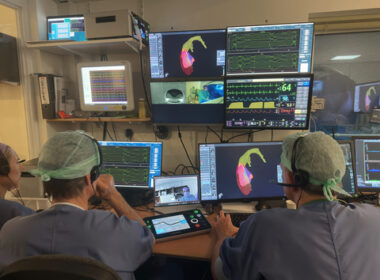
Nanosecond PFA 360° Cardiac Catheter for AF ablation to be highlighted in presentations and in a live case transmission HAYWARD, Calif.–(BUSINESS WIRE)–Pulse Biosciences, Inc. (Nasdaq: PLSE), a company leveraging its novel and proprietary Nanosecond Pulsed Field Ablation™ (nanosecond PFA or nsPFA™) technology, today announced that its Nanosecond PFA technology will […]
Author: Ken Dropiewski
Imricor Commences VISABL-VT Trial
MINNEAPOLIS–(BUSINESS WIRE)–Imricor Medical Systems, Inc. (Company or Imricor) (ASX: IMR) is pleased to announce that it has commenced the VISABL-VT clinical trial by completing the first-in-human ventricular ablation guided by real-time MRI with the Company’s NorthStar Mapping System. The procedure was performed by the team at the Amsterdam University Medical Centre (AUMC), ranked […]
PrepMD OMNI: The Most Comprehensive CIED Software Solution, Now Listed in the Epic Showroom
BRAINTREE, Mass., April 23, 2025 /PRNewswire/ — PrepMD OMNI’s inclusion in the Epic Showroom establishes it as a trusted, integrated solution within one of the nation’s leading EHR ecosystems. This visibility gives hospitals and cardiac care centers across the country easier access to…
FLEX Vessel Prep™ System FLEX FIRST AV Registry 6-Month Data and the Hamburg Vessel Prep Experience Prior to Angioplasty presented at Charing Cross Symposium
MINNEAPOLIS, April 23, 2025 /PRNewswire/ — VentureMed Group, Inc., a privately held leader in medical device innovations for arteriovenous (AV) access and peripheral arterial disease (PAD), announced data presented at the Charing Cross Symposium, April 23 – 25th, London, England….
Orchestra BioMed Announces AVIM Therapy-Focused Satellite Symposium at HRS 2025 Annual Meeting
NEW HOPE, Pa., April 23, 2025 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO, “Orchestra BioMed” or the “Company”), a biomedical company accelerating high-impact technologies to patients through risk-reward sharing partnerships, today announced it will host an industry-sponsored satellite symposium at the Heart Rhythm Society (“HRS”) 2025 Annual Meeting, taking place April 24–27, 2025, in San Diego, California featuring recent advancements in the Company’s atrioventricular interval modulation (“AVIM”) therapy program. The April 25th, 6:45 am PT symposium titled “The Future of Cardiac Pacing: Unlocking the Potential of Atrioventricular Interval Modulation (AVIM) Therapy” will convene leading electrophysiologists, hypertension and heart failure specialists to discuss the unmet need in hypertension, AVIM therapy mechanism of action, and growing body of clinical evidence supporting this novel therapy for the treatment of patients with uncontrolled hypertension who have increased cardiovascular risk with or without an indication for a pacemaker.
