Mavacamten Is a Potential First-in-Class Medicine with Compelling Data in the Treatment of Patients with Symptomatic Obstructive Hypertrophic Cardiomyopathy Mavacamten Will Be a Medium- and Long-Term Growth Driver Presenting a Significant Commercial Opportunity upon Approval Promising Portfolio of Pipeline Candidates Strengthens and Extends Bristol Myers Squibb’s Leading Cardiovascular Franchise Expected to […]
Author: Ken Dropiewski
NorthStar Medical Technologies, LLC Announces Promotion of CFO Paul Estrem to Executive Vice President and Appointment to NorthStar Board of Managers
BELOIT, Wis.–(BUSINESS WIRE)–NorthStar Medical Technologies, LLC, a global innovator in the development, production and commercialization of diagnostic and therapeutic radiopharmaceuticals, today announced the promotion of Chief Financial Officer Paul Estrem, to Executive Vice President. Mr. Estrem has also been appointed to the NorthStar Board of Managers, effective October 12, 2020, […]
BioVentrix Announces the Appointment of Cardiac Device Veteran Chris Wolff as Vice President of U.S. Sales
SAN RAMON, Calif.–(BUSINESS WIRE)–BioVentrix, Inc., a privately held medical device company focused on treating congestive heart failure (CHF) via Transcatheter Ventricular Restoration (TCVR), today announced that Chris Wolff has been named Vice President of U.S. Sales. In this role, Mr. Wolff will be responsible for driving enrollment in the ongoing ALIVE Trial […]
Amgen Announces Collaboration With Two NHS Organisations to Help Improve Cholesterol Management for People at High Risk of a Heart Attack or Stroke
‘Control Cholesterol’ to Support the NHS in Delivering on Long-Term Plan Ambitions and Improve Health Outcomes for Patients in the North West Coast of England Quality Management Approach Aims to Help Predict and Prevent Heart Attacks and Strokes For More Than 300,000 People With Dangerously High Cholesterol Levels CAMBRIDGE, England–(BUSINESS […]
Medicure announces an agreement with Reliance Life Sciences for the marketing rights of a cardiovascular biosimilar
WINNIPEG, AB, Oct. 5, 2020 /PRNewswire/ – Medicure Inc. (“Medicure” or the “Company”) (TSXV: MPH) (OTC: MCUJF), a pharmaceutical company, announces that through its wholly-owned subsidiary, Medicure International Inc., it has entered into a License, Manufacture and Supply Agreement (the “Agreement”) with Reliance Life Sciences Private Limited (“RLS”) for a cardiovascular biosimilar (the […]
Silk Road Medical Names Rick Anderson to Board of Directors
SUNNYVALE, Calif., Oct. 01, 2020 (GLOBE NEWSWIRE) — Silk Road Medical, Inc. (Nasdaq: SILK), a company focused on reducing the risk of stroke and its devastating impact, today announced that medical device industry veteran Rick Anderson has joined its board of directors. Anderson brings over 30 years of senior executive […]
Cytokinetics Announces Publication of Manuscript Detailing Baseline Characteristics of GALACTIC-HF in European Journal of Heart Failure
Heart Failure Patients in Phase 3 Clinical Trial of Omecamtiv Mecarbil Are at High Risk of Cardiovascular Events and Symptom Burden SOUTH SAN FRANCISCO, Calif., Oct. 02, 2020 (GLOBE NEWSWIRE) — Cytokinetics, Incorporated (Nasdaq: CYTK) today announced that a manuscript detailing the baseline characteristics from GALACTIC-HF (Global Approach to Lowering Adverse Cardiac Outcomes Through Improving Contractility in Heart Failure), the Phase […]
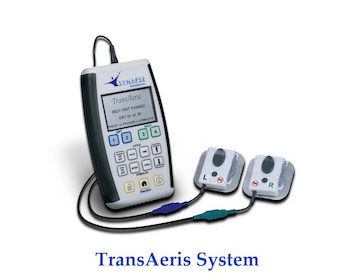
Diaphragm Pacing can Enhance Recovery and Weaning from Mechanical Ventilation (MV) in Cardiac Surgery
First Report of TransAeris® Diaphragm Pacer (DP) in High Risk Cardiac Surgeries under FDA Emergency Use Authorization OBERLIN, Ohio, Oct. 1, 2020 /PRNewswire/ — Synapse Biomedical, Inc®. (www.synapsebiomedical.com) announced today that surgeons at University Hospitals Cleveland Medical Center have improved the prognosis of several cardiac patients after the use of TransAeris DP […]
Medtronic Resolute Onyx™ Drug Eluting Stent (DES) First to Receive One-Month DAPT Labeling in the U.S. with Expanded Indication for High Bleeding Risk Patients
DUBLIN, Oct. 1, 2020 /PRNewswire/ — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received U.S. Food and Drug Administration (FDA) approval for new one-month of dual-antiplatelet therapy (DAPT) labeling with an expanded indication for high bleeding risk (HBR) patients implanted with the Resolute Onyx™ DES. The Resolute Onyx DES […]
China National Medical Products Administration (NMPA) Grants Innovative Medical Device Designation for VEST in China
TEL AVIV, Israel, Oct. 1, 2020 /PRNewswire/ — Vascular Graft Solutions Ltd. (VGS), an Israel-based innovative commercial-stage medical device company, announced today that the China National Medical Products Administration (NMPA) granted Innovative Medical Device Designation for VEST™, an external stent for vein grafts in coronary artery bypass surgery. VEST™ was proven to mitigate […]