptional image quality, enabling them to see exactly what they need, while it aims to provide reassurance that X-ray dose and contrast levels are optimized. “With this new imaging technology, we can go really low in dose and achieve even better image quality. It’s a win-win,” said Dr. Mark Winkens, Interventional Cardiologist at Elisabeth-TweeSteden Hospital (ETZ) in Tilburg, the Netherlands.Philips will showcase SmartIQ at EuroPCR 2026 (May 19–22, Paris, France), where attendees can experience the technology firsthand and hear directly from early clinical adopters. During dedicated clinical sessions, clinicians will share initial case experiences and present real-world multicenter results. Leveraging Philips’ strong track record in evidence generation, the first blinded clinical pilot study evaluating SmartIQ was recently published in Journal of the Society for Cardiovascular Angiography & Interventions. The study suggests that the algorithm scored higher on image quality and was preferred in the vast majority of blinded comparisons, while maintaining or lowering radiation and contrast dose. The technology builds on Philips’ industry-leading ClarityIQ platform, which has previously been associated with radiation exposure reductions between 23% and 83%, depending on the clinical area [3]. In parallel, further strengthening Philips’ evidence-generation efforts, the ongoing RADIQAL trial is designed to assess the impact of SmartIQ on radiation dose compared to ClarityIQ while maintaining procedural performance. The study has recently reached 60% of total enrollment and completed enrollment at European sites, marking an important milestone. Final study completion and results are expected in due course. SmartIQ represents the next step forward in image quality and dose management for coronary imaging. It includes an ultra-low dose protocol for coronary procedures that employs over 50% less X-ray radiation dose compared with the lowest dose settings available with Philips ClarityIQ [1]. This setting aims to enable acquisition of coronary cine runs at fluoroscopy dose levels, depending on system and clinical conditions [4]. “For too long, clinicians have had to choose between image quality and radiation dose during coronary procedures,” said Mark Stoffels, Business Leader Image-Guided Therapy Systems at Philips. “With our breakthrough SmartIQ technology, we believe that trade-off no longer has to define coronary imaging. This is not an incremental step forward – it represents one of Philips’ boldest advances yet in helping clinicians see what they need while aiming to further reduce exposure for patients and clinical teams.” “Image quality is the foundation of every decision we make in the cath lab. SmartIQ has genuinely surprised us. It delivers the clarity we need at very low radiation dose levels,” said Dr. Nicolaj Brejnholt Støttrup, Interventional Cardiologist at Aarhus University Hospital in Aarhus, Denmark. SmartIQ is the latest example of Philips’ long-term commitment to reducing radiation exposure across image-guided therapy. Together with innovations such as DoseAware, LumiGuide and EchoNavigator, it reflects a system-wide approach designed to help clinicians achieve the image quality they need while continuing the aim to reduce radiation exposure for patients and staff. SmartIQ is CE-marked and available in Europe and selected markets on both new and existing Azurion systems. The technology is not yet cleared for use in the United States. [1] Compared with the low ClarityIQ setting on Azurion systems, SmartIQ ultra-low left coronary 15 fps cine runs specify average reference air kerma reductions of 58% on Azurion M12 and 62% on Azurion M20 across all field sizes as stated in the IFU.[2] Assar et al., Exploring a Novel Processing Algorithm to Improve Image Quality and Reduce X-Ray and Contrast Dose for Coronary Angiography: A Blinded Pilot Study. Journal of the Society for Cardiovascular Angiography & Interventions, 2026, 105343[3] In 37 individual comparative studies, Philips ClarityIQ was associated with reductions in patient radiation exposure. The results of the application of dose reduction techniques will vary depending on the clinical task, patient size, anatomical location and clinical practice. The interventional radiologist assisted by a physicist as necessary has to determine the appropriate settings for each specific clinical task. See website: https://www.philips.com/clinicallyproven [4] The radiation dose reduction potential of SmartIQ is currently investigated in the RADIQAL trial. The RADIQAL trial is a multi-center randomized clinical trial to assess the impact of SmartIQ on radiation dose compared to the current ClarityIQ while maintaining procedural performance. This includes a comparison to the limitation of the reference air kerma rate in fluoroscopy of