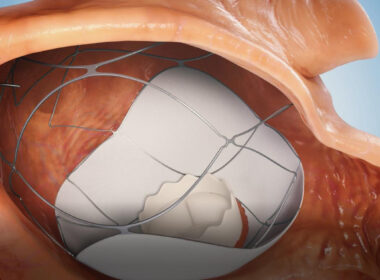
AllMeD Solutions (TASE:ALMD) subsidiary TruLeaf Medical receives Helsinki Ethics Committee’s approval in Uzbekistan to conduct clinical trial in human subjects, paving the way for minimally invasive heart valve treatmentsCAESAREA, Israel, July 11, 2024 /PRNewswire/ — AllMeD Solutions announced subsidiary TruLeaf Medical’s receipt of the Helsinki Ethics Committee’s approval in Uzbekistan to conduct clinical trial in human subjects. As part of the trial, a prosthetic mitral valve will be implanted via two needle sticks in the groins in a two-stage catheterization procedure without the need for open-heart surgery (transcatheter mitral valve replacement, TMVR). The implantation of the innovative platform (the RoseDoc) developed by TruLeaf, which replaces the patient’s leaky heart valve, will be carried out in two stages. In the first stage, a docking station will be implanted in the left atrium, followed a few weeks later by implantation of an artificial ‘biological’ mitral valve prosthesis.
Continue Reading
Trueleaf’s Medical Device
Today, there are tens of millions of patients with severe, life-threatening mitral valve regurgitation (leaky valve) across the world. This leak causes heart failure, heart arrhythmia, brain strokes leading to high mortality. About 10% of the world’s population over the age of 75 suffer from a leaky mitral heart valve. In the U.S. alone there are about 4 million patients.
These patients experience a substantial decrease in their functional capacity manifesting, such as fatigue, shortness of breath on exertion with lower and lower exertion and arrhythmias, progressively impairing their daily routine. Today, the most effective treatment for these patients is complex open-heart surgery to repair or replace the leaky heart valve. However, it is only offered to about 2% of patients due to the high surgical risk.
The unique RoseDoc platform developed by TruLeaf is the first-of-its-kind technology allowing implantation of a biological bioprosthesis to replace the diseased valve through catheterization only. This ground-breaking procedure is minimally invasive, performed on a beating heart via two needle punctures without surgery or the use of a heart-lung machine. As such, it is associated with substantially lower risk compared to the traditional open-heart mitral valve surgery. As a result, millions of patients around the world, who until now were deemed inoperable, will be able to get receive a new valve and experience a significant improvement in their functional capacity, quality of life and life expectancy, allowing them to resume normal life.As part of the preparations for human implantations in clinical trials in Uzbekistan, TruLeaf conducted additional implantations in large animals with the participation of Dr. Horst Sievert, one of the world’s leading interventional cardiologists, who is expected to lead TruLeaf’s clinical trials.TruLeaf Medical, Ltd was founded in 2017 by three Israeli entrepreneurs – Benjamin Spencer, Nathaniel Benisho and the late Dr. Uri Rosenstein. Benjamin and Nathaniel’s played a seminal role in the development of the first ever transcatheter aortic bioprosthesis, the Sapien valve, initially within the Israeli company PVT, that was later acquired by the medical technology giant Edwards Lifesciences. Today, the aortic valve that Benjamin and Nathaniel developed saves thousands of lives of patients with aortic stenosis every year all over the world.Benjamin Spencer, TruLeaf Medical CEO, explains, “The main challenge with existing transcatheter TMVR technologies is achieving optimal anchoring of the valve prosthesis to the heart, given the complex anatomy and physiology of the native mitral valve. The RoseDoc TMVR platform is technically simple, safe, and has proven effective in long-term animal testing. Completely eliminating the leak prevents the progressive dilation of the heart, which otherwise worsens the leak in a vicious cycle, leading to further weakening of the heart muscle and intractable heart failure. Currently, patients with severe mitral valve leaks that are unresponsive to maximal medical treatment have no effective options. The vast majority of these patients are declined surgery due to prohibitive risk. The unique RoseDoc TMVR platform provides a potential lifeline for these patients.”Professor Oz Shapira, AllMeD Solutions CEO, adds, “As a heart surgeon who has performed hundreds of open-heart surgeries to treat leaky heart valves, the possibility of replacing the mitral valve through a simple and quick needle puncture operation is a true revolution that may offer a solution to millions who currently have no other option.””The first-in-human trial is both exciting and mission-critical for TruLeaf’s success. Given the outstanding results of the preclinical experiments, I am confident that TruLeaf’s innovative RoseDoc TMVR platform will perform exceptionally well in humans and eventually save countless lives of patients who currently have no alternatives. AllMeD Solutions will continue to demonstrate its ability to identify early-stage startups and leverage its vast knowledge, experience, and expertise in the med-tech space to lead these companies to engineering, clinical, and business success.”Photo: https://mma.prnewswire.com/media/2459133/AllMeD_Solutions.jpgLogo: https://mma.prnewswire.com/media/2459132/AllMeD_Solutions_Logo.jpgSOURCE AllMeD Solutions