THE HEART RHYTHM SOCIETY ANNOUNCES FRAMEWORK FOR ESTABLISHING ATRIAL FIBRILLATION CENTERS OF EXCELLENCE AND KEY OPERATIONAL STANDARDS
WASHINGTON, April 23, 2025 /PRNewswire/ — Today, the Heart Rhythm Society (HRS) released a framework outlining criteria for establishing an Atrial Fibrillation (AF) Center of Excellence (CoE) and key operational standards to provide multidisciplinary care for AF patients. AF, the most…
Conavi Medical Corp. Announces Closing of $20M Public Offering
NOT FOR DISTRIBUTION TO U.S. NEWS WIRE SERVICES OR FOR DISSEMINATION IN THE UNITED STATES TORONTO, April 23, 2025 (GLOBE NEWSWIRE) — Conavi Medical Corp. (TSXV: CNVI; OTC: CNVIF) (“Conavi” or the “Company”), a commercial stage medical device company focused on designing, manufacturing, and marketing imaging technologies to guide common minimally invasive cardiovascular procedures, is pleased to announce that it has closed its previously announced, upsized equity offering for aggregate gross proceeds of $20 million (the “Offering”). The net proceeds from the Offering will be used to advance and complete the development and pre-clinical testing of its Novasight 3.0 technology, with the goal of submitting a 510(k) clearance application to the U.S. Food and Drug Administration in Q3 of 2025. The Company also intends to use the net proceeds for working capital and other general corporate purposes. Bloom Burton Securities Inc. acted as sole and exclusive agent for the Offering. Under the Offering, subscribers either purchased common shares at $0.40 per common share (the “Common Shares”) or pre-funded common share purchase warrants for $0.39999 per pre-funded common share purchase warrant (“Pre-Funded Warrants” and, together with the Common Shares, the “Securities”). Investors purchased a total of 50,000,000 Securities (consisting of 32,500,000 Common Shares and 17,500,000 Pre-Funded Warrants) for gross proceeds of $20 million. Each Pre-Funded Warrant issued in lieu of a Common Share at the election of a subscriber entitles the holder thereof to acquire one Common Share at an exercise price of $0.00001 per Common Share. The Pre-Funded Warrants will not expire. In Canada, the Securities purchased pursuant to the Offering were qualified for sale by way of a short form prospectus dated April 15, 2025, which was filed in British Columbia, Alberta and Ontario. The Securities were purchased by way of private placement in the United States, pursuant to exemptions from the registration requirements under the U.S. Securities Act of 1933 (the “U.S. Securities Act”), and pursuant to all applicable U.S. state securities laws. In addition, the Securities were also sold by way of private placement in certain other jurisdictions outside of Canada and the United States pursuant to and in compliance with applicable securities laws. CPOINT Capital Corp., an insider of the Company, purchased 625,000 Common Shares under the Offering and Juno Pharmaceuticals LP, an insider of the Company, purchased 1,250,000 Common Shares under the Offering. The subscriptions for Common Shares by CPOINT Capital Corp. and Juno Pharmaceuticals LP are related party transactions within the meaning of applicable Canadian securities laws. The subscriptions by such insiders are exempt from the formal valuation and minority approval requirements applicable to related party transactions on the basis that the value of the transactions insofar as they involve related parties is less than 25% of the Company’s market capitalization. The Board of Directors of the Company has approved the Offering. A material change report in respect of the related party transactions could not be filed earlier than 21 days prior to the closing of the Offering due to the limited time between the commitment by such insiders to purchase the subject Common Shares and the closing of the Offering. The securities described herein have not been, and will not be, registered under the U.S. Securities Act, or any U.S. state securities laws, and accordingly, may not be offered or sold to, or for the account or benefit of, persons in the United States or to U.S. Persons (as such terms are defined in Regulation S under the U.S. Securities Act), except in compliance with the registration requirements of the U.S. Securities Act and applicable U.S. state securities requirements or pursuant to exemptions therefrom. This press release does not constitute an offer to sell or a solicitation of an offer to buy any of the Company’s securities. About Conavi Medical Conavi Medical is focused on designing, manufacturing, and marketing imaging technologies to guide common minimally invasive cardiovascular procedures. Its patented Novasight Hybrid™ System is the first system to combine both intravascular ultrasound (IVUS) and optical coherence tomography (OCT) to enable simultaneous and co-registered imaging of coronary arteries. The Novasight Hybrid System has 510(k) clearance from the U.S. Food and Drug Administration; and regulatory approval for clinical use from Health Canada, China’s National Medical Products Administration, and Japan’s Ministry of Health, Labor and Welfare. For more information, visit http://www.conavi.com/. Notice on forward-looking statements: This press release includes forward-looking information or forward-looking statements within the meaning of applicable securities laws regarding Conavi and its business, which may