LAS VEGAS, Nov. 1, 2023 /PRNewswire/ — The VIVA Foundation, a not-for-profit organization dedicated to advancing the field of vascular medicine and intervention through education and research, announces the results for the last of four Late-Breaking Clinical Trial sessions at the VIVA23…
Author: Ken Dropiewski
Inari Medical Expands Its Patient-Focused Mission with Agreement to Acquire LimFlow
IRVINE, Calif., Nov. 01, 2023 (GLOBE NEWSWIRE) — Inari Medical, Inc. (NASDAQ: NARI) (“Inari”), a medical device company with a mission to treat and transform the lives of patients suffering from venous and other diseases, today announced that it has entered into a definitive agreement to acquire LimFlow, S.A., a […]
Third Round of Late-Breaking Clinical Trial Results Announced at VIVA23
LAS VEGAS, Nov. 1, 2023 /PRNewswire/ — The VIVA Foundation, a not-for-profit organization dedicated to advancing the field of vascular medicine and intervention through education and research, announces the results for the third of four Late-Breaking Clinical Trial sessions at the VIVA23…
InspireMD Presents Positive 30-Day Follow-Up Results from the C-GUARDIANS U.S. Investigational Device Exemption (IDE) Clinical Trial at VIVA23
Data demonstrate a low major adverse event rate of 0.95% through 30 days post-procedure TEL AVIV, Israel and MIAMI, Nov. 01, 2023 (GLOBE NEWSWIRE) — InspireMD, Inc. (Nasdaq: NSPR), developer of the CGuard™ Embolic Prevention Stent System (EPS) for the prevention of stroke, today presented 30-day results from the C-GUARDIANS […]
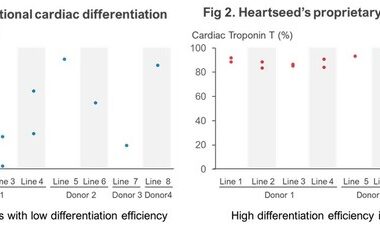
Using iPS Cells from I Peace, Heartseed Succeeds in Stable Production of High Purity Cardiomyocytes, A Major Step Forward in Advancing Autologous Cardiac Regenerative Medicine
PALO ALTO, Calif., Nov. 1, 2023 /PRNewswire/ — Leading GMP cell CDMO I Peace, Inc. (https://www.ipeace.com), specializing in induced pluripotent stem cells (iPSCs) and iPSC-derived cell therapies based in Palo Alto, CA, and Heartseed Inc. (“Heartseed”), a Tokyo-based biotechnology…
Covera Health Announces Up to $50M in Additional Series C Funding to Redefine the Role of Radiology Nationwide
Company Expands Industry’s Most Sophisticated AI-Enabled Radiology Performance Platform with Acquisition of CoRead NEW YORK, Nov. 1, 2023 /PRNewswire/ — Covera Health, the leading AI-enabled diagnostic technology company focused on radiology performance measurement and impact, today…
Silence Therapeutics Announces Positive Topline Results from Phase 1 Multiple Dose Study of Zerlasiran in Subjects with High Lipoprotein(a) and Stable Atherosclerotic Cardiovascular Disease
LONDON–(BUSINESS WIRE)—- $SLN #SenseofSilence–Silence Therapeutics plc, Nasdaq: SLN (“Silence” or the “Company”), an experienced and innovative biotechnology company committed to transforming people’s lives by silencing diseases through precision engineered medicines, today announced positive topline results from the multiple dose component of the APOLLO phase 1 study of zerlasiran (formerly SLN360) in 36 adults with baseline lipoprotein(a), or Lp(a), levels at or over 150 nmol/L and stable atherosclerotic cardiova
Second Round of Late-Breaking Clinical Trial Results Announced at VIVA23
LAS VEGAS, Oct. 31, 2023 /PRNewswire/ — The VIVA Foundation, a not-for-profit organization dedicated to advancing the field of vascular medicine and intervention through education and research, announces the results for the second of four Late-Breaking Clinical Trial sessions at the…
World’s First Patient Enrolled in SELUTION4DeNovo DEB IDE Trial
MIAMI LAKES, Fla., Oct. 31, 2023 /PRNewswire/ — Cordis has announced enrollment of the first patient in its SELUTION4DeNovo IDE trial, the coronary de novo study for SELUTION SLR™ Drug-Eluting Balloon (DEB), the company’s novel sirolimus-eluting balloon. SELUTION4DeNovo is the first DEB…
Endovascular Engineering, Inc. Announces Positive Initial Results in ENGULF Study for Hēlo™ Thrombectomy System
ENGULF Study utilizing next generation technology in targeting pulmonary embolism MENLO PARK, Calif., Oct. 31, 2023 /PRNewswire/ — Endovascular Engineering (“E2”), a mission driven, venture-backed, medical device company focused on the development and deployment of next generation clot…