Study Evaluates Patients With Kidney Disease After Undergoing TAVR By Cardiac Interventions Today October 16, 2017—The American College of Cardiology (ACC) announced the publication of a study demonstrating a relatively low rate of patients with chronic kidney disease undergoing transcatheter aortic valve replacement (TAVR) who eventually need to start dialysis. […]
Author: Ken Dropiewski
REVA Announces Commercial Expansion and the First Implant of the Fantom Bioresorbable Scaffold in Switzerland
SAN DIEGO, Oct. 16, 2017 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX:RVA) (“REVA” or the “Company”) announced today the commercial expansion of its bioresorbable scaffold (“BRS”), Fantom®, with the first implant in Switzerland. The first implant of Fantom in Switzerland was conducted at Kantonsspital Baselland in Liestal, Switzerland by Dr. Gregor Leibundgut. “Following my […]
Vivasure Enrolls First Patient in Frontier IV Clinical Trial
GALWAY, Ireland–(BUSINESS WIRE)–Vivasure Medical® (“Vivasure”) is pleased to announce the successful enrollment of the first patient in the Frontier IV clinical study, a non-randomized multicenter international trial, designed to expand the indications of its proprietary PerQseal® large arteriotomy closure technology. The patient was enrolled by Dr. Peter Crean at the […]
BIOLIFE4D Appoints Texas Heart Institute Surgeon Dr. Jeffrey Morgan Chief Medical Officer, Bolstering Leadership Team
CHICAGO, Oct. 13, 2017 /PRNewswire/ — Bioprinting startup BIOLIFE4D named accomplished thoracic and cardiac surgeon Jeffrey Morgan M.D., FACS, FACC as Chief Medical Officer, further bolstering its world-class leadership team. BIOLIFE4D is a pioneering biotech company focused on leveraging advances in life sciences and tissue engineering to 3D bioprint a viable human heart suitable for transplant. Joining […]
Boston Scientific presents 3 year DES MAJESTIC data
Boston Scientific shared the 3-year MAJESTIC DES data online at CardioVascular and Interventional Radiology MAJESTIC 3-Year Data Published for Boston Scientific’s Eluvia DES, by Endovascular Today October 11, 2017—The 3-year results of the MAJESTIC first-in-human study of the Eluvia paclitaxel-eluting vascular stent system (Boston Scientific) for treating femoropopliteal artery lesions were published […]
Endologix, Inc. to Announce Third Quarter 2017 Financial Results on November 7, 2017
IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (Nasdaq: ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced that it will report financial results for the third quarter of fiscal year 2017 after the market close on Tuesday, November 7, 2017. The Company’s management will host a conference call at 4:30 […]
Elixir Medical Corporation to Unveil Game-Changing DynamXTM Metallic Stent at the 29th Transcatheter Cardiovascular Therapeutics Conference
MILPITAS, Calif.–(BUSINESS WIRE)–Elixir Medical Corporation, a leader in the development of breakthrough adaptive remodeling technologies designed to mimic the normal arterial function after cardiovascular and peripheral vascular disease intervention, announced today it will unveil a game-changing metallic drug eluting stent (DES) platform at this year’s Transcatheter Cardiovascular Therapeutics (TCT) conference […]
Silk Road Medical Appoints World Class Stroke Prevention Champion to Serve as Vice President, Medical Affairs and Professional Education
SUNNYVALE, Calif., Oct. 11, 2017 /PRNewswire/ — Silk Road Medical, Inc., a company dedicated to preventing the devastating burden of stroke through surgical innovation, announces the appointment of Tammy Leitsinger to the position of Vice President, Medial Affairs and Professional Education. In this new role, Tammy will further develop and refine global, peer-to-peer TCAR […]
CryoLife Announces Definitive Agreement to Acquire JOTEC
ATLANTA, Oct. 10, 2017 /PRNewswire/ — CryoLife, Inc. (“CryoLife”; NYSE: CRY), a leading medical device and tissue processing company focused on cardiac and vascular surgery, announced today that it has entered into a definitive agreement to acquire JOTEC AG (“JOTEC”). JOTEC is a German-based, privately-held developer of technologically differentiated endovascular stent grafts, […]
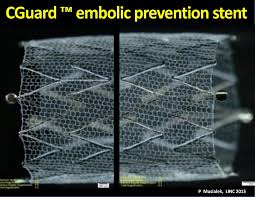
InspireMD Announces The Start Of Patient Enrollment In An Investigator Initiated Trial Of Cguard EPS In Russia
TEL AVIV, ISRAEL–(Marketwired – October 09, 2017) – InspireMD, Inc. (NYSE American: NSPR) (NYSE MKT: NSPR), a leader in Embolic Prevention Systems (EPS) / thrombus management technologies and neurovascular devices, today announced the start of patient enrollment in an investigator initiated trial in Russia, entitled: Independent Randomized Trial in Carotid Artery Revascularization Comparing […]