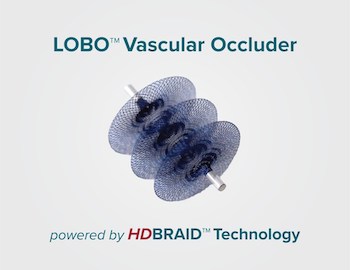
ALISO VIEJO, Calif., June 24, 2020 /PRNewswire/ — Okami Medical Inc., announced today the close of a Series D financing totaling $7.2 million. The financing was led by U.S. Venture Partners and was joined by members of Okami’s board of directors and medical device industry veterans. The financing provides Okami, the second portfolio company of […]
Author: Ken Dropiewski
Windtree Announces Initial European Trial Sites Activated in AEROSURF® Bridging Study and a Collaboration for a Concomitant Independent Cerebral Physiology Study
WARRINGTON, Pa., June 24, 2020 /PRNewswire/ — Windtree Therapeutics, Inc. (NasdaqCM: WINT), a biotechnology and medical device company focused on developing drug product candidates and medical device technologies to address acute cardiovascular and pulmonary diseases, today announced that all initial European trial sites are active and enrolling or able to enroll patients into […]
BAROSTIM™ NEO Associated with Very High Responder Rates in Women
BeAT-HF Trial Results Presented as Late-Breaking Clinical Science at ESC Heart Failure 2020 Meeting MINNEAPOLIS, June 23, 2020 /PRNewswire/ — CVRx, Inc., a private medical device company, announced new BeAT-HF trial results from late-breaking clinical science presented virtually at the European Society of Cardiology Heart Failure 2020 Scientific Sessions. The BAROSTIM NEO […]
Biomerics Launches Online Store for Medical Balloons
SALT LAKE CITY, June 24, 2020 /PRNewswire/ — Biomerics, LLC, a mid-market medical device contract manufacturer specializing in interventional and radiology markets, launched an online store to sell medical balloons. This new e-Commerce platform will sell a range of off-the-shelf Nylon, PET, and LDPE medical balloons. The fast-turn store enables engineers to […]
preCARDIA, Inc. Receives FDA Breakthrough Device Designation for Novel, Catheter Based Heart Failure Treatment
Pioneering technology is designed to address Acute Decompensated Heart Failure ST. PAUL, Minn., June 23, 2020/PRNewswire/ — preCARDIA, Inc., has announced that the company’s catheter based system for treating volume overload in patients with acutely decompensated heart failure (ADHF) has been designated for the Breakthrough Devices Program by the U.S. […]
Ablacon, Inc. Announces the Formation of Scientific Advisory Board
Scientific advisory board composed of thought leaders in electrophysiology and treatment of atrial fibrillation MENLO PARK, Calif., June 23, 2020 /PRNewswire/ — Ablacon, Inc., an Ajax Health-backed company, announced today the formation of its Scientific Advisory Board (SAB). The SAB will be composed of four internationally recognized thought leaders in the field of electrophysiology […]
Bendit Technologies Announces Senior Management Appointments
Richard Cappetta has been appointed Chairman of the Board and Yossi Mazel will be the new CEO TEL AVIV, Israel, June 23, 2020 /PRNewswire/ — Bendit Technologies, an industry leader in the development of steerable microcatheters, announced today that Richard Cappetta has been appointed as its Chairman of the Board. Together with this appointment, it was announced that Yossi […]
Philips receives FDA premarket approval for its HeartStart FR3 and HeartStart FRx automated external defibrillators
Amsterdam, the Netherlands and Cambridge, MA – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that the Center for Devices and Radiological Health (CDRH) of the U.S. Food and Drug Administration (FDA) has granted premarket approval (PMA) for the company’s HeartStart FR3 [1] and HeartStart FRx [2] […]
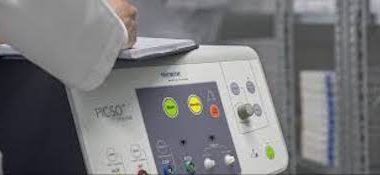
Miracor Medical’s PiCSO® System Receives CE Mark
AWANS, Belgium–(BUSINESS WIRE)–Miracor Medical SA (Miracor Medical) has announced the award of the CE Mark for its latest generation of PiCSO Impulse Catheter and PiCSO Impulse Console, which is indicated for the treatment of anterior STEMI patients. The new system features improvements in ease of use. PiCSO therapy is delivered […]
TARA Biosystems Announces $10 Million Series A2 Financing to Expand its Cardiac Disease Drug Discovery and Development Capabilities
NEW YORK–(BUSINESS WIRE)–TARA Biosystems, Inc., a company generating in vitro human biology to increase the success of cardiac disease drug discovery and development, today announced a $10 million Series A2 financing. This financing will be used to scale TARA’s cardiac tissue engineering platform technology and advance its drug discovery programs. New investors […]