ISSUED ON BEHALF OF VENTRIPOINT DIAGNOSTICS LTD. USANewsGroup.com News Commentary VANCOUVER, BC, May 12, 2026 /PRNewswire/ — Cardiovascular disease is about to become the most expensive problem in American medicine. The American Heart Association fired a warning shot in late April: U.S….
Author: Ken Dropiewski
Adagio Medical Reports First Quarter 2026 Results and Meaningful Clinical Progress
LAGUNA HILLS, Calif.–(BUSINESS WIRE)—- $ADGM #ARRHYTHMIA–Adagio Medical Holdings, Inc (Nasdaq: ADGM) (“Adagio” or “the Company”), a leading innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, today announced financial results for the first quarter ended March 31, 2026. Recent Business Highlights: Pivotal results from the FULCRUM-VT trial presented in late-breaking session at Heart Rhythm Society 2026 – the first large-scale, rigorously executed pivotal trial in patients with bot
Humacyte Appoints Dr. Todd E. Rasmussen as Chief Surgical Officer
– Distinguished vascular surgeon brings decades of clinical experience, innovation, and leadership to new role at cutting-edge biotech manufacturer –
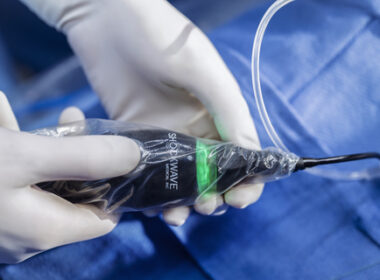
Johnson & Johnson Advances the Standard of Calcium Modification with Global Launch of Shockwave™ C2 Aero Coronary IVL Catheter
SANTA CLARA, Calif.–(BUSINESS WIRE)–Johnson & Johnson Advances the Standard of Calcium Modification with Global Launch of Shockwave™ C2 Aero Coronary IVL Catheter
NeuWire Medical Appoints Ramin Mousavi as Executive Chairman Following Stroke Journal Publication.
DALLAS–(BUSINESS WIRE)–NeuWire Medical, a medical device company advancing the development of a targeted plasticity therapy platform for individuals living with chronic ischemic stroke, today announced the appointment of Ramin Mousavi as Executive Chairman of the Board. His arrival marks an important step in the company’s next phase of growth as NeuWire Medical prepares to advance its clinical, regulatory, and financing strategy. Ramin joins NeuWire Medical following his role as President, Ch
CVRx Reports First Quarter 2026 Financial and Operating Results
MINNEAPOLIS, May 11, 2026 (GLOBE NEWSWIRE) — CVRx, Inc. (NASDAQ: CVRX) (“CVRx”), a commercial-stage medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases, today announced its financial and operating results for the first quarter of 2026. Recent Highlights Total revenue for the first quarter of 2026 was $14.8 million, an increase of approximately 20% over the prior year quarterU.S. revenue for the first quarter of 2026 was $13.7 million, an increase of 22% over the prior year quarterActive implanting centers in the U.S. grew to 257 as of March 31, 2026, as compared to 227 as of March 31, 2025First site activated and first patient enrolled in BENEFIT-HF clinical trial “We delivered a strong start to 2026, with U.S. revenue growing 22% as the investments we made in our team and programs throughout 2025 begin to translate into results,” said Kevin Hykes, President and Chief Executive Officer of CVRx. “We are seeing continued progress from our sales organization and the successful activation of the first site and our first patient enrolled in our BENEFIT-HF clinical trial. Together, these developments reinforce our confidence in the path ahead and in our ability to make Barostim more accessible to heart failure patients.” First Quarter 2026 Financial and Operating Results Revenue was $14.8 million for the three months ended March 31, 2026, an increase of $2.4 million, or 20%, over the three months ended March 31, 2025. Revenue generated in the U.S. was $13.7 million for the three months ended March 31, 2026, an increase of $2.4 million, or 22%, over the three months ended March 31, 2025. Revenue units in the U.S. totaled 429 and 359 for the three months ended March 31, 2026 and 2025, respectively. The increases were primarily driven by continued growth in the U.S. HF business as a result of the expansion into new sales territories, new accounts, and increased physician and patient awareness of Barostim. As of March 31, 2026, the Company had a total of 257 active implanting centers in the U.S., as compared to 252 as of December 31, 2025. Active implanting centers are customers that have completed at least one commercial HF implant in the last 12 months. The number of sales territories in the U.S. increased by three to a total of 56 during the three months ended March 31, 2026. Revenue generated in Europe was $1.1 million for the three months ended March 31, 2026, a decrease of $27,000, or 2%, over the three months ended March 31, 2025. Total revenue units in Europe decreased to 56 for the three months ended March 31, 2026 from 59 in the prior year period. The