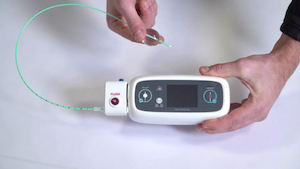
Remote blood pressure monitoring service standardizes BP collection PHILADELPHIA, June 14, 2021 /PRNewswire/ — ERT, the global leader in clinical endpoint data collection, today announced the expansion of its clinical trial endpoint data collection portfolio with at-home blood pressure (BP) monitoring solution. This completes a suite of blood pressure assessment services developed by […]
Author: Ken Dropiewski
BioCardia to Be Added to the Russell Microcap® Index
SAN CARLOS, Calif., June 14, 2021 (GLOBE NEWSWIRE) — BioCardia®, Inc. (Nasdaq: BCDA), a leader in the development of comprehensive solutions for cardiovascular and pulmonary cell based therapies, today announced it is to join the Russell Microcap® Index. This milestone will take place at the conclusion of the 2021 Russell Indexes annual reconstitution, […]
Steven Schoch Elected to Arena Pharmaceuticals Board of Directors
PARK CITY, Utah–(BUSINESS WIRE)–Arena Pharmaceuticals, Inc. (Nasdaq: ARNA) today announced the appointment of Steven Schoch to the Company’s Board of Directors and as Chair of the Audit Committee. Mr. Schoch brings more than 20 years of financial and operational leadership and expertise across a range of areas in biopharmaceuticals, media, and […]
Neovasc Provides Corporate Update
VANCOUVER and MINNEAPOLIS, June 10, 2021 (GLOBE NEWSWIRE) — via NewMediaWire — Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN) today announced that the Company has implemented a series of strategic initiatives focused on enhancing current shareholder value, minimizing dilution, extending its cash runway well into 2024, focusing investments on near term value drivers, […]
Caelum and Alexion Present Additional Phase 2 Data Reinforcing Safety and Tolerability of CAEL-101 in AL Amyloidosis at the European Hematology Association Congress 2021
– Exploratory biomarker analyses suggest possible cardiac disease improvements and renal response – BORDENTOWN, N.J. & BOSTON–(BUSINESS WIRE)–Caelum Biosciences and Alexion Pharmaceuticals, Inc. (NASDAQ:ALXN) today announced new Phase 2 safety and tolerability data for CAEL-101, a potentially first-in-class amyloid fibril targeted therapy, in combination with standard-of-care (SoC) therapy in patients with AL […]
Heartseed Raises $37 Million in Series C Funding to Accelerate Development of iPSC-derived Stem Cell Therapy for Heart Failure
TOKYO–(BUSINESS WIRE)–Heartseed, a Tokyo-based biotechnology company developing iPSC-derived cardiomyocytes for heart failure (HF), today announced it has raised 4 Billion-yen (approx. $37 Million) at Series C round, bringing its total financial backing to 8.2 Billion yen (approx. $75 Million) since its foundation in 2015. New investors are UTokyo Innovation Platform […]
Neurescue’s Breakthrough Intelligent Balloon Catheter FDA 510(k) Cleared for Hemorrhage Control and IDE Approved for Cardiac Arrest
Device is FDA IDE approved to start a clinical study in the U.S. to investigate a novel cardiac arrest treatment indication COPENHAGEN, Denmark–(BUSINESS WIRE)–Neurescue, a medical device company developing innovative cardiovascular solutions to improve the outcomes for emergency patients, today announced that the U.S. Food and Drug Administration (FDA) has […]
Peijia Medical Partners with inQB8 for US Incubator and Transcatheter Tricuspid Replacement (TTVR) Product
SUZHOU, China, June 9, 2021 /PRNewswire/ — Peijia Medical (HKEX:9996, or “Peijia“), a leading player in China for medical technology, announced a partnership with inQB8, a Boston based medical technology incubator, to explore innovative solutions for Structural Heart Disease. This partnership includes Peijia’s acquisition of a transcatheter tricuspid replacement technology (“TTVR”, or “inQB8 TTVR”) from inQB8, which is currently in […]
CVRx® Announces First Clinical Procedure with a New Ultrasound-Guided Implant Approach, the Latest Advancement in Barostim™ for the Treatment of Heart Failure Symptoms
MINNEAPOLIS, June 10, 2021 (GLOBE NEWSWIRE) — CVRx®, developer of the world’s first FDA-approved neuromodulation device to treat the symptoms of heart failure (HF), announced the completion of the first clinical procedure with the company’s new lead implantation approach. The novel ultrasound-guided technique is the latest advancement of CVRx’s Barostim™ Baroreflex Activation […]
Arcoma receives first order on the new product, OMNERA 500A
2021-06-10 PRESSRELEASE Arcoma receives first order on the new product, OMNERA 500A Arcoma AB has received the first order on the new product OMNERA 500A. The OMNERA 500A is a new X-ray system developed with focus on efficient workflow, safety and ease of use. The ergonomic design simplifies and optimizes […]