World’s First AI to Detect 35 Cardiac Determinations, Including Heart Attack, Using a Reduced LeadsetPowered by this AI, the Pocket-Sized Kardia 12L ECG System Enables Faster, Easier Detection of Life-Threatening Cardiac ConditionsMOUNTAIN VIEW, Calif., June 25, 2024 /PRNewswire/ — AliveCor, the global leader in AI-powered cardiology, today announced the U.S. Food & Drug Administration (FDA) clearance and commercial launch of KAI™ 12L AI technology and the Kardia™ 12L ECG System. This is the world’s first AI that can detect life-threatening cardiac conditions, including heart attacks, using a reduced leadset. The Kardia 12L ECG System, featuring a game-changing patented technology, is the world’s first AI-powered handheld 12-lead electrocardiogram (ECG) system with a unique single-cable design.
Continue Reading
Kardia 12L ECG System Enables Faster, Easier Detection of Life-Threatening Cardiac Conditions Using Reduced Leadset
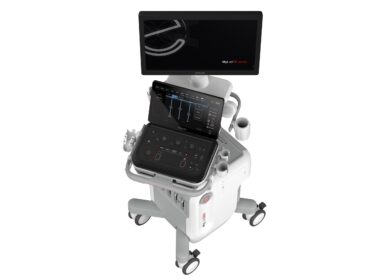
Pocket-Sized Kardia 12L ECG System
KAI 12L employs multiple deep neural network algorithms, trained and validated on more than 1.75 million ECGs from leading US medical centers. This AI technology can detect 35 cardiac determinations (14 arrhythmias and 21 morphologies), including serious conditions like acute myocardial infarction (MI) and the most common types of cardiac ischemia, using a reduced leadset. KAI 12L can seamlessly integrate with compatible devices, including the Kardia 12L ECG System, expanding critical access to accurate heart data within a broad range of healthcare settings.
“Not only is it the first FDA-cleared AI that can detect a heart attack on a reduced leadset, but it also returns determinations for our broadest range of conditions yet,” said Priya Abani, CEO of AliveCor. “Paired with our pocket-sized Kardia 12L ECG System, this offering is poised to disrupt traditional care pathways and represents a leap forward in cardiac care.”
Kardia 12L ECG System is an innovative solution that requires only a single cable with 5 electrodes to acquire 8 high quality diagnostic bandwidth leads. Conventional 12-lead ECG machines are the standard of care for detecting and diagnosing many heart conditions, but their availability can be limited due to their size and complexity. Using advanced KAI 12L and a reduced leadset, Kardia 12L offers outstanding clinical efficiency and performance substantially equivalent to state-of-the-art ECG analysis solutions. Its speed and simplicity assists healthcare providers in rapid disease detection and enhancing patient experience. “The AI innovations in the Kardia 12L ECG System offer healthcare providers a clinically-validated handheld 12-lead ECG system and expands this critical technology into more resource-limited settings,” said Dr. Stavros Stavrakis, MD, PhD, Professor of Medicine, Division of Cardiology at the University of Oklahoma Health Sciences Center. “By streamlining the process of recording a 12-lead ECG, Kardia 12L has significant implications for rapid ECG diagnosis in clinical practice.”Kardia 12L is battery-operated, weighs just 0.3 pounds and can fit in a pocket – making it significantly smaller, more portable and more convenient than conventional 12-lead ECG machines. Its streamlined leadset also makes it less invasive for patients, who do not need to fully disrobe during a reading. The device requires minimal self-guided training and is simpler to use than standard 12-lead ECG machines. These features put 12-lead ECG data within reach of more healthcare providers than ever before in a variety of healthcare facilities and acute settings, including primary and urgent care offices, employer clinics, and other under-resourced or rural venues.For healthcare providers interested in purchasing a Kardia 12L, please visit alivecor.com/products/kardia12L.About AliveCorAliveCor, Inc., the leading provider of FDA-cleared personal electrocardiogram (ECG) technology, is transforming cardiology with its medical-grade AI solutions. AliveCor is committed to providing innovative devices and services that empower patients and physicians with personalized and actionable heart data. With over 250 million ECGs recorded, the company’s Kardia devices are the most clinically validated personal ECGs in the world and can remotely detect six of the most common heart arrhythmias in just 30 seconds. The company’s latest offering, Kardia 12L ECG System, powered by KAI 12L to detect 35 cardiac conditions (14 arrhythmias and 21 morphologies), was designed exclusively for use by healthcare providers. AliveCor’s enterprise platform allows third-party providers to manage their patients’ and customers’ heart conditions simply using state-of-the-art tools that provide easy front-end and back-end integration to AliveCor technologies, addressing gaps in care and improving the treatment experience for patients across a range of disease areas. AliveCor is a privately held company headquartered in Mountain View, Calif. For more information, visit alivecor.com and follow us on LinkedIn, X, Instagram and Facebook.Media ContactsSu Leeslee191@alivecor.com(562) 544-4634Ben Rickles[email protected](404) 502-6766SOURCE AliveCor, Inc.