KIRKLAND, Wash.–(BUSINESS WIRE)–Prevencio, Inc., a leader in AI-powered blood tests for cardiovascular diagnostics, has been selected to join the 2024 Accelerator Cohort of MedTech Innovator, the world’s largest accelerator for medical technology companies. This honor places Prevencio among the top 5% of 1,300 applicants, reflecting the company’s innovative approach to […]
Author: Ken Dropiewski
AliveCor’s AI-backed ECG Offerings Featured in New Film Series on Innovative Technologies Presented by the Consumer Technology Association
The Human Component series, produced by BBC StoryWorks Commercial Productions, is now available to stream
MOUNTAIN VIEW, Calif., June 5, 2024 /PRNewswire/ — AliveCor, the leading innovator in FDA-cleared personal electrocardiogram (ECG) technology, today announced its participation in The Human Component, a new online branded series developed for the Consumer Technology Association (CTA) and produced by BBC StoryWorks Commercial Productions, the commercial content division of BBC Studios.
The series showcases innovative technologies that are supporting healthier communities, stronger societies and a more resilient planet. Through compelling, human-led films, The Human Component celebrates global efforts to maximize the full potential of technology.
“We are incredibly proud to take part in The Human Component alongside innovators who are just as committed to introducing game-changing ideas to improve lives as we are,” said Priya Abani, CEO of AliveCor. “The series beautifully depicts the real-life impacts of technologies like our AI-backed ECG solutions and services, and how they are helping transform society for the better.”
AliveCor’s film shares the story of Dr. Cleveland Francis, a cardiologist and musician who was diagnosed with a heart condition after collapsing at home. After his episode, Dr. Francis used one of AliveCor’s FDA-cleared Kardia™ devices to take a real-time, clinical-grade ECG that he shared with his doctor, leading to a definitive diagnosis. Capable of remotely identifying six of the most common arrhythmias in just 30 seconds, Kardia personal ECGs place the power of advanced AI technology directly into users’ hands. They also offer healthcare providers a way to capture patients’ elusive arrhythmias, contributing to earlier diagnoses and improving clinical decision-making.
This is the second series that BBC StoryWorks has created for CTA, a trade association that brings together companies that improve lives through technology. By highlighting how innovators across industries are advancing groundbreaking new technologies, The Human Component explores how we can solve some of the most pressing challenges facing the world today.
To learn more, watch AliveCor’s full film at alivecor.com/cleveland-cardiology-ai.
About AliveCorAliveCor, Inc., the leading provider of FDA-cleared personal electrocardiogram (ECG) technology, is transforming cardiology with its medical-grade AI solutions. AliveCor is committed to providing innovative devices and services that empower patients and physicians with personalized and actionable heart data. With over 250 million ECGs recorded, the company’s Kardia devices are the most clinically validated personal ECGs in the world and can remotely detect six of the most common heart arrhythmias in just 30 seconds. AliveCor’s enterprise platform allows third-party providers to manage their patients’ and customers’ heart conditions simply using state-of-the-art tools that provide easy front-end and back-end integration to AliveCor technologies, addressing gaps in care and improving the treatment experience for patients across a range of disease areas. AliveCor is a privately held company headquartered in Mountain View, Calif. For more information, visit alivecor.com and follow us on LinkedIn, X, Instagram and Facebook.
SOURCE AliveCor, Inc.
First Commercial Use of Baylis Medical Technologies’ PowerWire® Pro for In-Stent Restenosis
MISSISSAUGA, ON, June 5, 2024 /PRNewswire/ – Baylis Medical Technologies today announced the completion of its first clinical use of the PowerWire® Pro Radiofrequency (RF) Guidewire to safely cross a chronically occluded peripheral stent.
Continue Reading
Photograph of Dr. Iafrati and his team holding up a PowerWire® Pro box after their successful case. (CNW Group/Baylis Medical Technologies Inc.)
The procedure was performed in April 2024, by Mark Iafrati, MD, Professor of Vascular Surgery in Nashville, TN. The patient was suffering from a chronically occluded stent that extended from the inferior vena cava (IVC) down to the left common iliac vein. Previous attempts to cross the occlusion with mechanical tools were unsuccessful, likely owing to the dense fibrotic nature of the occlusion. The patient was brought back and Dr. Iafrati performed the procedure with the PowerWire Pro RF Guidewire, which utilizes RF energy to vaporize a channel through occlusions, allowing for successful crossing and subsequent revascularization of the iliocaval stent.
“This marks a significant advancement in the treatment of patients with in-stent restenosis”, commented Dr. Iafrati. “I was able to easily cross the occluded stents with the PowerWire Pro RF Guidewire when my standard tools had failed.”
