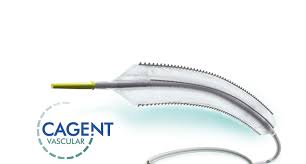
Feb. 7, 2018 14:30 UTC WAYNE, Pa.–(BUSINESS WIRE)– Cagent Vascular, a developer of next generation technology for vessel dilatation in cardiovascular interventions announces positive results from the First-in-Human PRELUDE Study. Dr. Andrew Holden (Auckland, New Zealand), Principal Investigator, presented 30-day and preliminary 6-month results using the FDA 510(k) cleared Serranator® Alto PTA Serration Balloon […]
Author: Ken Dropiewski
Global Vascular Grafts Market 2017-2025 by Type, Material, and Application – Market Expected to Grow at 4.9% CAGR due to the Rising Prevalence of CVD
DUBLIN, Feb. 7, 2018 /PRNewswire/ — The “Global Vascular Grafts Market Size, Market Share, Application Analysis, Regional Outlook, Growth Trends, Key Players, Competitive Strategies and Forecasts, 2017 to 2025” report has been added to ResearchAndMarkets.com’s offering. The global vascular grafts market was valued at US$ 2,352 million in 2016, and is expected to reach US$ 3,636.9 millionby 2025, expanding […]
JenaValve Raises $5 Million
JenaValve raises $5m FEBRUARY 6, 2018 BY FINK DENSFORD of MassDevice As first reported by Fink Densford of MassDevice: JenaValve Technology has raised $4.9 million in a new round of debt financing, according to an SEC filing posted this week. The Irvine, Calif.-based company is one of several smaller players looking to enter the […]
Access Scientific Receives Innovative Technology Contract from Vizient, Inc. For POWERWAND Extended Dwell Catheters
SAN DIEGO–(BUSINESS WIRE)– Access Scientific announced it has received an Innovative Technology contract from Vizient, Inc. the largest member-driven health care performance improvement company in the country, for their POWERWAND Extended Dwell Catheter (EDC). The contract was based on a recommendation of POWERWAND EDC by hospital experts in this category who serve on one […]
LimFlow Taps a New Chairman of the Board and Expands Senior Management Team
LimFlow Nominates Pierre Guyot as Chairman of Board of Directors and Expands Senior Management Team PARIS–(BUSINESS WIRE)– LimFlow SA, a Paris-based medical device company dedicated to transforming treatment options for Critical Limb Ischemia (CLI), announced today changes to their board of directors by naming Pierre Guyot as the new chairman of […]
BTG plc Release: OPTALYSE PE Long Term Results Reinforce Safety and Efficacy of Shorter, Lower Dose EKOS Therapy For Pulmonary Embolism
BOTHELL, Wash.–(BUSINESS WIRE)– BTG plc (LSE: BTG), the global healthcare company, today highlighted OPTALYSE PE one year trial results, presented at the International Society on Endovascular Therapy in Florida. The findings confirm that bilateral pulmonary embolism patients treated in as little as 2 hours with a total tissue plasminogen activator (tPA) dose […]
REVA Announces Appointment of Stephen Oesterle, MD to Board of Directors
SAN DIEGO, Feb. 05, 2018 (GLOBE NEWSWIRE) — REVA Medical (ASX:RVA) (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular applications, announces the appointment of Stephen Oesterle, MD to its Board of Directors, effective immediately. Dr. Oesterle brings significant clinical, medical device and board expertise to REVA. He is […]
Endologix, Inc. Completes Patient Enrollment in the ELEVATE IDE Clinical Study
IRVINE, Calif.–(BUSINESS WIRE)– Endologix Inc. (Nasdaq: ELGX), a developer and marketer of innovative treatments for aortic disorders, announced today the completion of enrollment in the Expanding Patient Applicability with Polymer Sealing Ovation Alto Stent Graft (ELEVATE) IDE clinical study. The objective of the 75-patient study is to evaluate the safety and effectiveness of […]
Teleflex Announces U.S. Launch of Two Next-Generation Versions of Its 0.035” Snares for Peripheral Procedures
Expro Elite™ Snare features a helical loop and Sympro Elite™ Snare features a simple 90-degree loop – both devices are available in five loop diameters for clinical versatility in retrieving or repositioning intravascular devices – preassembled, one-piece design allows rapid deployment through any 0.035” compatible lumen. WAYNE, Pa.–(BUSINESS WIRE)– Teleflex Incorporated (NYSE: […]
JenaValve Technology Appoints Laura A. Brenton as Vice President of Clinical and International Market Development
Feb. 2, 2018 14:00 UTC IRVINE, Calif.–(BUSINESS WIRE)– JenaValve Technology, Inc., a developer, manufacturer and marketer of differentiated transcatheter aortic valve replacement (TAVR) systems for the treatment of aortic valve disease, today announced the appointment of veteran medical device executive Laura A. Brenton as Vice President of Clinical and International Market […]