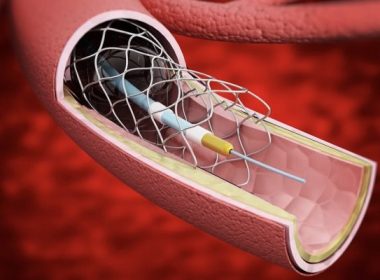
GPX is a biocompatible gel-particle embolic device that transforms from low-viscosity particles in a syringe to a solid particle after injection into a vessel LEIPZIG, GERMANY, February 01, 2018 /24-7PressRelease/ — Two scientific abstracts of a new vascular embolic device were presented this week at the 2018 Leipzig Interventional Congress […]
Author: Ken Dropiewski
Scandinavian Real Heart Selects AdaCore Embedded Software Development Platform for Revolutionary Artificial Heart
NEW YORK & PARIS & NUREMBERG, Germany–(BUSINESS WIRE)–Embedded World–AdaCore, a trusted provider of software development and verification tools with headquarters in New York and Paris, today announced that Scandinavian Real Heart AB in Sweden is using a suite of AdaCore software solutions to develop reliable embedded software for its revolutionary Total Artificial […]
Global Embolic Protection Devices Market 2018-2023 by Type, Material, Application, Indication – ResearchAndMarkets.com
DUBLIN–(BUSINESS WIRE)–The “Embolic Protection Devices Market by Type (Distal Filter, Distal Occlusion, Proximal Occlusion), Material (Nitinol, Polyurethane), Application (Cardiovascular, Neurovascular, Peripheral), Indication (PCI, SVD, TAVR) – Global Forecast to 2023” report has been added to ResearchAndMarkets.com’s offering. The Global Embolic Protection Devices Market is Projected to Reach USD 604.9 Million By 2023 from USD […]
Abbott and Surmodics Announce Agreement for Next-Generation Drug-Coated Balloon
ABBOTT PARK, Ill. and EDEN PRAIRIE, Minn., Feb. 27, 2018 /PRNewswire/ — Abbott (NYSE: ABT) and Surmodics (NASDAQ: SRDX) today announced that the companies have entered into an agreement whereby Abbott will have exclusive worldwide commercialization rights for Surmodics’ SurVeil® drug-coated balloon to treat the superficial femoral artery, which is currently being evaluated in a U.S. pivotal […]
CardioFocus Announces Intiation of HeartLight® X3 Clinical Evaluation
MARLBOROUGH, Mass., Feb. 26, 2018 /PRNewswire/ — CardioFocus, Inc., a medical device innovator and manufacturer dedicated to advancing ablation treatment for cardiac disorders such as atrial fibrillation (AF), today announced the initiation of a clinical evaluation of the new HeartLight X3 System for the treatment of AF. Building upon the advanced features of the […]
REVA ANNOUNCES CE MARK AND FIRST IMPLANT OF THE FANTOM ENCORE BIORESORBABLE SCAFFOLD
2.5 millimeter Diameter Size with Market-Leading 95 micron Strut Profile Secures Early Approval San Diego, California (Monday, February 26, 2018 – PST) – REVA Medical, Inc. (ASX: RVA) (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular applications, announces CE Mark of the 2.5 millimeter diameter size […]
Medtronic Launches First 2.0 mm Drug-Eluting Stent in United States Designed to Treat Patients with Small Vessels
FDA Approved, Resolute Onyx 2.0 mm DES Technology Tackles Clinical Challenge of Treating Coronary Artery Disease in Previously Untreatable Patients DUBLIN – February 26, 2018 – Designed specifically for small vessels, Medtronic plc (NYSE: MDT) today announced the U.S. Food and Drug Administration (FDA) approval and U.S. launch of the Resolute […]
Texas Cardiac Arrhythmia Institute at St. David’s Medical Center to host EPLive 2018
AUSTIN, Texas, Feb. 26, 2018 /PRNewswire/ — On March 1 and 2, 2018, the Texas Cardiac Arrhythmia Institute (TCAI) at St. David’s Medical Center will host its fourth international symposium on complex arrhythmias, EPLive 2018. EPLive is an intensive, two-day educational meeting for practicing clinical cardiac electrophysiologists, electrophysiologist fellows and […]
NaviGate Cardiac Structures Inc. (“NCSI”) reports GATE™ bioprosthesis as the first transcatheter valve replacement in Canada to treat tricuspid regurgitation
LAKE FOREST, Calif.–(BUSINESS WIRE)–NaviGate Cardiac Structures Inc. (“NCSI”) announced today that on 2 Feb. 2018, its catheter-guided GATE™ valved-stent bioprosthesis beame the first Canadian orthotopic valve replacement to treat severe tricuspid regurgitation. The procedure was performed at the Quebec Heart and Lung Institute, Laval University (Quebec City, Canada) by the Institute’s […]
Vascular Graft Solutions: External Stenting of Vein Grafts is Associated With High Early Patency Rates
TEL AVIV, Israel, Feb. 23, 2018 /PRNewswire/ — Vascular Grafts Solutions announced today the interim results from the VEST III study, a post-marketing trial which evaluates the effectiveness of the VEST technology (Venous External SupporT) in treatment of saphenous vein graft (SVG) disease after coronary artery bypass grafting. The Interim […]