Conference Focuses on Role of Digital Technology in Healthcare; David Fischel Named Panelist in Robotics Session ST. LOUIS, Nov. 22, 2019 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, announced today its Chairman and Chief Executive Officer, David Fischel, will present […]
Author: Ken Dropiewski
PAVmed Reports Third Quarter 2019 Financial Results and Provides Business Update
Conference call to be held on November 21, 2019 at 4:30 p.m. Eastern time NEW YORK, Nov. 21, 2019 (GLOBE NEWSWIRE) — PAVmed Inc. (Nasdaq: PAVM, PAVMZ) (the “Company” or “PAVmed”), a highly differentiated, multiproduct medical device company, today reported financial results for the three and nine months ended September […]
Neovasc Reducer™ Featured During Therapy Development Program in Coburg and Session at Maria Cecilia Hospital in Cotignola
VANCOUVER, CA, COBURG, DE, and COTIGNOLA, IT, Nov. 22, 2019 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, announced […]
Data Show Zilver® PTX® Leads to Fewer Complications and Shorter Hospital Stays Than Traditional Bypass Surgery
BLOOMINGTON, Ind.–(BUSINESS WIRE)–At this year’s VEITHsymposium®, Dr. Marc Bosiers presented data that show that patient treatment with the Zilver® PTX® stent has several benefits when compared to traditional bypass surgery. The data, which were gathered from a randomized controlled trial, show that treatment with Zilver PTX results in fewer complications and shorter […]
Study Uses Masimo Patient SafetyNet™ and rainbow Acoustic Monitoring® to Investigate the Incidence of Desaturation and Bradypnea in Postoperative Patients
NEUCHATEL, Switzerland–(BUSINESS WIRE)–Masimo (NASDAQ: MASI) announced today that in a study recently published in the Journal of Clinical Monitoring and Computing, researchers at Nippon Medical School in Tokyo, Japan used Masimo Patient SafetyNet™ and rainbow Acoustic Monitoring® (RAM®), acoustic respiration rate monitoring with RRa®, as a centralized continuous monitoring system to identify the […]
David Pacitti Elected to Siemens Foundation Board of Directors
WASHINGTON–(BUSINESS WIRE)–The Siemens Foundation today announced that David Pacitti, president of Siemens Medical Solutions USA, Inc. and head of the Americas for Siemens Healthineers, has been named to the Siemens Foundation Board of Directors. “The Siemens Foundation is focused on innovation, research and continuous learning to narrow the opportunity gap for young […]
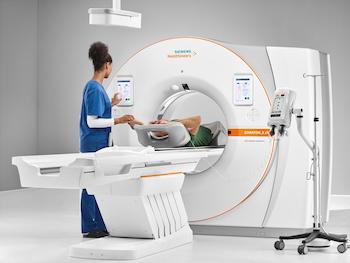
FDA Clears Siemens Healthineers SOMATOM X.cite CT Scanner With Intelligent User Interface Concept
myExam Companion intelligent user interface concept guides technologists through complex clinical tasks myExam Companion automatically adapts scans based on A.I.-trained clinical decision trees to simplify image acquisition parameters and reconstruction tasks SOMATOM X.cite improves patient comfort with an 82cm gantry bore and delivers powerful performance with Vectron tube MALVERN, Pa.–(BUSINESS […]
Medtronic Drug-Coated Balloon Receives U.S. FDA Approval to Treat Arteriovenous Fistula Lesions
Clinical Data Demonstrates IN.PACT™ AV DCB Is Safe, Reduces Reinterventions, and Maintains Access for End-Stage Renal Disease Patients Undergoing Dialysis DUBLIN, Nov. 21, 2019 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced U.S. Food and Drug Administration (FDA) approval of the IN.PACT™ AV drug-coated balloon (DCB), a paclitaxel-coated balloon indicated for the […]
Merit Medical Receives FDA Breakthrough Device Designation for WRAPSODY™ Endovascular Stent Graft System
SOUTH JORDAN, Utah, Nov. 21, 2019 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ: MMSI), a leading manufacturer and marketer of proprietary disposable devices used in interventional, diagnostic and therapeutic procedures, particularly in cardiology, radiology, oncology, critical care and endoscopy, announced today that it has been granted Breakthrough Device Designation […]
BioSig Commences Clinical Trial Enrollment for PURE EP System
Westport, CT, Nov. 21, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM) (“BioSig” or the “Company”), a medical technology company developing a proprietary biomedical signal processing platform designed to improve signal fidelity and uncover the full range of ECG and intra-cardiac signals, today announced that the Company commenced its […]