number of sales territories in Europe remained consistent at five for the three months ended March 31, 2026. Gross profit was $12.9 million for the three months ended March 31, 2026, an increase of $2.6 million, or 25%, over the three months ended March 31, 2025. Gross margin was 87% and 84% for the three months ended March 31, 2026 and March 31, 2025, respectively. R&D expenses increased $0.6 million, or 23%, to $3.1 million for the three months ended March 31, 2026 compared to the three months ended March 31, 2025. This change was driven by a $0.4 million increase in consulting expenses, a $0.3 million increase in compensation expenses, and a $0.1 million increase in non-cash stock-based compensation expenses, partially offset by a $0.2 million decrease in clinical trial expenses. SG&A expenses increased $0.7 million, or 3%, to $22.0 million for the three months ended March 31, 2026, compared to the three months ended March 31, 2025. This change was primarily driven by a $1.0 million increase in compensation expenses and a $0.3 million increase in non-cash stock-based compensation expenses, partially offset by a $0.3 million decrease in consulting expenses and a $0.3 million decrease in advertising expenses. Interest expense increased $0.1 million for the three months ended March 31, 2026, compared to the three months ended March 31, 2025, driven by the increased borrowings under the term loan agreement with Innovatus Capital Partners. Other income, net was $0.6 and $1.1 million for the three months ended March 31, 2026 and 2025, respectively. These balances consisted of interest income on our interest-bearing accounts. The decrease was primarily driven by the lower cash balance. Net loss was $13.1 million, or $0.50 per share, for the three months ended March 31, 2026, compared to a net loss of $13.8 million, or $0.53 per share, for the three months ended March 31, 2025. Net loss per share was based on 26.4 million weighted average shares outstanding for three months ended March 31, 2026 and 25.9 million weighted average shares outstanding for the three months ended March 31, 2025. As of March 31, 2026, cash and cash equivalents were $72.3 million. Net cash used in operating and investing activities was $12.3 million for the three months ended March 31, 2026 as compared to $12.9 million for the three months ended March 31, 2025. BENEFIT-HF Clinical Trial Update On March 31, 2026, the first site was activated in the BENEFIT-HF trial and the first patient was enrolled in the second quarter of 2026. This trial, as previously disclosed, is a landmark randomized controlled trial designed to evaluate Barostim’s impact on all-cause mortality and heart failure decompensation events in an expanded population of heart failure patients with left ventricular ejection fractions up to 50% and NT-proBNP levels up to 5,000 pg/mL. If successful, the BENEFIT-HF trial could expand the indicated patient population for Barostim approximately three times, significantly broadening access to this proven neuromodulation-based approach to heart failure management. Business Outlook For the full year of 2026, the Company maintained its revenue and expense guidance, and updated its guidance range for gross margin, as follows: Total revenue between $63.0 million and $67.0 million;Gross margin between 85% and 87%, compared to prior guidance of 84% and 86%;Operating expenses between $103.0 million and $107.0 million. For the second quarter of 2026, the Company expects to report total revenue between $15.1 million and $16.1 million. Webcast and Conference Call Information The Company will host a conference call to review its results at 4:30 p.m. Eastern Time today. A live webcast of the investor conference call will be available online at the investor relations page of the Company’s website at ir.cvrx.com. To listen to the conference call on your telephone, please dial 1-877-704-4453 for U.S. callers, or 1-201-389-0920 for international callers, approximately ten minutes prior to the start time. About CVRx, Inc. CVRx is a commercial-stage medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases. Barostim™ is the first medical technology approved by FDA that uses neuromodulation to improve the symptoms of patients with heart failure. Barostim is an implantable device that delivers electrical pulses to baroreceptors located in the wall of the carotid artery. The therapy is designed to restore balance to the autonomic nervous system and thereby reduce the symptoms of heart failure. Barostim received the FDA Breakthrough Device designation and is FDA-approved for use in heart failure patients in the U.S. It has been certified as compliant with the