IRVINE, Calif., Oct. 09, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, announced today that it has entered into a Sponsored Research Agreement with Texas Heart Institute for the development of HJLI’s CoreoGraft®, an off […]
Peripheral/Endo
Eximo Medical Receives FDA Clearance for B-Laser™ Atherectomy System to Treat Peripheral Artery Disease (Including ISR)
REHOVOT, Israel–(BUSINESS WIRE)–Eximo Medical Ltd. announced today it has received 510(k) clearance from the U. S. Food & Drug Administration (FDA) for its B-Laser™ Atherectomy System for Peripheral Artery Disease (PAD). B-Laser™ is a transformative 355nm wavelength laser technology designed to address unmet clinical needs for treating multiple vascular indications. The specific indication cleared by the FDA is: […]
Intact Vascular Announces Presentation of TOBA II Pivotal Data
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced that data from its Tack Optimized Balloon Angioplasty (TOBA) II pivotal clinical trial will be presented at the 15th Annual VIVA Conference in Las Vegas, NV on November 5-8. The TOBA II trial […]
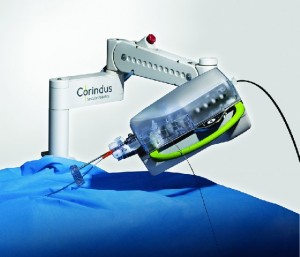
Corindus Technology Successfully used in Multiple Live Complex Robotic-Assisted Coronary Interventions at Transcatheter Cardiovascular Therapeutics 2018
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that its CorPath GRX System was used for multiple complex robotic-assisted percutaneous coronary interventions (PCI) at the recent Transcatheter Cardiovascular Therapeutics (TCT) 2018 Conference. In the first case at University of Washington […]
LeMaitre Vascular Acquires Clot Management Business from Applied Medical
BURLINGTON, Mass., Sept. 26, 2018 (GLOBE NEWSWIRE) — LeMaitre Vascular, Inc. (Nasdaq:LMAT) announced today that it has acquired the vascular clot management business of Applied Medical Resources Corporation for $14.2 million, of which $11.0 million was paid at closing and $3.2 million will be paid in two post-closing installments. As […]
Inari Medical Announces First Patient Enrolled in CLOUT Registry Using the ClotTriever Thrombectomy System
IRVINE, CALIFORNIA – September 27, 2018 — Inari Medical, Inc. announced today the enrollment of the first patient in the ClotTriever Outcomes Registry (“CLOUT”) using the ClotTriever® Thrombectomy System. CLOUT is a 500-patient prospective, multicenter, single-arm registry to evaluate real world outcomes after treatment of patients with thrombosis in the […]
Positive Data On BioCardia’s Investigational CardiAMP Cell Therapy For Heart Failure Presented At Transcatheter Cardiovascular Therapeutics (TCT) Conference
SAN DIEGO and SAN CARLOS, Calif., Sept. 24, 2018 /PRNewswire/ — BioCardia®, Inc. (OTC :BCDA ), a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today announced that positive data from two studies of its investigational CardiAMP stem cell therapy were presented today at the Transcatheter Cardiovascular Therapeutics (TCT) conference. CardiAMP cell therapy […]
Cook Medical Receives FDA Approval for First 5 mm Diameter SFA Drug-Eluting Stent
BLOOMINGTON, Ind.–(BUSINESS WIRE)–Cook Medical announced that a new 5 mm diameter version of Zilver® PTX® was approved by the FDA. It is the first 5 mm drug-eluting stent in the U.S. with lengths available up to 140 mm that is indicated to treat vessels as small as 4mm in diameter. The range […]
Hancock Jaffe Submits Application to INVIMA for First-In-Human VenoValve Trial in Colombia
IRVINE, Calif., Sept. 24, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, announced today that it has completed and submitted its application to INVIMA (the Colombian equivalent of the US FDA) for approval of its […]
Boston Scientific Receives U.S. FDA Approval for the Eluvia™ Drug-Eluting Vascular Stent System
MARLBOROUGH, Mass., Sept. 24, 2018 /PRNewswire/ — Today, Boston Scientific (NYSE :BSX ) announced that the U.S. Food and Drug Administration (FDA) has approved its Premarket Approval (PMA) application to market the Eluvia™Drug-Eluting Vascular Stent System, specifically developed for the treatment of peripheral artery disease (PAD). The Eluvia stent utilizes a drug-polymer combination to offer […]