IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (NASDAQ:ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced financial results for the third quarter ended September 30, 2017. “Third quarter results were in line with our expectations, despite weather related disruptions,” commented John McDermott, Endologix’s Chief Executive Officer. “We are very […]
Author: Ken Dropiewski
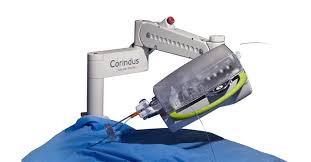
Corindus Vascular Robotics Reports Third Quarter 2017 Results
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, today reported financial results for the quarter ended September 30, 2017. Recent Highlights and Accomplishments Revenue of $2.4 million in the third quarter 2017 CorPath® GRX installed base increased to 23 systems with the installation […]
Surmodics Reports Fourth Quarter Fiscal 2017 Results and Provides Fiscal 2018 Guidance
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today announced results for its fiscal 2017 fourth quarter, ended September 30, 2017 and financial outlook for fiscal 2018. Recent Highlights First Patient Enrolled in TRANSCEND Pivotal Clinical […]
The Biggest Breakthrough Since Statins
A new study shows that anti-inflammatory injections may lower the risk of heart attacks. Researchers are saying this is the biggest cardiology breakthrough since statins. In the study, heart attack survivors that were given injections of the anti-inflammatory drug canakinumab had fewer attacks in the future. Incidentally, death from cancer decreased by […]
Clinical Trial Targets Devastating Heart Failure Diagnosis
CHICAGO, Nov. 6, 2017 /PRNewswire-USNewswire/ — Lynne Austin found she was increasingly out of breath doing even the simplest of tasks as 2015 came to a close. She couldn’t walk more than a few steps at a time. She couldn’t breathe. Her health was rapidly deteriorating and she wasn’t sure she would get […]
Boston Scientific To Participate In 26th Annual Credit Suisse Healthcare Conference
MARLBOROUGH, Mass., Nov. 6, 2017 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) will participate in the 26th Annual Credit Suisse Healthcare Conference on November 7, 2017 in Scottsdale, Arizona. Joe Fitzgerald, executive vice president & president, rhythm management, and Susie Lisa, vice president, investor relations, will participate in a 30-minute presentation and question-and-answer session with the host […]
CryoLife Announces Enrollment of First Patients in BioGlue Clinical Trial in China
ATLANTA, Nov. 6, 2017 /PRNewswire/ — CryoLife, Inc. (NYSE: CRY),a leading medical device and tissue processing company focused on cardiac and vascular surgery, announced today that enrollment has started in the Company’s BioGlue clinical trial in China. The results from the trial will serve as the basis of the Company’s regulatory submission […]
Vesalio Announces NeVa™ Stroke Treatment Product CE Mark Certification
Nashville, TN, October 31, 2017 –(PR.com)– Vesalio announces the CE Mark certification of its first product line, the proprietary NeVa™ neurothrombectomy platform for the treatment of strokes. Developed by a team of physicians that treat stroke, the NeVa mechanical thrombectomy technology was designed to improve “first pass” success, by effectively […]
HeartFlow Announces Decision by Centers for Medicare & Medicaid Services to Assign a New Technology Payment Classification to HeartFlow® FFRct Analysis
REDWOOD CITY, Calif.–(BUSINESS WIRE)–HeartFlow, Inc. today announced that the Centers for Medicare & Medicaid Services (CMS) has finalized a New Technology Ambulatory Payment Classification (APC) for the HeartFlow® FFRct Analysis, a first-of-its-kind non-invasive technology that helps clinicians diagnose and treat patients with suspected coronary artery disease (CAD). Under the APC payment system, […]
ABBOTT’S MITRACLIP APPROVED AS FIRST TRANSCATHETER MITRAL VALVE REPAIR DEVICE IN JAPAN
ABBOTT PARK, Ill., Nov. 6, 2017 /PRNewswire/ — Abbott (NYSE: ABT) today announced that Japan’s Ministry of Health, Labour and Welfare (MHLW) has approved the company’s MitraClip device for treatment of people with mitral regurgitation (MR), a serious, progressive heart disease in which the mitral valve does not close properly, allowing blood to flow backward […]