EU Medical Device Regulation (MDR) and holds CE Mark approval for heart failure and resistant hypertension in the European Economic Area. To learn more about Barostim, visit www.cvrx.com. Forward-Looking Statements This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts are forward-looking statements, including statements regarding our future financial performance (including our financial guidance regarding full year and second quarter 2026 results), our anticipated growth strategies (including statements regarding the expected timing, enrollment, scope and outcomes of the BENEFIT-HF clinical trial, potential expansion of the Barostim indication, and anticipated benefits of Barostim therapy), anticipated trends in our industry, our business prospects and our opportunities. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “outlook,” “guidance,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. The forward-looking statements in this press release are only predictions and are based largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition, and results of operations. These forward-looking statements speak only as of the date of this press release and are subject to a number of known and unknown risks, uncertainties and assumptions, including, but not limited to, our expectations regarding enrollment in BENEFIT-HF and the resulting impact on our addressable market; our history of significant losses, which we expect to continue; our limited history operating as a commercial company and our dependence on a single product, Barostim; our limited commercial sales experience marketing and selling Barostim; our ability to continue demonstrating to physicians and patients the merits of our Barostim; any failure by third-party payors to provide adequate coverage and reimbursement for the use of Barostim; our competitors’ success in developing and marketing products that are safer, more effective, less costly, easier to use or otherwise more attractive than Barostim; any failure to receive access to hospitals; our dependence upon third-party manufacturers and suppliers, and in some cases a limited number of suppliers; a pandemic, epidemic or outbreak of an infectious disease in the U.S. or worldwide; product liability claims; future lawsuits to protect or enforce our intellectual property, which could be expensive, time consuming and ultimately unsuccessful; any failure to retain our key executives or recruit and hire new employees; impacts on adoption and regulatory approvals resulting from additional long-term clinical data about our product, including those resulting from the BENEFIT-HF trial; and other important factors that could cause actual results, performance or achievements to differ materially from those that are found in “Part I, Item 1A. Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025, as such factors may be updated from time to time in our other filings with the Securities and Exchange Commission. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Investor Contact:Mark Klausner or Mike VallieICR Healthcare443-213-0501ir@cvrx.com Media Contact:Emily Meyers CVRx, Inc. 763-416-2853emeyers@cvrx.com CVRx, INC.Condensed Consolidated Balance Sheets(In thousands, except share and per share data)(Unaudited) March 31, December 31, 2026 2025Assets Current assets: Cash and cash equivalents $72,303 $75,708 Accounts receivable, net of allowances of $869 and $871, respectively 9,104 10,665 Inventory 12,403 12,205 Prepaid expenses and other current assets 2,940 3,069 Total current assets 96,750 101,647 Property and equipment, net 2,159 2,243 Operating lease right-of-use asset 793 878 Other non-current assets 26 26 Total assets $99,728 $104,794 Liabilities and Stockholders’ Equity Current liabilities: Accounts payable $2,998 $3,833 Accrued expenses 6,488 9,484 Total current liabilities 9,486 13,317 Long-term debt 58,490 49,514 Operating lease liability, non-current portion 544 638 Other long-term liabilities 2,099 2,001 Total liabilities 70,619 65,470 Commitments and contingencies Stockholders’ equity: Common stock, $0.01 par value, 200,000,000 authorized as of March 31, 2026 and December 31, 2025; 26,428,767 and 26,311,607 shares issued and outstanding as of March 31, 2026 and December 31, 2025, respectively 264 263 Additional paid-in capital 632,820 629,916 Accumulated deficit (603,772) (590,652)Accumulated other comprehensive loss (203) (203)Total stockholders’ equity 29,109 39,324 Total liabilities and stockholders’ equity $99,728 $104,794 CVRx, INC.Condensed Consolidated Statements of Operations and Comprehensive Loss(In thousands, except share and per share data)(Unaudited) Three months ended March 31, 2026 2025Revenue $14,769 $12,348 Cost of goods sold 1,888 2,036 Gross profit 12,881 10,312 Operating expenses: Research and development 3,084 2,517 Selling, general and administrative 21,958 21,232 Total operating expenses 25,042 23,749 Loss from operations (12,161) (13,437)Interest expense (1,551) (1,457)Other income, net 593 1,123 Loss before income taxes (13,119) (13,771)Benefit (provision) for income taxes (1) 5 Net loss (13,120) (13,766)Cumulative translation adjustment — — Comprehensive loss $(13,120) $(13,766)Net loss per share, basic and diluted $(0.50) $(0.53)Weighted-average common shares used to compute net loss per share, basic and diluted 26,355,591 25,876,062
