Microbot Medical® Announces a Notable Health System as the First in Michigan to Adopt the LIBERTY® Endovascular Robotic System
Peripheral/Endo
InspireMD Announces First Patient Enrolled in the CGUARDIANS III Pivotal Study of the SwitchGuard Neuro Protection System for Use in TCAR Procedures
MIAMI, June 08, 2026 (GLOBE NEWSWIRE) — InspireMD, Inc. (Nasdaq: NSPR) (“InspireMD” or the “Company”), developer of the CGuard® Prime carotid stent system for the prevention of stroke, today announced that the first patient has been enrolled in the CGUARDIANS III pivotal study of its SwitchGuard neuro protection system (“NPS”), for use with its CGuard Prime 80 cm stent platform, in transcarotid artery revascularization (“TCAR”) procedures. The patient was enrolled by Dr. Patrick Muck at Good Samaritan Hospital, part of the TriHealth System in Cincinnati, Ohio.
Society for Vascular Surgery Annual Meeting to Unveil Cutting-Edge Research Shaping the Future of Vascular Care
International gathering of more than 1,400 vascular specialists will feature the latest data on limb-threatening diseases, novel technology and integration of specialty within larger healthcare system BOSTON, MA, MAY 27, 2026 – The Society for Vascular Surgery’s Vascular Annual Meeting (VAM) will commence on Wednesday, June 10, 2026, uniting the greatest […]
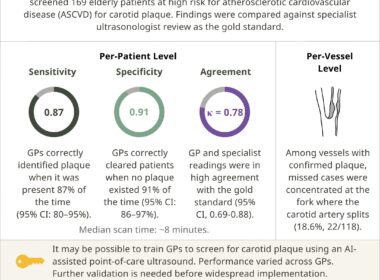
Annals of Family Medicine: Researchers in China Examine Use of AI-Assisted Ultrasound by General Practitioners To Detect Carotid Artery Plaque
PROVIDENCE, R.I., May 27, 2026 /PRNewswire/ — In a new feasibility study published in Annals of Family Medicine, general practitioners (GPs) in China were able to accurately detect carotid artery plaque — an indicator of cardiovascular disease — in high-risk patients using an AI-assisted…
Jupiter Endovascular Announces Positive Results From SPIRARE II Pivotal Trial at EuroPCR 2026
MENLO PARK, Calif.–(BUSINESS WIRE)– #TFX–Jupiter Endovascular announced positive results from its SPIRARE II pivotal trial at EuroPCR showing the trial met both of its primary endpoints.
Amplifi Vascular Announces Major Clinical Milestone by Launching the AMPLIFI-1 Pivotal Trial to Expand Forearm Fistula Options for Hemodialysis Patients
Study evaluates whether Amplifi therapy enables AVF maturation at two weeks, potentially reducing catheter dependence and transforming the dialysis access pathway BOSTON, May 19, 2026 /PRNewswire/ — Amplifi Vascular, a clinical-stage medical technology company developing the Amplifi™…
Endologix Acquires Pounce™ Thrombectomy System, Expanding Its Vascular Intervention Portfolio
SANTA ROSA, Calif.–(BUSINESS WIRE)–Endologix LLC, a privately held, global medical device company focused on advancing innovative therapies for the interventional treatment of vascular disease, today announced the acquisition of the Pounce™ Thrombectomy System from Surmodics, Inc. The transaction closed on May 18, 2026. The Pounce System is an FDA-cleared, fully mechanical thrombectomy system for the non-surgical removal of thrombi and emboli from peripheral arteries. Built around proprietary
FastWave Medical Secures Four New International Patents
Newly issued patents in Australia and Japan cover the company’s Artero™ E-IVL and Sola™ L-IVL platforms as FastWave advances toward peripheral and coronary IDE trials. MINNEAPOLIS, May 19, 2026 /PRNewswire/ — FastWave Medical has added four international patents to its intellectual…
DeepQure Reports Encouraging Clinical Data from World’s First Extravascular RDN System at AUA 2026
WASHINGTON, May 14, 2026 /PRNewswire/ — DeepQure, a clinical-stage medical device company, announced the completion of its featured presentation at the American Urological Association (AUA) Innovation Nexus Forum, held during the AUA Annual Meeting. DeepQure was selected to present the…
Solaris Endovascular Receives FDA Breakthrough Device Designation for Solaris DE™, a new Drug-Eluting Covered Stent
DALLAS, TX, UNITED STATES, May 5, 2026 /EINPresswire.com/ — Solaris Endovascular, Inc, a medical device company focused on next-generation endovascular therapies, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its first-in-class Solaris DE™ drug-eluting covered stent indicated for use in hemodialysis patients for […]