This spring, the FDA cleared Baylis Medical Technologies’ PowerWire Pro RF Guidewire, a device designed to cross occluded peripheral vessels, including those with stents, using radiofrequency technology.Frank Baylis, Executive Chairman of Baylis Medical Technologies, expressed enthusiasm about the PowerWire® Pro RF Guidewire’s potential impact, stating, “This innovation aims to simplify the often challenging and time-consuming process of crossing chronic occlusions in revascularization procedures, ensuring uninterrupted patient treatment.”
About Baylis Medical Technologies Inc.
Baylis Medical Technologies seeks to improve the lives of patients through the conception and commercialization of state-of-the-art medical devices. The company proudly carries forward Gloria Baylis’ legacy to enhance access to care through its divisions of Endovascular and Design and Manufacturing Services. Baylis Medical Technologies’ clinical solutions are utilized by healthcare professionals worldwide to improve patient outcomes for individuals with cardiovascular and other medical conditions. Learn more about our innovative products and ongoing mission at www.baylismedtech.com.For any inquiries, please contact [email protected] or call 1-888-505-4885
PRM-00910 J-1 V-1 © Baylis Medical Technologies Inc., 2024. PowerWire is a trademark and/or registered trademark of Baylis Medical Technologies Inc. in the United States and other countries. Baylis Medical Technologies Inc. reserves the right to change specifications or to incorporate design changes without notice and without incurring any obligation relating to equipment previously manufactured or delivered. Patents Pending and/or issued. CAUTION: Federal Law (USA) restricts the sale of these devices to or by the order of a physician. Before use, consult product labels and inserts for any indications, contraindications, hazards, warnings, cautions and instructions for use.
SOURCE Baylis Medical Technologies Inc.
Nanox Launches Artificial Intelligence Functionality in Second Opinions Platform
Nanox integrates FDA-cleared population health AI solutions into its Second Opinions service New AI capabilities available to patients who submit chest and abdominal CT scans for second opinions Offering the power of Artificial Intelligence to their second opinion CT report to promote early detection of chronic conditions PETACH TIKVA, Israel, June 05, 2024 (GLOBE NEWSWIRE) — NANO-X IMAGING LTD (“Nanox” or the “Company,” Nasdaq: NNOX), an innovative medical imaging technology company, today announced that its deep-learning medical imaging analytics subsidiary, Nanox AI Ltd., has launched an artificial intelligence (AI) functionality in the Second Opinions online medical consultation service. Second Opinions is a platform provided by USARAD Holdings INC, a subsidiary of Nano-X Imaging Ltd., that provides teleradiology services. The platform connects patients with radiologists and other subspecialty physicians for additional consultation on their medical diagnoses. Second Opinions has integrated three of Nanox.AI’s FDA 510(k)-cleared AI solutions, enabling patients to conveniently get second opinions from experts in various medical and surgical subspecialties including radiology, neurology, oncology and orthopedic surgery. The integration of Nanox.AI’s tools is intended to promote the early detection of chronic conditions on chest and abdominal CT scans: AI Cardiac solution (HealthCCSng) detects coronary artery calcium, an early sign of coronary artery diseaseAI Bone solution (HealthOST) assesses bone mineral density, and highlights vertebral spine compression fractures which can indicate risk of osteoporosisAI Liver solution (HealthFLD) measures liver density, which can indicate fatty liver disease These AI-driven insights are reviewed and approved by Second Opinions physicians and incorporated into reports for patients who submit eligible chest and abdominal CT scans. “We are excited to bring AI-powered, early detection through the Second Opinions platform to patients seeking peace of mind concerning their health and diagnoses,” said Erez Meltzer, Nanox Chief Executive Officer. “The integration of Nanox.AI’s solutions into the Second Opinions service will help empower radiologists and other healthcare providers by providing them with advanced AI tools that aim to improve patient outcomes. We will continue exploring opportunities to leverage our AI technology to promote accessible early diagnosis and preventative management.” Learn more about Second Opinions and its new AI capabilities at Artificial Intelligence (AI) Service – Second Opinions. About USARADUSARAD is a U.S.-based teleradiology company with a network of radiologists, certified by the American Board of Radiology. USARAD provides imaging interpretation and database services to radiology practices, hospitals, medical clinics, diagnostic imaging centers, urgent care facilities and multi-specialty physician groups in the U.S and additional countries, improving service levels, streamlining practice economics and enhancing physician efficiency. About Nanox.AINanox AI is the deep-learning medical imaging analytics subsidiary of Nanox. Nanox.AI solutions are developed to target highly prevalent chronic and acute diseases affecting large populations around the world. Leveraging AI technology, Nanox AI helps clinicians extract valuable and actionable clinical insights from routine medical imaging that otherwise may go unnoticed, potentially initiating further medical assessment to establish individual preventative care pathways for patients. For more information, please visit https://www.nanox.vision/ai. About NanoxNanox (NASDAQ: NNOX) is focused on applying its proprietary medical imaging technology and solutions to make diagnostic medicine more accessible and affordable across the globe. Nanox’s vision is to increase access, reduce costs and enhance the efficiency of routine medical imaging technology and processes, in order to improve early detection and treatment, which Nanox believes is key to helping people achieve better health outcomes, and, ultimately, to save lives. The Nanox ecosystem includes Nanox.ARC— a multi-source Digital Tomosynthesis system that is cost-effective and user-friendly; an AI-based suite of algorithms that augment the readings of routine CT imaging to highlight early signs often related to chronic disease (Nanox.AI); a cloud-based infrastructure (Nanox.CLOUD); and a proprietary decentralized marketplace, through Nanox’s subsidiary, USARAD Holdings Inc., that provides remote access to radiology and cardiology experts; and a comprehensive teleradiology services platform (Nanox.MARKETPLACE). Together, Nanox’s products and services create a worldwide, innovative, and comprehensive solution that connects medical imaging solutions, from scan to diagnosis. For more information, please visit www.nanox.vision. Contacts Media Contact:Ben ShannonICR WestwickeNanoxPR@icrinc.com Investor Contact:Mike CavanaughICR Westwickemike.cavanaugh@westwicke.com
Eko Health Raises $41 Million to Scale AI-Driven Heart and Lung Disease Detection
Supporting more than 500,000 U.S. healthcare professionals, Eko combines FDA-cleared algorithms with digital medical devices to enhance the early detection of cardiac and pulmonary diseases during physical exams.SAN FRANCISCO, June 5, 2024 /PRNewswire/ — Eko Health, a pioneer in applying artificial intelligence for early detection of heart and lung diseases, today announced it has raised $41 million in Series D financing. With participation by ARTIS Ventures, Highland Capital Partners, NTTVC, and Questa Capital, the funding round will be used to expand U.S. and global access to the company’s early disease detection platform.
Continue Reading
Supporting more than 500,000 U.S. healthcare professionals, Eko combines FDA-cleared algorithms with digital medical devices to enhance the early detection of cardiac and pulmonary diseases during physical exams.
The funding builds on recent commercial and clinical milestones, including U.S. FDA clearances for its structural heart murmur and low ejection fraction (Low EF) detection algorithms. Eko’s murmur detection algorithm was clinically validated in a Massachusetts General Hospital study and found to double identification rates of structural heart murmurs versus conventional practice in primary care. The low EF detection algorithm, developed with Mayo Clinic, was shown in an Imperial College London study to significantly enhance the identification of heart failure with reduced ejection fraction in GP clinics. By enhancing detection capabilities, Eko’s platform significantly reduces diagnostic bottlenecks, leading to earlier interventions and improved patient outcomes.
“Eko harnesses AI to unlock universal access to expert-level cardiac and pulmonary disease detection for patients everywhere,” said Connor Landgraf, CEO and co-founder of Eko Health. “Just as Ring transformed doorbells into home security systems, Eko has reinvented the world’s most ubiquitous medical tool into a powerful early disease detection platform, creating the world’s largest install base of professional AI-enabled cardiology devices.”
Cardiovascular and pulmonary diseases are among the leading causes of death worldwide, underscoring the critical need for early detection. Millions of patients remain unaware of their risk factors, often due to limited access to effective detection tools. Eko is addressing this gap by introducing sophisticated detection capabilities into any physical exam — from primary care exams at NCH Healthcare System in the U.S. to maternal-fetal health exams in Nigeria.”Eko has spent the past decade building an unparalleled dataset of digital heart and lung sounds, which it leverages to develop clinical AI for the physical world,” said Vas Bailey, PhD, Chair of the Board at Eko Health and Partner at ARTIS Ventures. “Like countless others, I lost a parent much too early to undetected heart disease. I am deeply inspired by the team’s dedication to saving lives by equipping hundreds of thousands—and soon millions—of clinicians worldwide with our groundbreaking early detection platform.”Eko will use the new capital to deepen its presence in the U.S. and accelerate its expansion into key international markets, supported by new strategic investments from Double Point Ventures in the U.S., Singapore-based global investor EDBI (the corporate investment arm of the Singapore Economic Development Board), and LG Technology Ventures, backed by the LG Group of South Korea. With the new funding and regulatory clearances, Eko is poised to rapidly expand access to its AI-enabled cardiac and pulmonary disease detection platform, empowering millions of healthcare professionals to improve patient outcomes in the coming years.About Eko HealthEko Health is a leading digital health company advancing how healthcare professionals detect and monitor heart and lung disease with its portfolio of digital stethoscopes, patient and provider software, and AI-powered analysis. Its FDA-cleared platform, used by over 500,000 healthcare professionals worldwide, allows them to detect earlier and with higher accuracy, diagnose with more confidence, manage treatment effectively, and ultimately give their patients the best care possible. Eko Health is headquartered in Emeryville, California, with over $165 million in funding from ARTIS Ventures, DigiTx Partners, Double Point Ventures, EDBI, Highland Capital Partners, LG Technology Ventures, Mayo Clinic, Morningside Technology Ventures Limited, NTTVC, Questa Capital, and others.Media Contact:Sam Moore[email protected]SOURCE Eko Health