include, but are not limited to, statements with respect to the anticipated use of proceeds from the Offering. All statements that are, or information which is, not historical facts, including without limitation, statements regarding future estimates, plans, programs, forecasts, projections, objectives, assumptions, expectations or beliefs of future performance, are “forward-looking information or statements”. Often but not always, forward-looking information or statements can be identified by the use of words such as “shall”, “intends”, “anticipate”, “believe”, “plan”, “expect”, “intend”, “estimate” “anticipate” or any variations (including negative variations) of such words and phrases, or state that certain actions, events or results “may”, “might”, “can”, “could”, “would” or “will” be taken, occur, lead to, result in, or, be achieved. Such statements are based on the current expectations and views of future events of the management of the Company. They are based on assumptions and subject to risks and uncertainties. Although management believes that the assumptions underlying these statements are reasonable, they may prove to be incorrect. The forward-looking events and circumstances discussed in this release, may not occur and could differ materially as a result of known and unknown risk factors and uncertainties affecting the Company, including, without limitation, those listed in the “Risk Factors” section of the short form prospectus dated April 15, 2025 and the joint information circular of the Company dated August 30, 2024 (both of which are on the Company’s profile at www.sedarplus.ca). Although Conavi has attempted to identify important factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements, there may be other factors that cause actions, events or results to differ from those anticipated, estimated or intended. Accordingly, readers should not place undue reliance on any forward-looking statements or information. No forward-looking statement can be guaranteed. Except as required by applicable securities laws, forward-looking statements speak only as of the date on which they are made and Conavi does not undertake any obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise. No regulatory authority has approved or disapproved the content of this press release. Neither the TSX Venture Exchange nor its Regulatory Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this press release. CONTACT: CONTACT:
Stefano Picone
Chief Financial Officer
(416) 483-0100
FIELD MEDICAL ANNOUNCES FIRST-IN-HUMAN DATA FOR FOCAL PFA IN SCAR-RELATED VT AT HEART RHYTHM 2025
Eight presentations, including late-breaking science, live cases, and a landmark symposium CARDIFF-BY-THE-SEA, Calif., April 23, 2025 /PRNewswire/ — Field Medical Inc., a leader in cardiac pulsed field ablation (PFA) technology, announced today its FieldForce™ Ablation System will be…
Catheter Precision to Acquire Assets in Development for Late-Stage Treatment for Acute Decompensated Heart Failure
Initial Clinical Studies Completed with Significant Positive Results
Kestra Medical Technologies to Showcase Innovation in Sudden Cardiac Arrest Protection and Recovery at Heart Rhythm 2025
Company to spotlight ASSURE WCD performance and expanded clinical applicationsKIRKLAND, Wash., April 23, 2025 (GLOBE NEWSWIRE) — Kestra Medical Technologies, Ltd. (Nasdaq: KMTS) (“Kestra”), a wearable medical device and digital healthcare company, announced today it will exhibit at Heart Rhythm 2025, the annual meeting of the Heart Rhythm Society (HRS), taking place April 24-27 at the San Diego Convention Center. This marks Kestra’s first major industry showcase following its successful IPO earlier this year. Kestra will debut an immersive in-booth experience designed to bring the ASSURE® system to life—demonstrating how this innovative technology is redefining protection for patients at risk of sudden cardiac arrest. By combining lifesaving defibrillation therapy with intuitive, intelligent, and connected diagnostic and patient support capabilities, the ASSURE system is a key part of a broader vision for a holistic cardiac care ecosystem that supports patients and providers across the recovery journey. “Our presence at Heart Rhythm 2025 comes at a pivotal time for Kestra,” said Brian Webster, President and Chief Executive Officer of Kestra. “Following our IPO, we’re moving forward with increasing momentum—and this year’s HRS meeting is an opportunity to demonstrate how the ASSURE system goes beyond protection to offer a smarter, more connected recovery experience for both patients and care teams.” In addition to exhibiting, Kestra will also present new real-world clinical data highlighting the impact of the ASSURE system. Kestra is also proud to sponsor the Women in EP Luncheon for the fourth consecutive year—underscoring its ongoing commitment to leadership, innovation, and equity in cardiovascular care. About KestraKestra Medical Technologies, Ltd. is a commercial-stage wearable medical device and digital healthcare company focused on transforming patient outcomes in cardiovascular disease using monitoring and therapeutic intervention technologies that are intuitive, intelligent, and connected. For more information, please visit www.kestramedical.com. Media contactRhiannon Pickusrhiannon.pickus@kestramedical.com Investor contactNeil Bhalodkarneil.bhalodkar@kestramedical.com