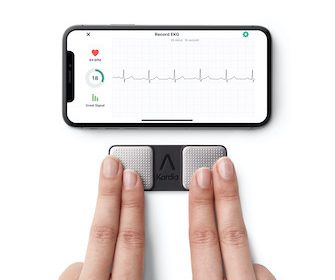
AliveCor’s KardiaMobile® is the only personal electrocardiogram (ECG) to be recommended by the National Institute for Health and Care Excellence (NICE) for use within the NHS[1] NICE’s positive recommendation[1] offers patients and physicians across England and Wales access to a medical-grade ECG to support at home monitoring of paroxysmal atrial fibrillation (AF) without the requirements […]
Author: Ken Dropiewski
SERIAL INNOVATOR, EITAN KONSTANTINO JOINS GENESIS MEDTECH AS CTO OF VASCULAR INTERVENTIONS FRANCHISE
SINGAPORE, Jan. 7, 2022 /PRNewswire/ — Genesis MedTech Group (Genesis or Group) has appointed Dr. Eitan Konstantino (Eitan) as CTO of Vascular Interventions to propel Genesis’ technology innovation within the neurovascular, cardiovascular and peripheral vascular spaces. His role will include developing and implementing the Group’s global Vascular Interventions R&D strategy, driving the enhancement of […]
Cytokinetics and Royalty Pharma Announce Funding Agreements Totaling up to $450 Million
Royalty Pharma Extends Capital to Cytokinetics to Support the Commercial Launch of Omecamtiv Mecarbil and the Further Development of Aficamten Royalty Pharma Purchases Royalties on Future Sales of Aficamten Cytokinetics to Host Conference Call and Webcast Today at 8:30 am Eastern Time SOUTH SAN FRANCISCO, Calif. and NEW YORK, Jan. […]
AngioDynamics Reports Fiscal 2022 Second Quarter Financial Results; Reaffirms Revenue Guidance; Revises Gross Margin and Adjusted EPS Guidance
Fiscal 2022 Second Quarter Highlights Net sales of $78.3 million increased 7.6% compared to the prior-year quarter Med Tech growth of 36.4% and Med Device growth of 0.8% year over year Gross margin of 51.8% declined 340 basis points year over year GAAP loss per share of $0.21, and adjusted […]
HELIOS CARDIO ANNOUNCES US FDA 510(k) CLEARANCE FOR A NEW PERICARDIAL AND EPICARDIAL RECONSTRUCTION MATRIX
Offers cardiac surgeons with a best-in-class biomaterial indicated for pericardial repair and reconstruction, and epicardial support and repair WESTON, Mass., Jan. 5, 2022 /PRNewswire/ — Helios Cardio Inc. announces the Food and Drug Administration (FDA) 510(k) clearance for commercial distribution of CardiaMend™ Pericardial and Epicardial Reconstruction Matrix. CardiaMend™ Pericardial and Epicardial Reconstruction […]
LQT Therapeutics Announces Award by the European Joint Program for Rare Diseases for SGK1 inhibition as a novel therapeutic approach in Long QT syndrome
Closing the gap between industry and academia to place patients directly at the center of research efforts. , Jan. 6, 2022 /PRNewswire/ – LQT Therapeutics, a privately-held biopharmaceutical company advancing novel SGK1 kinase therapeutics, today announced that the project “SILENCE-LQTS: SGK1 inhibition as a novel therapeutic approach in Long QT syndrome” was selected for […]
Housatonic Partners Invests in Mobile Vascular Physicians
SAN FRANCISCO and BETHPAGE, N.Y., Jan. 5, 2022 /PRNewswire/ — Housatonic Partners, a growth-oriented private equity firm focused on investing in market-leading services companies, announced a strategic growth investment in Mobile Vascular Physicians (“MVP”), a New York-based concierge medical practice that utilizes a specialized, mobile approach to provide vascular and interventional radiology services to residents […]
Spacelabs Healthcare Introduces CardioPulse ECG Solutions for a Faster Path to Patient Care
SNOQUALMIE, Wash.–(BUSINESS WIRE)–Spacelabs Healthcare, a division of OSI Systems, Inc. (the “Company” or “OSI Systems”) (NASDAQ: OSIS), today announced the release of CardioPulse™ Go and CardioPulse™ Prime resting Electrocardiograph (ECG) devices. “Workflow fit is a critical factor for clinicians when choosing resting ECG devices,” said Spacelabs President Shalabh Chandra. “Keeping […]
Neovasc Announces First Patient Enrollment in COSIRA-II Trial
COSIRA-II Trial Designed to Study the Neovasc Reducer™ for Patients with Refractory Angina VANCOUVER and MINNEAPOLIS, Jan. 05, 2022 (GLOBE NEWSWIRE) — via NewMediaWire — Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN) today announced enrollment of the first patient in the COSIRA-II clinical trial. COSIRA-II (COronary SInus Reducer for the Treatment of Refractory Angina) is a pivotal trial […]
Adagio Medical Announces Successful First-In-Human Cases of Pulsed Field Cryoablation for Treatment of Atrial Fibrillation
LAGUNA HILLS, Calif., Jan. 4, 2022 /PRNewswire/ — Adagio Medical, Inc., a leading innovator in catheter ablation technologies for atrial fibrillation (AF) and ventricular tachycardia (VT), today announced first-in-human cases of pulsed field cryoablation (PFCA) for the treatment of atrial fibrillation (AF). The cases were performed at Medicover Hospital, Warsaw, Poland by Dr. Pawel Derejko, Head […]