NEW YORK, Sept. 13, 2017 (GLOBE NEWSWIRE) — Delcath Systems, Inc. (NASDAQ:DCTH), an interventional oncology company focused on the treatment of primary and metastatic liver cancers, announces that its Board of Directors has approved the voluntary delisting from NASDAQ. As a result of the shareholder non-approval of its reverse stock […]
Author: Ken Dropiewski
Gecko Biomedical receives CE Mark Approval for SETALUM™ Sealant
Approval of CE Mark paves the way for application expansion and the exploration of new therapeutic areas for ground-breaking surgical solutions. Paris, France, September 11, 2017 – Gecko Biomedical (“Gecko”), a medical device company developing innovative polymers to support tissue reconstruction, announced today that it has received CE Mark approval […]
Boston Scientific to Establish Regional Manufacturing Hub in Malaysia
PENANG, 11 April 2016 – Expanding its manufacturing footprint in the Asia Pacific region, Boston Scientific Corporation (NYSE: BSX) today marked the commencement of construction work on a regional manufacturing hub during a groundbreaking ceremony in Penang. Located at the Batu Kawan Industrial Park, the completion of the facility is planned […]
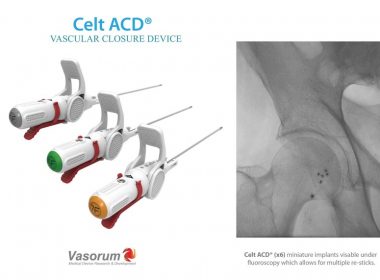
Vasorum Launches Celt ACD® Second Generation Vascular Closure Device in the USA
DUBLIN, Ireland, September 12, 2017 /PRNewswire/ — Vasorum Ltd, following FDA approval of its PMA supplement, the developer and manufacturer of the novel Celt ACD®vascular closure device has added a 7F sized Celt ACD® device to its Celt ACD® 6F and Celt ACD® 5F size range in the USA. Celt ACD® is indicated for arterial puncture closure in […]
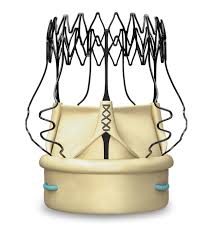
Neovasc Provides Tiara™ Clinical Update
VANCOUVER, Sept. 12, 2017 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company“) (NASDAQ, TSX: NVCN) today provided an update on the study progress and clinical performance of the TiaraTM valve, a self-expanding mitral bioprosthesis for transcatheter implantation in patients with Mitral Regurgitation (“MR”), one of the most prevalent valvular heart diseases in western […]
LivaNova Announces Publication of Literature Review Highlighting the Clinical Value and Safety of the Perceval Sutureless Valve for Aortic Valve Replacement
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN) (“LivaNova” or the “Company”), a market-leading medical technology company, today announced the publication of the analysis, “The Perceval® Sutureless Aortic Valve: Review of Outcomes, Complications and Future Direction,” by Powell, Pelletier, Chu, Bouchard, Melvin and Adams in the latest issue of Innovations1. The literature review, which included […]
Corindus Announces First Patient Enrolled in PRECISION GRX Registry
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. [NYSE American: CVRS], a leading developer of precision vascular robotics, today announced first patient enrollment in the PRECISION GRX Registry, a post-market study to continue market surveillance of the Company’s second generation CorPath GRX System. The PRECISION GRX Registry will include up to 25 sites […]
AtriCure Announces U.S. Launch of the AtriClip® PRO•V™ Device
MASON, Ohio–(BUSINESS WIRE)–AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in treatments for atrial fibrillation (Afib) and left atrial appendage management, today announced that it has launched the AtriClip PRO•V™ Left Atrial Appendage (LAA) Exclusion System in the United States. The new device offers an open-ended design combined with a tip-first closure […]
PQ Bypass to Unveil New Data From DETOUR I at VIVA 17
SUNNYVALE, Calif.–(BUSINESS WIRE)–PQ Bypass today announced that data from a subset analysis of the DETOUR I clinical trial will be presented during a late-breaking clinical trial session at Vascular InterVentional Advances (VIVA 17), taking place September 11-14 in Las Vegas. The study evaluated the safety and effectiveness of the DETOUR […]
Endologix Announces Collaboration Agreements with Japan Lifeline for the Development and Commercialization of Thoracic Endovascular Systems in Japan
IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (NASDAQ: ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced that it has signed a joint research and development agreement, as well as an exclusive distribution agreement, with Japan Lifeline Co., Ltd. (“JLL”) pertaining to the development and distribution of novel endovascular […]