Expansion includes adolescent patients, as young as 12 years of age, who can benefit from cryo nerve block therapy during surgical procedures involving the chest wall. MASON, Ohio–(BUSINESS WIRE)–AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in treatments for atrial fibrillation (Afib) and left atrial appendage (LAA) management, today announced that […]
Author: Ken Dropiewski
BioCardia Issued New U.S. Patent for Technology that Enhances Control for Cardiac Therapies
Technology Anticipated to Impact Cardiac Biologic Therapies, Cardiac Arrhythmia, and Structural Heart Procedures SAN CARLOS, Calif., Dec. 30, 2020 (GLOBE NEWSWIRE) — BioCardia®, Inc. [Nasdaq: BCDA] a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today announced the issuance of U.S. Patent No: 10,874, 831 for “Devices and Methods for […]
Teleflex Completes Acquisition of Market Leader in Hemostatic Products
WAYNE, Pa., Dec. 28, 2020 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE:TFX), a leading global provider for healthcare supplies and services, announced today that it has completed the previously announced acquisition of Z-Medica, LLC, an industry-leading manufacturer of hemostatic products. On October 28, 2020, Teleflex announced a definitive agreement to acquire Z-Medica. Under the […]
CARMAT Receives the CE Marking for Its Total Artificial Heart
CE marking allows the company to market its total artificial heart in the EU as a bridge to transplant Virtual press conference scheduled on January 6, 2021 PARIS–(BUSINESS WIRE)–Regulatory News: CARMAT (FR0010907956, ALCAR) (Paris:ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to fulfill an […]
Cytokinetics Regains European Rights to Omecamtiv Mecarbil
Company is Planning Regulatory Interactions in 2021 to Discuss Results of GALACTIC-HF and Expects to Evaluate Strategic Options for Co-Commercialization and Licensing SOUTH SAN FRANCISCO, Calif., Dec. 23, 2020 (GLOBE NEWSWIRE) — Cytokinetics, Incorporated (Nasdaq: CYTK) announced that Amgen Inc. (“Amgen”) notified the company that Les Laboratoires Servier and Institut […]
MicroVention, Inc. Files Trade Secret Lawsuit Against Balt USA, LLC
ALISO VIEJO, Calif., Dec. 23, 2020 /PRNewswire/ — MicroVention, Inc., a U.S. based subsidiary of Terumo and leading global neurovascular company, announced today the filing of a second lawsuit against Balt USA, LLC and four former employees for trade secret misappropriation. This lawsuit follows an earlier patent lawsuit filed against Balt USA in July 2019 that is ongoing. […]
Rapid Medical Receives FDA Clearance for the World’s First Steerable Neurovascular Guidewire
DRIVEWIRE™ provides efficient access in difficult neuro and peripheral vascular procedures YOKNEAM, Israel & MIAMI–(BUSINESS WIRE)–Rapid Medical, a company focused on the development of the first class of responsive, adjustable neurovascular devices, announces the FDA clearance of DRIVEWIRE, a novel guidewire with a steerable distal tip that handles complex anatomical […]
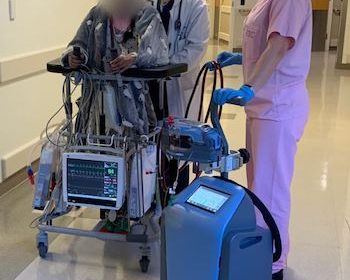
First Patients, Including a COVID-19 Patient, Treated with Abiomed’s Innovative ECMO Technology
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed (NASDAQ: ABMD) announces the first two patients in the world have been treated with the Abiomed Breethe OXY-1 System, a compact cardiopulmonary bypass system. The advanced ECMO technology pumps, oxygenates, and removes carbon dioxide from blood for patients whose lungs can no longer provide sufficient end organ oxygenation. The […]
The Children’s Heart Foundation funds new congenital heart defect research
Since 1996, the Foundation has funded nearly $14 million in research on congenital heart defects–America’s most common birth defect. NORTHBROOK, Ill., Dec. 22, 2020 /PRNewswire/ — The Children’s Heart Foundation, the nation’s leading organization dedicated to funding congenital heart defect (CHD) research, will fund over $735,000 in CHD research and scientific collaborations this year across […]
TricValve® Transcatheter Bicaval Valves System granted Designation as Breakthrough Device by the U.S. Food and Drug Administration (FDA)
TricValve® made in Europe VIENNA, Dec. 22, 2020 /PRNewswire/ — P+F Products + Features GmbH has been granted designation as a Breakthrough Device for the company’s lead product, the TricValve® Transcatheter Bicval Valves System by the U.S. Food & Drug Administration on December 15th 2020. The TricValve® is a system of two self-expanding biological valves for […]