4Q Revenues up 8% year-over-year, Organic revenues* up 13% 4Q Net income of $554 million versus $564 million for the prior year, Adjusted EBIT* of $844 million versus $827 million 4Q EPS were $1.21 versus $1.24 in the prior year, Adjusted EPS* were $1.31 versus $1.36 Reaffirmed 2023 guidance disclosed […]
Author: Ken Dropiewski
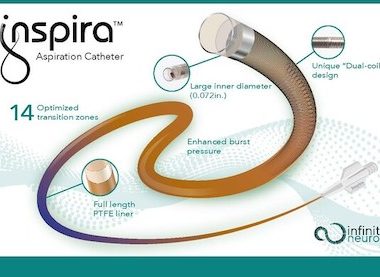
Infinity Neuro Receives CE Mark Approval for Their First Device to Treat Stroke
LAKE FOREST, Calif., Jan. 27, 2023 Infinity Neuro announced today that its Inspira™ aspiration catheters received CE Mark approval and are now commercially available in Europe. This is the first offering from Infinity Neuro which plans to launch a full range of products for the treatment of ischemic and hemorrhagic stroke throughout 2023 […]
Orchestra BioMed Debuts as Nasdaq-Traded Company with Lead Programs Targeting Hypertension and Artery Disease and Novel Partnership-Enabled Business Model
Business combination with Health Sciences Acquisitions Corporation 2, a special purpose acquisition company sponsored by an affiliate of RTW Investments, LP, completed on January 26, 2023 The combination was announced simultaneously with Orchestra’s strategic collaboration with Medtronic to develop BackBeat Cardiac Neuromodulation Therapy™ as potential integrated hypertension treatment for cardiac […]
CVRx Reports Fourth Quarter and Full Year 2022 Financial and Operating Results
Fourth Quarter 2022 Revenue of $7.2 million, a 96% Increase Over Prior Year MINNEAPOLIS, Jan. 26, 2023 (GLOBE NEWSWIRE) — CVRx, Inc. (NASDAQ: CVRX) (“CVRx”), a commercial-stage medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases, today announced its financial and operating […]
Amarin Announces VAZKEPA® (Icosapent Ethyl) Approved By Medsafe In New Zealand
Marks 7th Regulatory Approval for VASCEPA®/VAZKEPA® Franchise in Key International Markets Since 2022 DUBLIN, Ireland and BRIDGEWATER, N.J., Jan. 27, 2023 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN) today announced that Medsafe in New Zealand has granted approval to VAZKEPA (icosapent ethyl) to reduce the risk of cardiovascular (CV) events […]
JACC Publication Guides Use of CareDx’s HeartCare Transplant Rejection Surveillance in Clinical Practice
Guide Helps Transplant Centers Continue the Evolution from Endomyocardial Biopsies to Noninvasive Surveillance Using AlloMap GEP and AlloSure dd-cfDNA BRISBANE, Calif.–(BUSINESS WIRE)–CareDx, Inc. (Nasdaq: CDNA) – The Transplant Company™ focused on the discovery, development, and commercialization of clinically differentiated, high-value healthcare solutions for transplant patients and caregivers – today announced […]
Alleviant Medical Closes $75M in Financing; Receives FDA IDE Approval for Pivotal Clinical Trial
Novel technology shows potential to treat heart failure—leading cause of hospitalizations worldwide—with less invasive approach January 26, 2023 09:00 AM Eastern Standard Time AUSTIN, Texas–(BUSINESS WIRE)–Alleviant Medical, Inc., a privately-held medical device company developing a no-implant interatrial shunt for heart failure, today announced the closing of a $75 million equity […]
Clinical Study Shows Vista.ai Significantly Reduces Cardiac MRI Scan Time
Real-world cardiac MRI data gathered at Brigham and Women’s Hospital to be discussed at 26th Annual SCMR Scientific Sessions SAN DIEGO, Jan. 26, 2023 /PRNewswire/ — Vista.ai, a pioneer and leader in automated MRI solutions, today announced results from a clinical adaptation study on the company’s One Click MRI™ software, led by Dr. Raymond Kwong, Director, […]
XyloCor Therapeutics Reports Positive Topline Safety and Efficacy Results from Phase 2 EXACT Clinical Trial of XC001 Novel Gene Therapy for Refractory Angina
-No serious adverse events related to drug product were reported -Patients demonstrated improvements in exercise capacity and reductions in episodes of chest pain -Cardiac imaging results provide mechanistic evidence supporting the therapeutic potential of XC001 in cardiovascular disease WAYNE, Pa.–(BUSINESS WIRE)–XyloCor Therapeutics, a clinical-stage biopharmaceutical company developing novel gene therapies […]
Cardiac Dimensions Appoints Angie Swenson as Vice President, Clinical Operations
KIRKLAND, Wash.–(BUSINESS WIRE)–Cardiac Dimensions®, a leader in the development of innovative, minimally invasive treatment modalities to address heart failure and related cardiovascular conditions, today announced the appointment of Angie Swenson as Vice President of Clinical Operations. Ms. Swenson has spent most of the last 20 years leading clinical trials in […]