Cytokinetics Announces New Data Related to MYQORZO® (aficamten) at ESC Heart Failure 2026
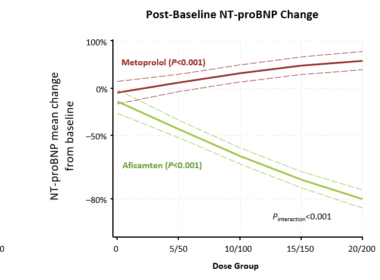
Multinational Real-world Data Reinforce Favorable Clinical Profile and Long-Term Safety SOUTH SAN FRANCISCO, Calif., May 11, 2026 (GLOBE NEWSWIRE) — Cytokinetics, Incorporated (Nasdaq: CYTK) today announced the presentation of new data reinforcing the clinical profile of MYQORZO® (aficamten) at the European Society of Cardiology (ESC) Heart Failure 2026 Congress. The presentations include new analyses from SEQUOIA-HCM, the pivotal Phase 3 clinical trial of aficamten in patients with oHCM; MAPLE-HCM, the Phase 3 clinical trial of aficamten compared to metoprolol in patients with symptomatic obstructive HCM (oHCM); and FOREST-HCM, the open-label extension trial of aficamten. Collectively, the new evidence expands understanding of the effectiveness of cardiac myosin inhibition with aficamten compared directly to metoprolol, a beta blocker, as well as specific safety and durability characteristics of MYQORZO across patient demographics, clinical, and economic subgroups. “The breadth of research being presented at ESC Heart Failure 2026 reflects our commitment to deepening the scientific understanding of oHCM and heart failure,” said Stephen Heitner, M.D., Senior Vice President, Clinical Research and Development, Cytokinetics. “These new insights underscore the strength and consistency of the clinical profile of MYQORZO, adding to the growing body of real-world evidence informing physician treatment decisions.” New Analyses Show MYQORZO Outperforms Metoprolol Across Sex and Doses A dose-dependent analysis from MAPLE-HCM compared aficamten to the beta-blocker metoprolol in patients with symptomatic oHCM. Key findings showed significant improvements in exercise capacity, outflow gradients, and N-terminal pro-B-type natriuretic peptide (NT-proBNP) across all treatment doses. Conversely, metoprolol failed to show improvement in these outcomes regardless of the dose administered (Figure 1). Figure 1 A secondary analysis of sex differences in MAPLE-HCM showed consistent benefits of MYQORZO in women. Despite women entering the trial (42%) with more severe baseline characteristics, MYQORZO delivered nearly identical improvements in peak oxygen consumption (pVO2) for both sexes (+2.2 mL/kg/min). Both groups also saw significant gains in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and reductions in cardiac biomarkers and NT-proBNP. Long-term Data Reinforce Safety and Efficacy Profile of MYQORZO A prospective analysis of 122 patients who had interpretable ambulatory electrocardiogram (ECG) data at screening in the FOREST-HCM showed that long-term treatment with MYQORZO—up to 96 weeks—did not increase the incidence of arrhythmias in patients, including those who underwent withdrawal of beta-blocker therapy. Among 122 patients, the incidence of non-sustained ventricular tachycardia (NSVT) on ambulatory ECG remained stable through 96 weeks compared with baseline, with no increase in atrial fibrillation (AF) episodes and no newly identified subclinical AF. There was also no increase in arrhythmia frequency among a subgroup of 16 patients who discontinued beta-blocker therapy during aficamten treatment. These results represent the first prospective analysis of ambulatory ECG monitoring in patients treated with a cardiac myosin inhibitor and are consistent with the low incidence of clinically detected arrhythmias previously reported for patients with oHCM treated with aficamten. Additionally, an open-label extension study of aficamten in Chinese patients with symptomatic oHCM showed that aficamten was well tolerated using the same dosing strategy of individualized titration as was used globally. Through 48 weeks of treatment with aficamten, patients experienced no serious or severe treatment-emergent adverse events, no occurrences of LVEF
Hengrui Pharma and Braveheart Bio Announce Positive Phase 2 Results with HRS/BHB-1893 in Non-Obstructive Hypertrophic Cardiomyopathy
