Appointments Include: Chair, Clay Johnston, M.D., Ph.D. as well as Members Greg Albers, M.D. and Brett Cucchiara, M.D. AUSTIN, Texas, Jan. 19, 2021 (GLOBE NEWSWIRE) — XBiotech Inc. (NASDAQ: XBIT) announced today the first meeting of its Cerebrovascular Medical Advisory Board (CMAB). The CMAB is comprised of key opinion leaders […]
Author: Ken Dropiewski
InCarda Therapeutics Announces Enrollment of First U.S. Patient in Phase 2 INSTANT Trial of InRhythm™ for Treatment of Atrial Fibrillation
Ongoing Trial to Evaluate Optimal Dose Selected from Dose-Ranging Stage of the INSTANT Study for Rapid Conversion of Atrial Fibrillation to Normal Sinus Rhythm SAN FRANCISCO, Jan. 19, 2021 (GLOBE NEWSWIRE) — InCarda Therapeutics, Inc. (“InCarda”), a privately-held biopharmaceutical company developing first-of-their-kind inhaled therapies for cardiovascular diseases, today announced dosing […]
CHF Solutions Appoints Nestor Jaramillo, Jr. as President and Chief Executive Officer
EDEN PRAIRIE, Minn., Jan. 19, 2021 (GLOBE NEWSWIRE) — CHF Solutions (Nasdaq: CHFS), a medical device company dedicated to changing the lives of patients suffering from fluid overload, today announced the appointment of President and Chief Operating Officer Nestor Jaramillo, Jr. to President and Chief Executive Officer (CEO), effective immediately. […]
Caladrius Biosciences Treats First Patient in the Phase 2b FREEDOM Trial of CLBS16 for the Treatment of Coronary Microvascular Dysfunction
Initiation of the Phase 2b FREEDOM trial represents the next step in development of CLBS16 as a potential breakthrough treatment for the millions of sufferers of CMD in the U.S., most of whom are women Patient enrollment underway at The Christ Hospital Health Network in Cincinnati (OH) and Mayo Clinic […]
Teleflex continues to deliver COMPLETE confidence with new Arrow® ErgoPack® Complete MAC, PSI Systems
Teleflex expands value of ErgoPack® Complete enhancements to critical vascular access products WAYNE, Pa., Jan. 19, 2021 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE: TFX) continues to deliver COMPLETE confidence with the release of the Arrow® ErgoPack® Complete Multi-Lumen Access Catheter (MAC) System and the Arrow® ErgoPack® Complete Percutaneous Sheath Introducer (PSI) System. The release […]
Home-based Heart Monitoring Now Available to All Canadians Through Icentia
Canadian company drives at-home cardiac monitoring nationwide during pandemic QUEBEC CITY, Jan. 19, 2021 (GLOBE NEWSWIRE) — Icentia Inc., a leading Canadian technology company—specialised in cardiac ambulatory monitoring—will be providing a new home-based service nationwide that will enable patients with heart rhythm disorders to have access to an ambulatory cardiac […]
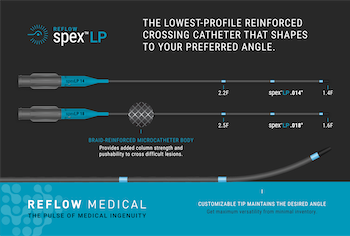
Reflow Medical Introduces the Spex™ LP, the Lowest Profile Shapeable Reinforced Support Catheter
SAN CLEMENTE, Calif.–(BUSINESS WIRE)–Reflow Medical, Inc., a California-based medical device company, introduces the Reflow™ Spex™ LP (Low Profile) 0.014 and 0.018-inch reinforced support catheters. The new Spex LP is engineered to provide the lowest profile tip for accessing and crossing the tightest and most complex lesions with a supportive system. It also […]
Microbot Medical Announces Positive Results of Additional Animal Feasibility Study with LIBERTYTM Robotic System
Results Validate Findings of Initial Study; End Points Achieved with No Intraoperative Adverse Events Management Presenting at Needham Growth Conference Today at 2:00pm ET HINGHAM, Mass., Jan. 14, 2021 (GLOBE NEWSWIRE) — The continued progression of Microbot Medical Inc.’s (Nasdaq: MBOT) LIBERTY™ Robotic System was further demonstrated as the Company announced […]
Marizyme Announces Approval for DuraGraft® in India
Latest Approval Joins a Growing List of Countries for Commercial Sales in 2021 JUPITER, Fla., Jan. 14, 2021 /PRNewswire/ — Marizyme, Inc. (OTCQB:MRZM), a publicly traded global biotechnology company developing products to reduce the burden of ischemia-reperfusion injury in tissue grafting, organ transplant, and other surgical indications, announced today that its flagship product DuraGraft® is […]
Lexicon Pharmaceuticals Provides Regulatory Update on Sotagliflozin in Heart Failure
THE WOODLANDS, Texas, Jan. 14, 2021 (GLOBE NEWSWIRE) — Lexicon Pharmaceuticals, Inc. (Nasdaq: LXRX) announced today that it has received U.S. Food and Drug Administration (FDA) regulatory feedback that the results of its SOLOIST and SCORED Phase 3 clinical studies can support a new drug application (NDA) submission for an indication to […]