TEL AVIV, Israel, Sept. 17, 2018 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE American: NSPR), developer of the CGuard™ Embolic Prevention System (EPS) for the prevention of stroke caused by the treatment of carotid artery disease, today announced that the United States Patent and Trademark Office (USPTO) has issued US Patents […]
Author: Ken Dropiewski
New Study Confirms Significant Potential U.S. Market for Hancock Jaffe’s VenoValve®
IRVINE, Calif., Sept. 17, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, has commissioned a research report from The Sage Group, a research organization specializing in peripheral vascular disease. The report, titled “Chronic Venous Disease: Epidemiology […]
First Patient in Europe Receives Implant of GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System and Enrolls in Post-Market Registry
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) today announced the first European patient implant of the GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System. This next-generation endovascular aneurysm repair (EVAR) device is indicated to treat the broadest range of abdominal aortic aneurysms (AAA) in patients with challenging anatomies. […]
Audio: Denis Harrington, CEO of NexGen Medical Systems speaks with CardiacVascularNews
Audio recording: Learn more about NexGen Medical System and the XCOIL System to remove blood clots from their CEO, Denis Harrington recorded at his office in Wayzata, MN.
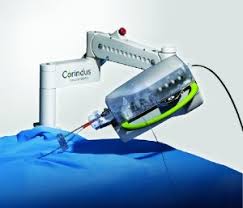
Corindus to Feature the CorPath® GRX System at the Transcatheter Cardiovascular Therapeutics 2018 Conference
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, will feature its CorPath® GRX System at the Transcatheter Cardiovascular Therapeutics (TCT) 2018 Conference, September 21 – 25, in San Diego, CA. Corindus’ CorPath GRX is the first FDA-cleared medical device for robotic-assisted vascular interventions. […]
Alnylam Announces Publication in Circulation of Exploratory Cardiac Endpoint Data from APOLLO Phase 3 Study of Patisiran
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Alnylam Pharmaceuticals, Inc. (Nasdaq: ALNY), the leading RNAi therapeutics company, announced today publication of data from exploratory cardiac assessments in the APOLLO Phase 3 study of patisiran, an RNAi therapeutic for the treatment of the polyneuropathy of hereditary transthyretin-mediated (hATTR) amyloidosis in adults. The results were published online today in the […]
FDA approves device for treatment of acute coronary artery perforations
SILVER SPRING, Md., Sept. 14, 2018 /PRNewswire/ — The U.S. Food and Drug Administration today approved a device intended to treat acute coronary artery perforations, or tears in the blood vessels of the heart. The PK Papyrus Covered Coronary Stent System is the first device approved by the FDA for this indication in […]
BioCardia Receives Two New U.S. Patents
SAN CARLOS, Calif., Sept. 14, 2018 /PRNewswire/ — BioCardia®, Inc. [OTC :BCDA ], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today announced the issuance of two United Statespatents. U.S. Patent No. 10,035,982 relates to methods of preparing culture-expanded cells for the treatment of heart failure. The patent has broad claims on […]
Endotronix Raises $45 Million in Series D Financing for the Treatment of Heart Failure
LISLE, Ill., Sept. 14, 2018 /PRNewswire/ — Endotronix, Inc., a digital health, medtech company dedicated to advancing the treatment of heart failure, today announced the close of a Series D financing round totaling $45 million. The round was led by LSP, investing from its LSP Health Economics Fund 2, and included Aperture Venture Partners, BioVentures Investors, Lumira Ventures, OSF […]
3 Ways Small Medical Device Companies Are Using Their MDET Savings… (Or Not)
Ken Dropiewski Principal Consultant Life Science/Medical Device Practice Executive Summary: In January 2018, President Trump issued an Executive Order suspending the medical device tax (2.3% on revenue) for an additional 24 months. Further, because the moratorium is retroactive to January 1, 2018 -when the tax did commence, medical device […]