Results with next-generation cardiac myosin inhibitor suggest a potentially disease-modifying therapy in non-obstructive hypertrophic cardiomyopathy HRS/BHB-1893 treatment resulted in rapid and sustained reductions in key biomarkers, structural remodeling and meaningful improvements in…
Acoramidis Continues to Demonstrate Disease-Modifying Effects in ATTR-CM, Reducing sTTR Variability and Outpatient Worsening Heart Failure
-Acoramidis increased sTTR early and significantly reduced intra-individual sTTR variability versus placebo (p
Longeveron Announces Independent Data Monitoring Committee Positive Review and Recommendation to Complete Phase 2b Stem Cell Therapy Clinical Trial in Hypoplastic Left Heart Syndrome (HLHS)
Nataliya Agafonova
Nataliya Agafonova, M.D., Chief Medical Officer, Longeveron
Phase 2b clinical trial (ELPIS II) evaluating laromestrocel as a potential adjunct treatment for HLHS, a rare pediatric disease and orphan-designated indicationDMC performed a risk-benefit assessment, identified no new safety concerns, and recommended continuation of the study as designed through completionThis review represents the final planned independent safety assessment prior to completion of the trialELPIS II top-line trial results anticipated in August 2026 MIAMI, May 11, 2026 (GLOBE NEWSWIRE) — Longeveron Inc. (NASDAQ: LGVN), a clinical stage biotechnology company developing cellular therapy for life-threatening, rare pediatric and chronic aging-related conditions, today announced that the independent Data Monitoring Committee (DMC) has completed its final prespecified data review for the ongoing, fully enrolled, Phase 2b clinical trial (ELPIS II) evaluating laromestrocel (Lomecel-B®) as a potential adjunct therapy for hypoplastic left heart syndrome (HLHS). The DMC based its review on available data for all participating patients enrolled. The DMC performed a risk-benefit assessment, indicated no safety concerns, and approved the study to continue as designed to completion. “We are grateful to the members of the DMC for their effort and diligence in this review,” said Nataliya Agafonova, M.D., Chief Medical Officer at Longeveron. “Based on the scientific rationale supporting laromestrocel and the clinical data generated to date, we remain encouraged by the potential of our investigational therapy in HLHS. With our Phase 2b clinical trial for HLHS having achieved full enrollment last June, we continue to anticipate top-line trial results in August 2026 and look forward to sharing the data.” ELPIS II enrolled 40 pediatric patients at 12 leading pediatric cardiac centers across the country. The clinical trial is being conducted in collaboration with the National Heart, Lung, and Blood Institute (NHLBI) through grants from the National Institutes of Health (NIH). ELPIS II builds on findings from ELPIS I study, in which treated children demonstrated 100% transplant-free survival up to five years of age. Published historical outcomes in HLHS populations have reported substantial mortality despite surgical palliation. The U.S. Food and Drug Administration (FDA) has granted laromestrocel Orphan Drug designation, Fast Track designation, and Rare Pediatric Disease designation, for development for HLHS. About Hypoplastic Left Heart Syndrome (HLHS) HLHS is a rare congenital heart defect that effects approximately 1,000 infants per year in the U.S. Infants with HLHS are born with an underdeveloped left ventricle, which creates a life-threatening condition due to the heart’s inability to pump adequate amounts of blood throughout the body. The current treatment requires infants to undergo a complex three-stage heart reconstruction surgery process over the first five years of their life. Despite staged surgical palliation, long-term mortality and morbidity remain substantial, with progressive right ventricular dysfunction representing a major contributor to adverse outcomes. There is clearly an important unmet medical need to improve right ventricular function in these infants to positively impact both short- and long-term patient outcomes. About laromestrocel (Lomecel-B®)Laromestrocel is a living cell product made from specialized cells isolated from the bone marrow of young healthy adult donors. These specialized cells, known as mesenchymal stem cells (MSCs), are essential to our endogenous biological repair mechanism. MSCs have been shown to perform a number of complex functions in the body and support tissue repair and regenerative signaling pathways. They also have been shown to respond to sites of injury or disease and secrete bioactive factors that are immunomodulatory and regenerative. We