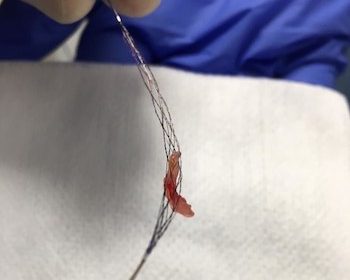
TIGERTRIEVER XL is the largest stentriever available, designed to safely remove clots from intracranial vessels up to 9mm in diameter YOKNEAM, Israel, July 27, 2020 /PRNewswire/ — Rapid Medical, a company focused on the development of next generation neurovascular devices, has announced that it received CE Mark for TIGERTRIEVER XL. In addition, the first […]
Author: Ken Dropiewski
Endonovo Therapeutics Announces Significant Results From Study Showing 70% Reduction of Morphine Equivalent Dose and 2 Day Reduction of Length of Stay at Hospitals Using SofPulse®
LOS ANGELES, July 27, 2020 (GLOBE NEWSWIRE) — Endonovo Therapeutics, Inc. (OTCQB: ENDV) (“Endonovo” or the “Company”), today announced that an investigator initiated study, authored by Dr. Samir Awad, Professor of Surgery at Baylor College of Medicine, entitled ‘The Impact of Pulsed Electromagnetic Field (SofPulse®) on Postoperative Pain in Patients […]
VASCEPA® (icosapent ethyl) Data from REDUCE-IT® REVASC, Including New Data on Timeframe to Demonstrated Benefit, Presented as Encore at The American Society for Preventive Cardiology 2020 Virtual Summit on CVD (Cardiovascular Disease) Prevention
VASCEPA showed significant reduction in coronary revascularization, including coronary stenting and cardiac bypass surgery, in prespecified and post hoc analyses of landmark REDUCE-IT study First and total coronary revascularization event reductions of 34% and 36%, respectively, shown with VASCEPA in prespecified tertiary endpoint analyses, with results consistent across different types […]
DalCor announces dal-GenE trial continues with final data expected in the first half of 2021
LONDON and MONTREAL, July 27, 2020 (GLOBE NEWSWIRE) — DalCor Pharmaceuticals today announced that the independent dal-GenE Data and Safety Monitoring Board (DSMB) recommends the dal-GenE pivotal outcomes trial should continue as planned with no modifications. This is an important milestone for DalCor and for cardiovascular patients with the ADCY9 […]
Transformative Raises $1.7M Seed Round After Developing Technology That Predicts Sudden Cardiac Arrest
NEW YORK–(BUSINESS WIRE)–Transformative, a healthcare startup whose software predicts sudden cardiac arrest, announced today that it has raised a $1.7 million seed round, led by Tera Ventures. The round includes funding from the Wellcome Trust, which provided Transformative a Healthcare Innovator Award, and from InHealth Ventures, which had previously invested […]
scPharmaceuticals Announces FDA Acceptance of FUROSCIX® New Drug Application Resubmission
FDA sets PDUFA date of December 30, 2020 BURLINGTON, Mass.–(BUSINESS WIRE)–scPharmaceuticals Inc. (Nasdaq: SCPH), a pharmaceutical company focused on developing and commercializing products that have the potential to optimize the delivery of infused therapies, advance patient care, and reduce healthcare costs, today announced that the U.S. Food and Drug Administration […]
Synchron Secures Further Funding from the Australian Government to Advance Commercialization of Stentrode
Synchron’s Stentrode is a fully-implantable brain-computer interface being designed to enable people with paralysis to restore functional independence by engaging in activities of daily living SAN FRANCISCO & NEW YORK CITY & MELBOURNE, Australia–(BUSINESS WIRE)–Synchron Inc., a neurovascular bioelectronic medicine company, today announced that the Company has been awarded nearly AUD$1M […]
SANUWAVE Health, Inc. Announces Successful Results of Annual Meeting of Stockholders
SUWANEE, GA, July 24, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — SANUWAVE Health, Inc. (OTCQB: SNWV) (“SANUWAVE” or the “Company”) today announced the results from the Company’s 2020 Annual Meeting of Stockholders, held Thursday, July 23, 2020. All director nominees, including Kevin A. Richardson II, John F. Nemelka, Alan L Rubino, A. Michael […]
LeMaitre Vascular Announces Q2 2020 Financial Results
BURLINGTON, Mass., July 23, 2020 (GLOBE NEWSWIRE) — LeMaitre Vascular, Inc. (Nasdaq:LMAT), a provider of vascular devices, implants and services, today reported Q2 2020 results and announced a $0.095/share quarterly dividend. Q2 2020 Results Sales of $24.9mm, -16% (-24% organic) vs. Q2 2019 Op. income of $4.9mm, -18% Adjusted op. income of $6.1mm, […]
Neovasc Announces Partial Prepayment of Convertible Debenture
VANCOUVER and MINNEAPOLIS, July 23, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN) announced today that Strul Medical Group LLC (“SMG”) exercised 1,424,049 of the 2,573,959 common share purchase warrants (the “May Warrants”) issued pursuant to a Securities Purchase Agreement dated May […]