WASHINGTON, March 7, 2026 /PRNewswire/ — Today at the Cardiovascular Research Technologies (CRT) annual conference taking place in Washington, DC, Dr. Christopher Chieh Yang Koo from National University Hospital Singapore, on behalf of investigators from Smith Center for Outcomes…
Author: Ken Dropiewski
Solid Biosciences Announces Oversubscribed $240 Million Private Placement
CHARLESTOWN, Mass., March 06, 2026 (GLOBE NEWSWIRE) — Solid Biosciences Inc. (Nasdaq: SLDB), a life sciences company developing precision genetic medicines for neuromuscular and cardiac diseases, today announced that it has entered into a securities purchase agreement with a select group of institutional accredited investors for an approximately $240 million private placement, before deducting placement agent fees and offering expenses, which is expected to close on or about March 9, 2026, subject to the satisfaction of customary closing conditions. The private placement is being conducted in accordance with applicable Nasdaq rules and was priced to satisfy the “Minimum Price” requirement (as defined in the Nasdaq rules). In the private placement, the Company is selling 14,973,257 shares of common stock at a price of $5.61 per share and, in lieu of common stock to investors who so choose, pre-funded warrants to purchase up to 27,807,482 shares of common stock at a price of $5.609 per pre-funded warrant. Each pre-funded warrant will have an exercise price of $0.001 per share, will be exercisable immediately, and will be exercisable until exercised in full. The private placement was anchored by existing and new investors, including Perceptive Advisors, Bain Capital Life Sciences, RA Capital Management, Invus, Vestal Point Capital, Janus Henderson Investors, and Deep Track Capital, among others. The Company expects to use net proceeds from the private placement to fund ongoing pipeline development programs, business development activities, and for working capital and other general corporate purposes. Leerink Partners and Citigroup are acting as joint lead placement agents for the financing. Cantor is acting as co-lead placement agent for the financing. Truist and H.C. Wainwright & Co. are acting as co-placement agents for the financing. The securities to be sold in the private placement have not been registered under the Securities Act of 1933, as amended (the “Securities Act”), or any state or other applicable jurisdiction’s securities laws, and may not be offered or sold in the United States absent registration or an applicable exemption from the registration requirements of the Securities Act and applicable state or other jurisdictions’ securities laws. The Company has agreed to file a registration statement with the U.S. Securities and Exchange Commission (the “SEC”) registering the resale of the shares of common stock issued in the private placement and the shares of common stock issuable upon the exercise of the pre-funded warrants issued in the private placement no later than the 30th day after the closing of the private placement. This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any offer, solicitation or sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful. Any offering of the securities under the resale registration statement will only be made by means of a prospectus. About Solid Biosciences Solid Biosciences is a precision genetic medicine company focused on advancing a portfolio of gene therapy candidates targeting rare neuromuscular and cardiac diseases, including SGT-003 for Duchenne muscular dystrophy (Duchenne), SGT-212 for Friedreich’s ataxia (FA), SGT-501 for catecholaminergic polymorphic ventricular tachycardia (CPVT), SGT-601 for TNNT2-mediated dilated cardiomyopathy and additional fatal, genetic cardiac diseases. The Company is also focused on developing innovative libraries of genetic regulators and other enabling technologies with promising potential to significantly impact gene therapy delivery cross-industry. Solid is advancing its diverse pipeline and delivery platform in the pursuit of uniting experts in science, technology, disease management, and care. Patient-focused and founded by those directly impacted by Duchenne, Solid’s mission is to improve the daily lives of patients living with devastating rare diseases. Cautionary Note Regarding Forward-Looking Statements This press release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, including statements regarding the anticipated closing of the private placement; the anticipated use of proceeds from the private placement; the filing of a registration statement to register the resale of the shares and pre-funded warrant shares to be issued and sold in the private placement; future expectations, plans and prospects for the Company; and other statements containing the words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “would,” “working” and similar expressions. Any forward-looking statements are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in, or implied by, such forward-looking statements. These risks and uncertainties include, but are not limited to: market and other financial conditions and the impact of general economic, industry or political conditions in the United States or internationally; whether the conditions for the closing of the private placement will be satisfied; the Company’s ability to advance SGT-003 and other clinical and preclinical programs, capsid libraries and other enabling technologies on the timelines expected or at all; obtain and maintain necessary approvals from the FDA and other regulatory authorities; replicate in clinical trials positive results found in preclinical studies of the Company’s product candidates; obtain, maintain or protect intellectual property rights related to its product candidates; enroll patients in ongoing trials; activate clinical trial sites; replicate preliminary or interim data from clinicals trials in the final data of such trials; compete successfully with other companies that are seeking to develop Duchenne, FA, CPVT and other neuromuscular and cardiac treatments and gene therapies; manage expenses; and raise the substantial additional capital needed, on the timeline necessary, to