First-of-its-kind medical device aims to improve vascular access for hemodialysis patients PRAIRIE VILLAGE, Kan., Jan. 27, 2021 /PRNewswire/ — Artio Medical, Inc., a medical device company developing innovative products for the peripheral vascular, neurovascular, and structural heart markets, announced today that it has successfully completed the first human use of its Amplifi™ Vein Dilation […]
Author: Ken Dropiewski
FLEX Vessel Prep™ System Data Presented During Leipzig Interventional Course (LINC) 2021
Trio of studies demonstrate that FLEX Vessel Prep System safely and effectively modifies obstructive plaque to facilitate delivery of definitive therapies MINNEAPOLIS, Jan. 27, 2021 /PRNewswire/ — VentureMed Group, Inc. (VentureMed), a privately-held medical device innovator, announced today that data from three studies evaluating the use of its FLEX Vessel Prep (VP) System were […]
Valcare Medical Announces First-in-Human Transseptal Implant of the AMEND™ Annuloplasty Ring for Mitral Valve Repair
HERZLIYA, Israel, Jan. 27, 2021 /PRNewswire/ — Valcare Medical Ltd. developer of transcatheter mitral and tricuspid valve repair and replacement solutions, announces that it has successfully completed its first-in-human transseptal delivery of the AMEND™ annuloplasty ring. The transseptal AMEND procedure was performed at the Schulich Heart Centre at Sunnybrook Health Sciences Centre in Toronto, under […]
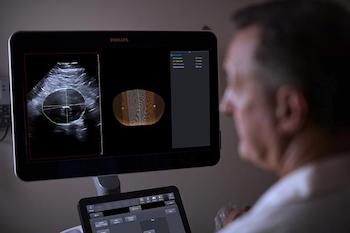
Philips integrates 3D ultrasound with innovative software for breakthrough in surveillance of abdominal aortic aneurysms
Philips Abdominal Aortic Aneurysm (AAA) Model helps increase diagnostic confidence and improved patient experience compared to current standard of care Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, has introduced the Philips Abdominal Aortic Aneurysm (AAA) Model, providing physicians a more patient-friendly solution compared to […]
Microbot Medical Announces Successful Outcome of its Discussions with FDA Regarding Regulation of Self-Cleaning Shunt
FDA Feedback Reflects Strength of Pre-Clinical Safety Data; Maintains Q3 2022 Projected Commencement of First-in-Human Clinical Trial HINGHAM, Mass., Jan. 27, 2021 (GLOBE NEWSWIRE) — Microbot Medical Inc. (Nasdaq: MBOT) announced the completion of successful discussions with the U.S. Food and Drug Administration (FDA) for its Self-Cleaning Shunt (SCS). After review […]
Elixir Medical Announces Outstanding 24-month Data for DynamX Coronary Bioadaptor System, Demonstrating Strong Safety With No Target Lesion Revascularization, Myocardial Infarction or Thrombosis Through 24 Months
Successful DynamX Bioadaptor Live Case Presented at Singapore Live MILPITAS, Calif.–(BUSINESS WIRE)–Elixir Medical, a developer of innovative, drug-eluting cardiovascular devices, today announced the 24-month clinical results for the DynamX™ Coronary Bioadaptor System, the first drug-eluting coronary artery implant that adapts to vessel physiology. The results were presented at the 30th Annual Live […]
ZOLL Foundation Grants to Nearly 70 Recipients in Resuscitation and Critical Care Research Surpass $2.5 Million
15 Grantees Awarded Nearly $500,000 in Final 2020 Application Round CHELMSFORD, Mass.–(BUSINESS WIRE)–In the application round ended September 30, 2020, the ZOLL Foundation, established in 2014 to support young investigators through research grants in the fields of resuscitation and acute critical care, received 35 applications from across the United States, […]
SoundBite Medical Solutions Announces First Use of its Novel 0.014” Active Wire to Successfully Treat Calcified Below-The-Knee CTOs
MONTREAL–(BUSINESS WIRE)–SoundBite Medical Solutions Inc. (SBMS) announced today the first use of its novel Active Wire 0.014” platform in the successful treatment of patients suffering from critical limb ischemia (CLI) with heavily calcified below-the-knee (BTK) chronic total occlusions (CTO). The procedures were performed by Professor Marianne Brodmann, Head of the […]
Foldax Biopolymer Shown to Possess Ideal Properties for Heart Valve in New Published Paper
Company’s LifePolymer Material Used in First Polymer Heart Valves Ever Approved by FDA for Use in Clinical Trials SALT LAKE CITY–(BUSINESS WIRE)–Foldax®, Inc. today announced publication of a research paper in Advanced NanoBiomed Research that concluded that its LifePolymer™ biopolymer “exhibits ideal biomaterial properties for the flexible leaflets of a totally synthetic […]
Caladrius Biosciences Closes $25.0 Million Private Placement
BASKING RIDGE, N.J., Jan. 25, 2021 (GLOBE NEWSWIRE) — Caladrius Biosciences, Inc. (Nasdaq: CLBS) (“Caladrius” or the “Company”), a clinical-stage biopharmaceutical company dedicated to the development of cellular therapies designed to reverse disease, today announced that it has closed on its previously announced sale of an aggregate of 12,500,000 shares of […]