believe that laromestrocel MSCs may have multiple potential mechanisms of action that may lead to anti-inflammatory, pro-vascular regenerative responses, and therefore may have broad application for a range of rare and aging related diseases. About Longeveron Inc. Longeveron is a clinical stage biotechnology company developing regenerative medicines to address unmet medical needs. The Company’s lead investigational product is laromestrocel (Lomecel-B™), an allogeneic mesenchymal stem cell (MSC) therapy product isolated from the bone marrow of young, healthy adult donors. Laromestrocel has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas. Longeveron is pursuing four pipeline indications: hypoplastic left heart syndrome (HLHS), Alzheimer’s disease (AD), Pediatric Dilated Cardiomyopathy (DCM) and Aging-related Frailty. Laromestrocel development programs have received five distinct and important FDA designations: for the HLHS program – Orphan Drug designation, Fast Track designation, and Rare Pediatric Disease designation; and, for the AD program – Regenerative Medicine Advanced Therapy (RMAT) designation and Fast Track designation. For more information, visit www.longeveron.com or follow Longeveron on LinkedIn, X, and Instagram. Forward-Looking StatementsCertain statements in this press release that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve known and unknown risks, uncertainties, and other important factors that could cause actual results, performance, or achievements to differ materially from those anticipated, expressed, or implied by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expects,” “intend,” “looks to,” “may,” “on condition,” “plan,” “potential,” “predict,” “preliminary,” “project,” “see,” “should,” “target,” “will,” “would,” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, our cash position and need to raise additional capital, the difficulties we may face in obtaining access to capital, and the dilutive impact it may have on our investors; our financial performance, and ability to continue as a going concern; the period over which we estimate our existing cash and cash equivalents will be sufficient to fund our future operating expenses and capital expenditure requirements; the ability of our clinical trials to demonstrate safety and efficacy of our product candidates, and other positive results; the timing and focus of our ongoing and future preclinical studies and clinical trials, and the reporting of data from those studies and trials; the size of the market opportunity for certain of our product candidates, including our estimates of the number of patients who suffer from the diseases we are targeting; our ability to scale production and commercialize the product candidate for certain indications; the success of competing therapies that are or may become available; the beneficial characteristics, safety, efficacy and therapeutic effects of our product candidates; our ability to obtain and maintain regulatory approval of our product candidates in the U.S. and other jurisdictions; our plans relating to the further development of our product candidates, including additional disease states or indications we may pursue; our plans and ability to obtain or protect intellectual property rights, including extensions of existing patent terms where available and our ability to avoid infringing the intellectual property rights of others; the need to hire additional personnel and our ability to attract and retain such personnel; and our estimates regarding expenses, future revenue, capital requirements and needs for additional financing. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2024, filed with the Securities and Exchange Commission on February 28, 2025, its Quarterly Reports on Form 10-Q, and its Current Reports on Form 8-K. The Company operates in highly competitive and rapidly changing environment; therefore, new factors may arise, and it is not possible for the Company’s management to predict all such factors that may arise nor assess the impact of such factors or the extent to which any individual factor or combination thereof, may cause results to differ materially from those contained in any forward-looking statements. The forward-looking statements contained in this press release are made as of the date of this press release based on information available as of the date of this press release, are inherently uncertain, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise. Investor and Media Contact:Derek ColeInvestor Relations Advisory Solutionsderek.cole@iradvisory.com A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/e9e96d9c-d6c3-4aec-ba85-36631b520211