HeartSciences Regains Compliance with Nasdaq Listing Requirements
Southlake, TX, June 04, 2024 (GLOBE NEWSWIRE) — Heart Test Laboratories, Inc. d/b/a HeartSciences (NASDAQ: HSCS; HSCSW) (“HeartSciences” or the “Company”), an artificial intelligence (AI)-powered medical technology company focused on transforming ECGs/EKGs to save lives through earlier detection of heart disease, today announced that on June 3, 2024, the Company received formal notice from the Listing Qualifications Staff of the Nasdaq Stock Market LLC indicating that HeartSciences has regained compliance with bid price requirement as set forth in Listing Rule 5550(a)(2), and that the Company is therefore in compliance with the Nasdaq Capital Market’s listing requirements and the scheduled hearing has now been cancelled. About HeartSciences Heart Test Laboratories, Inc. d/b/a HeartSciences is a medical technology company focused on applying innovative AI-based technology to an ECG (also known as an EKG) to expand and improve an ECG’s clinical utility. Millions of ECGs are performed every week and the Company’s objective is to improve healthcare by making it a far more valuable cardiac screening tool, particularly in frontline or point-of-care clinical settings. HeartSciences has one of the largest libraries of AI-ECG algorithms and is developing AI-ECG solutions to be made available on either a hardware agnostic cloud-based platform or its proprietary MyoVista® wavECG™ device, to help identify cardiovascular disease in any care setting worldwide in a manner to best suit different care providers. HeartSciences’ first product candidate for FDA clearance, the MyoVista® wavECG™, or the MyoVista®, is a resting 12-lead ECG that is also designed to provide diagnostic information related to cardiac dysfunction which has traditionally only been available through the use of cardiac imaging. The MyoVista® also provides conventional ECG information in the same test. For more information, please visit: https://heartsciences.com/. X: @HeartSciences Safe Harbor Statement This announcement contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements are made under the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995 and are relating to the Company’s future financial and operating performance. All statements, other than statements of historical facts, included herein are “forward-looking statements” including, among other things, statements about HeartSciences’ beliefs and expectations. These statements are based on current expectations, assumptions and uncertainties involving judgments about, among other things, future economic, competitive and market conditions and future business decisions, all of which are difficult or impossible to predict accurately and many of which are beyond the Company’s control. The expectations reflected in these forward-looking statements involve significant assumptions, risks and uncertainties, and these expectations may prove to be incorrect. Investors should not place undue reliance on these forward-looking statements, which speak only as of the date of this press release. Potential risks and uncertainties include, but are not limited to, risks discussed in HeartSciences’ Annual Report on Form 10-K for the fiscal year ended April 30, 2023, filed with the U.S. Securities and Exchange Commission (the “SEC”) on July 18, 2023, HeartSciences’ Quarterly Report on Form 10-Q for the fiscal quarter ended January 31, 2024, filed with the SEC on March 14, 2024 and in HeartSciences’ other filings with the SEC at www.sec.gov. Other than as required under the securities laws, the Company does not assume a duty to update these forward-looking statements. Contacts: HeartSciencesGene Gephart+1-682-244-2578 (US)info@heartsciences.com InvestorsGilmartin GroupVivian Cervantesinvestorrelations@heartsciences.com
OBVIUS Robotics™ Announces Growth Into New Development and Operations Facility
SUNRISE, Fla., June 4, 2024 /PRNewswire/ — Obvius Robotics, a medical device company developing an innovative technology platform for democratizing vascular access, today announced its move into a new development and operations facility. The South Florida facility will enable Obvius Robotics to support its growth as the company prepares for clearance and commercialization.
Obvius Robotics has developed technology that can be used with minimal training or experience, initially targeting central venous catheterization (CVC) procedures. The CERTA Access System is a hand-held, image guided device that enables clinicians to single-handedly target anatomical structures and access them with the push of a button using a proprietary targeting system and needle assembly. Early clinical use of the CERTA has demonstrated a 100% access success rate (link).
As the most common interventional procedure performed in the United States, CVC is a key platform from which to launch critical medical interventions for acutely ill patients and patients requiring surgery. The procedure involves gaining access to one of the great veins (internal jugular, subclavian, or femoral) to place a multi-lumen catheter for rapid replacement of blood volume, administration of emergency medicines and analgesics, and hemodynamic monitoring. Despite the broad use of CVC, these procedures carry a complication rate between 4% and 11% due to a lack of training and experience.