continue development of SGT-003 and its other candidates, achieve its other business objectives and continue as a going concern. For a discussion of other risks and uncertainties, and other important factors, any of which could cause the Company’s actual results to differ from those contained in the forward-looking statements, see the “Risk Factors” section, as well as discussions of potential risks, uncertainties and other important factors, in the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2025 and other filings that the Company may make with the SEC in the future. In addition, the forward-looking statements included in this press release represent the Company’s views as of the date hereof and should not be relied upon as representing the Company’s views as of any date subsequent to the date hereof. The Company anticipates that subsequent events and developments will cause the Company’s views to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. Solid Biosciences Investor Contact:Nicole AndersonSenior Director, Investor Relations and Corporate CommunicationsSolid Biosciences Inc.investors@solidbio.com Media Contact:Glenn SilverFINN Partnersglenn.silver@finnpartners.com
Valcare Medical Appoints Two New Members to Board of Directors
WILMINGTON, Del., March 6, 2026 /PRNewswire/ — Valcare Medical, Inc., a pioneer in transcatheter mitral valve repair solutions, today announced the appointment of two new members to its Board of Directors: Jeffrey B. Jump and Jacques Séguin, MD, PhD. These renowned experts in the…
VDyne Appoints Mike Buck as Chairman and CEO to Lead Next Phase of its TTVR Program
MAPLE GROVE, Minn.–(BUSINESS WIRE)–VDyne Inc. (“VDyne”), a privately held medical device company developing a transcatheter tricuspid valve replacement (TTVR) system to treat tricuspid regurgitation, today announced the appointment of Mike Buck as Chairman and Chief Executive Officer. Mr. Buck brings more than 30 years of leadership experience in cardiovascular medical […]
Harrison.ai Receives FDA Clearance for Acute Infarct Triage on Non-contrast CT Brain
Approval marks Harrison.ai’s second out of three Breakthrough designations to reach marketing authorization, and 9th FDA clearance covering a total of 13 radiological findings. SYDNEY & BOSTON–(BUSINESS WIRE)–Harrison.ai today announced FDA 510(k) clearance for acute infarct triage on non-contrast CT Brain. Acute infarct is one of the most difficult findings to […]
Profound Medical Reports Fourth Quarter and Full Year 2025 Financial Results
TORONTO, March 05, 2026 (GLOBE NEWSWIRE) — Profound Medical Corp. (NASDAQ:PROF; TSX:PRN) (“Profound” or the “Company”), a commercial-stage medical device company that develops and markets innovative interventional MRI (“iMRI”) procedures, today reported financial results for the fourth quarter and full year ended December 31, 2025. Unless specified otherwise, all amounts in this press release are expressed in U.S. dollars and are presented in accordance with U.S. generally accepted accounting principles (U.S. GAAP). Business Highlights Q4-2025 revenue grew 43% year-over-year and 13% sequentially quarter-over-quarter to a record $6.0 million. Profound’s TULSA-PRO® qualified sales pipeline also continues to grow, and currently stands at 110 new systems being classified within one of the “Verify, Negotiate and Contracting” stages.The Company’s TULSA-PRO installed base stood at 78 as of December 31, 2025 and, due to its strong capital sales pipeline, Profound currently expects to reach approximately 120 installs by the end of 2026.Profound continued to see a wide variety of prostate disease patients treated by its TULSA-PRO customers in the fourth quarter of 2025: 67% were treated for prostate cancer only, 17% were hybrid patients suffering from both prostate cancer and benign prostatic hyperplasia (“BPH”), 13% were salvage, and 3% were men with BPH only;For cancer grade, 5% were GG1, 57% were GG2, 29% were GG3, and 9% were GG4 & GG5;By intention-to-treat, 44% were whole gland; 24% were sub-total but more than half the gland; 24% were hemi-ablations, and 9% were focal therapy; andFor prostate size, 9% were
GAIA and Daiichi Sankyo Europe Enter Exclusive Partnership to Launch Next-Generation Digital Therapeutic for Cardiovascular Care in Europe.
HAMBURG, Germany & MUNICH–(BUSINESS WIRE)–GAIA, the pioneer in evidence-based digital therapeutics and Daiichi Sankyo Europe, today announced an exclusive strategic partnership to commercialize lipodia upon regulatory approval1. This digital therapeutic is designed to support adults living with hypercholesterolemia. The comprehensive collaboration brings together GAIA’s long-standing expertise in developing clinically validated […]
HeartBeam Announces First Patient Enrollments in Heart Attack Detection Pilot Study
HeartBeam’s technology uniquely capable of assessing possible heart attacks outside of traditional clinical settings ALIGN-ACS study expected to complete patient enrollment quickly Milestone signifies key step toward future FDA indication expansion for heart attack assessment Future indication expansion would unlock large market with over 20 million patients at risk of a […]
VentureMed Group Announces It Is Now Doing Business as Flex Vascular
MINNEAPOLIS, March 5, 2026 /PRNewswire/ — VentureMed Group, Inc., an innovative medical device company specializing in vessel preparation and access management for AV fistulas and grafts and peripheral vascular disease, today announced it is now doing business as Flex Vascular. The DBA…
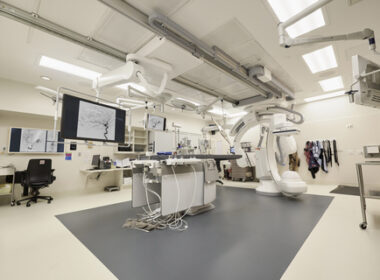
Children’s Minnesota Unveils Region’s First Dedicated Pediatric Neurointerventional Angiography Suite to Treat Stroke Patients
MINNEAPOLIS–(BUSINESS WIRE)–Children’s Minnesota is pleased to announce a $2.5 million, state-of-the-art biplane neuroangiography suite—the region’s only facility dedicated to pediatric neurointerventional procedures. This advanced imaging technology brings new treatment options for children with a variety of cerebrovascular diseases, further expanding Children’s Minnesota’s neurosurgical capabilities. “With this advanced biplane neuroangiography suite, we are raising the bar for children in