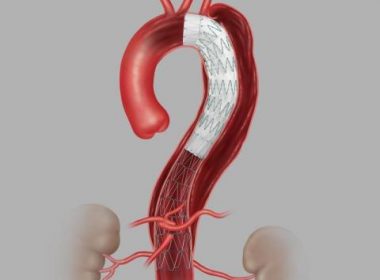
BLOOMINGTON, Ind.–(BUSINESS WIRE)–Today, Cook Medical announced its recent approval from the U.S. FDA for its Zenith Dissection Endovascular System. The system, consisting of a proximal stent-graft component and a distal bare stent component, provides physicians a less invasive alternative to open surgery for repair of Type B dissections of the […]
Author: Ken Dropiewski
First Atrial Fibrillation Patient Treated In Biosense Webster U.S. IDE Study Evaluating High Power, Short Duration Ablation Catheter
IRVINE, Calif., Feb. 4, 2019 /PRNewswire/ — Johnson & Johnson Medical Devices Companies* announced today that Biosense Webster, Inc., a worldwide leader in the diagnosis and treatment of heart arrhythmias, has enrolled and treated the first patient in its U.S. Investigational Device Exemption (IDE) study** which evaluates the company’s QDOT MICRO Radiofrequency […]
CHMP Recommends Approval of Praluent® (alirocumab) Injection to Reduce Cardiovascular Risk in People with Established Atherosclerotic Cardiovascular Disease
TARRYTOWN, N.Y. and PARIS, Feb. 4, 2019 /PRNewswire/ — Regeneron Pharmaceuticals, Inc. (NASDAQ: REGN) and Sanofi today announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion for Praluent® (alirocumab) Injection, recommending a new indication to reduce cardiovascular risk by lowering low-density lipoprotein cholesterol (LDL-C) levels as an […]
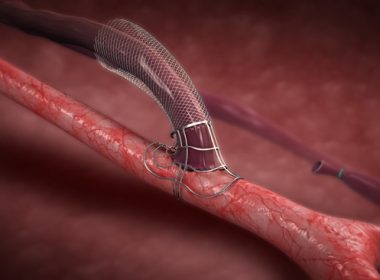
Ascyrus Medical Announces CE Mark Approval for the Ascyrus Medical Dissection Stent (AMDS)
BOCA RATON, FLA. (PRWEB) FEBRUARY 01, 2019 With approval, the AMDS offers physicians and patients in Europe the first marketed arch remodeling device and a breakthrough treatment for type A aortic dissections. Ascyrus Medical, a privately-owned medical device company focused on innovative treatments for aortic dissections, today announced the CE mark […]
CHF Solutions Announces Distribution Agreement in India
EDEN PRAIRIE, Minn., Jan. 31, 2019 (GLOBE NEWSWIRE) — CHF Solutions (Nasdaq: CHFS), today announced the initiation of a distribution agreement in the Indian market with Wayinia Lifesciences. This agreement expands the company’s sales outreach in Asia, while opening new access to one of the larger population centers in the […]
Altavant Sciences to Present Data Supporting Planned Phase 2 Clinical Program for Rodatristat Ethyl in Patients with Pulmonary Arterial Hypertension at PVRI World Congress
BASEL, Switzerland and DURHAM, N.C., Jan. 31, 2019 /PRNewswire/ — Altavant Sciences, a company in the Roivant family focused on clinical development of novel therapies for pulmonary arterial hypertension (PAH) and other indications, will present its latest nonclinical and clinical data at the Pulmonary Vascular Research Institute (PVRI) 13th Annual World Congress on Pulmonary Vascular Disease, […]
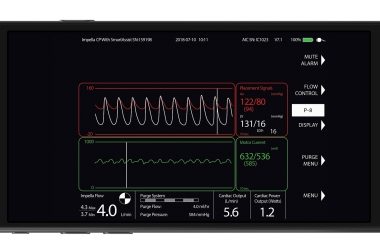
Impella Connect Achieves Regulatory Milestone
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed (NASDAQ:ABMD) has achieved CE Mark for Impella Connect, the first-of-its kind cloud-based technology that enables secure, real-time, remote viewing of the Impella console for physicians and hospital staff from anywhere with Internet connectivity. European CE Mark adds to Impella Connect’s previous U.S. FDA PMA approval. Impella Connect uses […]
Israeli Start-up Laminate, Developer of a Vascular Support Solution for Dialysis Patients, Has Completed a Capital Raising Round of $12 Million
TEL-AVIV, Israel, Jan. 31, 2019 /PRNewswire/ — The Israeli start-up Laminate Medical Technologies, developer of a vascular support device that is implanted in patients requiring dialysis, announces a capital raising round of $12 million. This round brings total investment in the company to date to $24 million. Participating in the current round is the leading dialyzer […]
Cardiovascular Systems, Inc. Reports Fiscal 2019 Second-Quarter Financial Results
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, today reported financial results for its fiscal second quarter, ended December 31, 2018. CSI’s second-quarter revenues were $60.2 million, an increase of […]
iSchemaView Introduces New Mobile and Notification Platform Features Across Its Family of RAPID Products
MENLO PARK, Calif.–(BUSINESS WIRE)–iSchemaView, the worldwide leader in advanced imaging for stroke, today announced significant upgrades to the RAPID APP as well as other messaging and notification capabilities. In its latest release, the world’s most powerful and feature-rich neuroimaging software, RAPID, now includes additional customization of notifications for hospitals and […]