“The goal of Obvius Robotics is to democratize access, improve outcomes, and become the new standard of care in CVC,” said Russell Seiber, president and CEO of Obvius Robotics. “The new facility will not only support development and production of the CERTA Access System for CVC, but will also be a center of innovation to expand the CERTA platform into new applications”.
The CERTA Access System is not approved for clinical use at this time, but the company is currently preparing its submission to the U.S. Food and Drug Administration (FDA) for market clearance.
About Obvius Robotics
Obvius Robotics is a privately held medical device company based in South Florida with clinical offices in the Center for Device Innovation at the Texas Medical Center, Houston, that is developing technology for image-guided access of structures within the body. The platform technology, the CERTA Access System, incorporates robotics and imaging to improve the accuracy, safety, and consistency of accessing targeted anatomy. The company’s first clinical application is central venous catheter placement, a high-volume procedure in which CERTA has the potential to aid clinicians of varying levels of training and experience in safely and effectively achieving vascular access. Note: The CERTA Access System is not approved or cleared for use in patients in any geography at this time.
SOURCE OBVIUS Robotics, Inc.
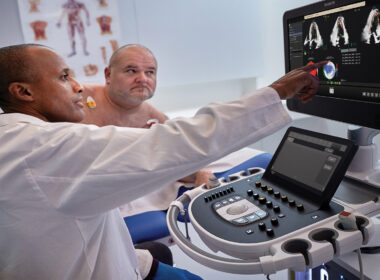
Deep integration of proven AI tools into Philips cardiovascular ultrasound systems to better diagnose more cardiac disease patients
June 4, 2024 New FDA-cleared AI-enabled applications include the industry’s first automated tool for segmental wall motion scoring of the heart to quickly and objectively identify disorders including coronary artery disease and cardio-oncology issues in secondsFirst fully automated 3D quantification of mitral regurgitation (MR) volumes* designed to provide reproducible, efficient analysis helps clinicians make better-informed decisions for patients with heart valve disease Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, announced its next-generation AI-enabled cardiovascular ultrasound platform to help speed up cardiac ultrasound analysis with proven AI technology and reduce the burden on echocardiography labs. Integrated into the company’s EPIQ CVx and Affiniti CVx ultrasound systems, the new FDA 510(k) cleared AI applications significantly advance Philips’ cardiovascular imaging and diagnosis solutions, automating measurements and speeding workflows to increase productivity. Heart failure is a rapidly growing health issue that affects an estimated 64 million people worldwide [1]. It’s also associated with high mortality and poor quality of life, and is a substantial burden on healthcare systems globally [2]. As the most common and least invasive way to check the structure and function of the heart, cardiovascular ultrasound has played a key role in diagnosing cardiac disease earlier. “As clinical cases get more complex and patient volumes increase, we read hundreds of echocardiography exams daily with thousands of data points,” said Roberto Lang, MD, Director of the Noninvasive Cardiac Imaging Lab, University of Chicago Medicine, USA. “With the integration of AI into echocardiography solutions, we can now automate some of the steps to support clinicians’ decision-making, allowing them to detect, diagnose, and monitor various cardiac conditions with greater confidence and efficiency in seconds.” Dr. Lang will join other clinicians to share the results of a new scientific abstract being presented at the American Society of Echocardiography (ASE2024) annual meeting (June 14 – 16, Portland, US), demonstrating how first-of-kind AI algorithms co-developed with Philips provide highly accurate detection of regional wall motion abnormalities (RWMA) on echocardiography. RWMAs can be an independent indicator of adverse cardiovascular events and death in patients with cardiovascular diseases like myocardial infarction (MI) and congenital heart disease. Automated machine learning-based assessment of RWMA has the potential to improve the efficiency of all readers. “An advantage of AI methods over conventional visual analysis is that it can be performed in seconds, providing rapid and accurate information to help augment expert reads by quickly highlighting areas of concern for RWMA, improving the ease and efficiency of interpretation,” Lang added. “By harnessing the power of AI into our echocardiography solutions, we empower clinicians with enhanced diagnostic capabilities, to ultimately improve patient care and outcomes in the management of coronary and valvular disease, while enhancing overall efficiency in cardiac practice,” said David Handler, VP and Business Leader for Global Cardiovascular Ultrasound at Philips. “For patients, this means consistent image interpretation which can lead to fewer re-scans, shorter and more effective interventional procedures, and potentially faster recovery times.” Integration of proven AI applications from Philips DiA Imaging Analysis Trained on anonymized patient data sets from real-life clinical environments, the AI features integrated across Philips cardiovascular ultrasound systems help improve the quality and reproducibility of cardiac imaging and enhance operator and departmental efficiency. These include FDA-cleared and CE-marked software solutions from DiA Imaging Analysis, a Philips company. Together, these AI features automate how users interpret ultrasound images, so clinicians with varying levels of ultrasound experience can automatically analyze images with increased speed, efficiency, and accuracy in real time. In addition to integrating these latest advanced AI features into the company’s cardiovascular ultrasound systems, Philips’ new mini ultrasound transducer X11 4t, introduced earlier this year, is fully compatible with the EPIQ CVx and Affiniti CVx systems. The innovative transducer allows interventional cardiologists to provide enhanced care to a wider range of patients, including pediatric patients as small as 5kg in weight. Through breakthrough innovation and collaborations with technology leaders including NVIDIA**, Philips continues to rapidly integrate AI into its cardiovascular ultrasound portfolio to help improve efficiency and productivity and mitigate staff shortages while delivering high-quality cardiology care to an increasing number of patients. These third-party solutions complement Philips’ proprietary AI-enabled solutions, integrating AI to enhance diagnostic confidence and decision-making. For more information, join Philips in Booth #441 at ASE2024 and visit Philips Cardiovascular Ultrasound for the latest AI-powered echocardiography applications. *Clinical performance and safety have not been established for some features which have 510(k) pending. Not available for sale in the USA.**NVIDIA MONAI for medical imaging AI and NVIDIA Holoscan for building software-defined medical devices. [1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10398425/ [2] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10398425/ For further information, please contact: Kathy O’ReillyPhilips Global External RelationsTel.: +1 978 221 8919E-mail : kathy.oreilly@philips.com About Royal Philips Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people’s health and well-being through meaningful innovation. Philips’ patient- and people-centric innovation leverages advanced technology and deep clinical and consumer insights to deliver personal health solutions for consumers and professional health solutions for healthcare providers and their patients in the hospital and the home. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, ultrasound, image-guided therapy, monitoring, and enterprise informatics, as well as in personal health. Philips generated 2023 sales of EUR 18.2 billion and employs approximately 69,100 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Attachments
Orchestra BioMed Announces Further Details on In-Person R&D Day in New York Featuring Key Opinion Leaders to Discuss AVIM Therapy Program in Hypertensive Pacemaker Patients on June 11, 2024
NEW HOPE, Pa., June 04, 2024 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO, “Orchestra BioMed” or the “Company”), a biomedical company accelerating high-impact technologies to patients through risk-reward sharing partnerships, today announced further details on its in-person R&D day at the Lotte New York Palace Hotel on Tuesday, June 11, 2024, at 10:00 AM ET. To register, click here. The R&D day will feature in-depth presentations focused on the Company’s lead program, atrioventricular interval modulation (AVIM) therapy, including: The high unmet need for treatment of uncontrolled hypertension in pacemaker-indicated patients and other older high-risk patientsThe AVIM therapy mechanism of action and supporting clinical and non-clinical mechanistic dataThe robust existing body of clinical evidence from the MODERATO I and II studiesDesign of and rationale for the BACKBEAT global pivotal study In addition to Orchestra BioMed management presentations, the event will include esteemed key opinion leading physicians: David Kandzari, M.D., FACC, FSCAI, Chief of the Piedmont Heart Institute and Cardiovascular Service Line; Director, Interventional Cardiology of the Piedmont Heart Institute; Chief Scientific Officer for Piedmont Healthcare in Atlanta, Georgia; and co-principal investigator on the BACKBEAT global pivotal study Dr. Kandzari specializes in cardiovascular disease, peripheral arterial disease and interventional cardiology. Board certified by the American Board of Internal Medicine and its subspecialty Board of Cardiovascular Disease and Board of Interventional Cardiology, Dr. Kandzari has held national and international leadership roles in clinical trials in cardiovascular disease and has participated in national and international program committees in cardiology. He has authored and coauthored more than 400 studies, book chapters and scientific reviews, and has delivered more than 800 lectures, both nationally and internationally, on a variety of issues related to both interventional and general cardiology. Dr. Kandzari has been consecutively voted as one of Atlanta’s Top Doctors by Atlanta Magazine from 2011 to 2024 and is peer-nominated in the top 1% of cardiologists by U.S News and World Report. Vivek Reddy, M.D., Director of Cardiac Arrhythmia Services for The Mount Sinai Hospital and the Mount Sinai Health System; Leona M. and Harry B. Helmsley Charitable Trust Professor of Medicine in Cardiac Electrophysiology at Icahn School of Medicine at Mount Sinai; and Clinical Steering Committee Advisor for the BACKBEAT global pivotal study Dr. Reddy is one of the nation’s premier cardiac electrophysiologists, leading a team of physician-scientists who are developing and testing advanced therapies for cardiac arrhythmias – including atrial fibrillation and ventricular tachycardias, the most common cause of sudden cardiac death. Dr. Reddy’s pioneering clinical research is changing the way cardiac patients are treated and cured. Under his leadership, Mount Sinai is the lead site on several multinational clinical trials exploring new arrhythmia procedures and technologies. In 2014, Dr. Reddy implanted the world’s first miniature leadless pacemaker in a patient’s heart in the United States at The Mount Sinai Hospital. A live question and answer session will follow formal presentations. About Orchestra BioMed Orchestra BioMed (Nasdaq: OBIO) is a biomedical innovation company accelerating high-impact technologies to patients through risk-reward sharing partnerships with leading medical device companies. Orchestra BioMed’s partnership-enabled business model focuses on forging strategic collaborations with leading medical device companies to drive successful global commercialization of products it develops. Orchestra BioMed’s lead product candidate is atrioventricular interval modulation (AVIM) therapy (also known as BackBeat Cardiac Neuromodulation Therapy (CNT™)) for the treatment of hypertension, a significant risk factor for death worldwide. Orchestra BioMed is also developing Virtue® Sirolimus AngioInfusion™ Balloon (SAB) for the treatment of atherosclerotic artery disease, the leading cause of mortality worldwide. Orchestra BioMed has a strategic collaboration with Medtronic, one of the largest medical device companies in the world, for the development and commercialization of AVIM therapy for the treatment of hypertension in pacemaker-indicated patients, and a strategic partnership with Terumo, a global leader in medical technology, for the development and commercialization of Virtue SAB for the treatment of artery disease. For further information about Orchestra BioMed, please visit www.orchestrabiomed.com, and follow us on LinkedIn. References to Websites and Social Media Platforms References to information included on, or accessible through, websites and social media platforms do not constitute incorporation by reference of the information contained at or available through such websites or social media platforms, and you should not consider such information to be part of this press release. About Hypertension and the Risk of High Blood Pressure in the Pacemaker Population Hypertension (“HTN”) is characterized by elevated blood pressure, which increases the force of blood pushing against blood vessels, requiring the heart to work harder and consume more oxygen. HTN accelerates the progression of atherosclerosis and leads to increased risk of major cardiac events like heart attack, heart failure, kidney disease and other end organ damage. HTN is the leading global risk factor for death, affecting an estimated 1.28 billion adults worldwide. In the United States, 122 million adults, or approximately 47% of all adults, are estimated to have HTN. While many patients do not notice high blood pressure, cardiovascular risk doubles for every 10-mmHg increase in systolic blood pressure and the mortality rate doubles with an increase of 20 mmHg in systolic blood pressure.1 It is estimated that more than 70% of the approximately 1.1 million people globally who are implanted with cardiac pacemakers each year are also diagnosed with HTN. Based on updated American College of Cardiology/American Heart Association guidelines, we estimate that an even higher percentage (approximately 80%) of U.S. patients who are indicated for a pacemaker implant have HTN. Pacemaker patients tend to be elderly and are more likely to suffer from co-morbidities, such as atherosclerosis, hyperlipidemia, diabetes mellitus and chronic kidney disease, and harder to treat effectively with medical therapy for many reasons, including co-morbidities and a high prevalence of isolated systolic HTN. About AVIM Therapy (BackBeat CNT™) AVIM therapy, also known as BackBeat CNT™, is an investigational therapy compatible with standard dual-chamber pacemakers designed to substantially and persistently lower blood pressure. It has been evaluated in pilot studies in patients with hypertension who are also indicated for a pacemaker. MODERATO II, a double-blind, randomized, pilot study, showed that patients treated with AVIM therapy experienced net reductions of 8.1 mmHg in 24-hour ambulatory systolic blood pressure (aSBP) and 12.3 mmHg in office systolic blood pressure (oSBP) at six months when compared to control patients. The IDE approved BACKBEAT (BradycArdia paCemaKer with atrioventricular interval modulation for Blood prEssure treAtmenT) global pivotal study will further evaluate the safety and efficacy of AVIM therapy in lowering blood pressure in a similar target population of patients who have been indicated for, and recently implanted with, a dual-chamber cardiac pacemaker. References: 1. Benjamin EJ, Blaha MJ, Chiuve SE, et al., Heart Disease and Stroke Statistics – 2017 Update: A Report from the American Heart Association. Circulation. 2017; 135: e146. Investor Contact:Bob YedidLifeSci Advisors(516) 428-8577Bob@lifesciadvisors.com Media Contact:Kelsey Kirk-EllisOrchestra BioMed(484) 682-4892Kkirkellis@orchestrabiomed.com
Amarin Board of Directors Announces CEO Transition
— Board Appoints Aaron Berg as President & CEO — — Patrick Holt to Step Down as President & CEO to Pursue Other Opportunities — DUBLIN and BRIDGEWATER, N.J., June 04, 2024 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN) today announced that the Company’s Board of Directors has appointed Aaron Berg, currently Amarin’s Executive Vice President and President of the U.S. Business, as President and Chief Executive Officer (CEO). The appointment of Mr. Berg follows the resignation of Patrick Holt as President & CEO of the Company. “On behalf of the Company’s Board of Directors, I thank Pat for his contributions to Amarin, and I welcome working closely again with Aaron as President & CEO,” said Odysseas Kostas, MD, the Chairman of the Board. “No one knows the company better than Aaron, and we look forward to him contributing meaningfully as CEO.” “As I begin this new role, my focus is clear – to find ways to deliver value for investors, and to maximize the potential of VASCEPA®/VAZKEPA for patients,” said Aaron Berg, President & CEO, Amarin. “I look forward to continuing to work with our Board of Directors and our tremendous team focusing on these priorities for the Company as we move forward.” Mr. Berg joined Amarin in November 2012 and has more than 30 years of biopharmaceutical industry experience. Mr. Berg initially served as Amarin’s Vice President, Marketing and Managed Care. He was promoted to Senior Vice President, Marketing and Sales in February 2014, and to the position of Senior Vice President and Chief Commercial Officer in April 2018. Before joining Amarin, Mr. Berg served as President and Chief Executive Officer for Essentialis, Inc., a development stage pharmaceutical company where he led the company’s work on triglyceride management. Prior to joining Essentialis, Mr. Berg served as Vice President of Marketing and Sales at Kos Pharmaceuticals (Kos), where he was instrumental in driving annual revenues approaching $1 billion. Mr. Berg worked at Kos until it was acquired by Abbott Laboratories in December 2006 for $3.7 billion. About Amarin Amarin is an innovative pharmaceutical company leading a new paradigm in cardiovascular disease management. We are committed to increasing the scientific understanding of the cardiovascular risk that persists beyond traditional therapies and advancing the treatment of that risk for patients worldwide. Amarin has offices in Bridgewater, New Jersey in the United States, Dublin in Ireland, Zug in Switzerland, and other countries in Europe as well as commercial partners and suppliers around the world. Forward-Looking Statements This press release contains forward-looking statements which are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, including beliefs about the potential for VASCEPA (marketed as VAZKEPA in Europe); beliefs about icosapent ethyl (IPE)’s role concerning appropriate patients suffering from cardiovascular disease (CVD) and potential population health impact, as well as general beliefs about the safety and effectiveness of VASCEPA. These forward-looking statements are not promises or guarantees and involve substantial risks and uncertainties. A further list and description of these risks, uncertainties and other risks associated with an investment in Amarin can be found in Amarin’s filings with the U.S. Securities and Exchange Commission, including Amarin’s annual report on Form 10-K for the full year ended 2023. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date they are made. Amarin undertakes no obligation to update or revise the information contained in its forward-looking statements, whether as a result of new information, future events or circumstances or otherwise. Amarin’s forward-looking statements do not reflect the potential impact of significant transactions the company may enter into, such as mergers, acquisitions, dispositions, joint ventures or any material agreements that Amarin may enter into, amend or terminate. Availability of Other Information About Amarin communicates with its investors and the public using the company website (www.amarincorp.com) and the investor relations website (amarincorp.com/investor-relations), including but not limited to investor presentations and FAQs, Securities and Exchange Commission filings, press releases, public conference calls and webcasts. The information that Amarin posts on these channels and websites could be deemed to be material information. As a result, Amarin encourages investors, the media and others interested in Amarin to review the information that is posted on these channels, including the investor relations website, on a regular basis. This list of channels may be updated from time to time on Amarin’s investor relations website and may include social media channels. The contents of Amarin’s website or these channels, or any other website that may be accessed from its website or these channels, shall not be deemed incorporated by reference in any filing under the Securities Act of 1933. Availability of Other Information About Amarin Investors and others should note that Amarin communicates with its investors and the public using the company website (www.amarincorp.com), the investor relations website (investor.amarincorp.com), including but not limited to investor presentations and investor FAQs, U.S. Securities and Exchange Commission filings, press releases, public conference calls and webcasts. The information that Amarin posts on these channels and websites could be deemed to be material information. As a result, Amarin encourages investors, the media, and others interested in Amarin to review the information that is posted on these channels, including the investor relations website, on a regular basis. This list of channels may be updated from time to time on Amarin’s investor relations website and may include social media channels. The contents of Amarin’s website or these channels, or any other website that may be accessed from its website or these channels, shall not be deemed incorporated by reference in any filing under the Securities Act of 1933. Amarin Contact Information Investor & Media Inquiries: Mark Marmur Amarin Corporation plc PR@amarincorp.com