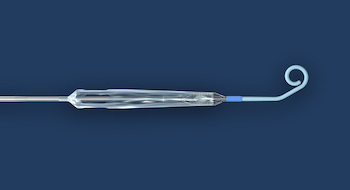
Results indicate that 12 months after carotid intervention the CGuard EPS MicroNET-covered stent delivers sustained protection against postprocedural neurologic events TEL AVIV, Israel, June 10, 2020 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE American: NSPR), developer of the CGuard™ Embolic Prevention System (EPS) for the prevention of stroke caused by carotid […]
Author: Ken Dropiewski
Saranas Announces Peer-Reviewed Publications Highlighting Accuracy and Clinical Utility of Early Bird® Bleed Monitoring System
HOUSTON–(BUSINESS WIRE)–Saranas, Inc., developer of the Early Bird® Bleed Monitoring System for the monitoring and early detection of internal bleed complications during endovascular procedures, announced the online publication of two peer-reviewed articles highlighting the system’s accuracy and performance. The publications provide evidence of the Early Bird’s reliability to detect the early […]
LindaCare Appoints Donald Deyo to Executive Chairman and Head of US Operations
LEUVEN, Belgium and STAMFORD, Conn., June 9, 2020 /PRNewswire/ — LindaCare is pleased to announce the appointment of Donald L. Deyo into the new roles of Executive Chairman of the LindaCare NV and President and CEO of the US subsidiary, LindaCare Inc. Deyo has served as Chairman of the Board for the Company since 2017. LindaCare […]
Ardelyx Appoints Veteran Biopharma Executive, Justin Renz, as Chief Financial Officer
FREMONT, Calif., June 8, 2020 /PRNewswire/ — Ardelyx, Inc. (Nasdaq: ARDX), a specialized biopharmaceutical company focused on developing innovative first-in-class medicines to improve treatment for people with kidney and cardiovascular diseases, today announced the appointment of Justin Renz to the position of Chief Financial Officer. Mr. Renz, a highly accomplished leader with over 20 years of […]
XyloCor Therapeutics Names Alexander Gaidamaka, PhD, Senior Vice President of Technology, Manufacturing & Quality
PHILADELPHIA–(BUSINESS WIRE)–XyloCor Therapeutics, a private clinical‑stage biopharmaceutical company focused on the development of gene therapy for unmet needs in cardiovascular disease, strengthens its leadership team with the appointment of Alexander Gaidamaka, PhD, as Senior Vice President of Technology, Manufacturing & Quality. Dr. Gaidamaka brings a wealth of product development, regulatory […]
Shockwave Medical Announces That CMS Has Created New Codes for Intravascular Lithotripsy
Centers for Medicare & Medicaid Services Issues New Codes for Peripheral Intravascular Lithotripsy Performed in Hospital Outpatient and Inpatient Settings SANTA CLARA, Calif., June 08, 2020 (GLOBE NEWSWIRE) — Shockwave Medical, Inc. (Nasdaq: SWAV), a pioneer in the development and commercialization of Intravascular Lithotripsy (IVL) to treat complex calcified cardiovascular […]
FDA Approves Abiomed’s First-in-Human Trial of Impella ECP, World’s Smallest Heart Pump
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed (NASDAQ: ABMD) announces the United States Food and Drug Administration (FDA) has approved the company’s investigational device exemption application to start an early feasibility study with a first-in-human trial of the 9 French (Fr) Impella ECP™ heart pump. Impella ECP, which stands for expandable cardiac power, will be […]
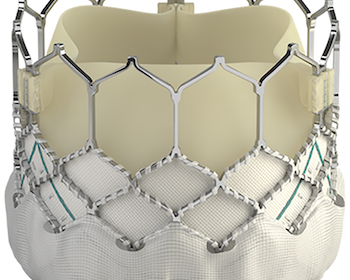
Edwards SAPIEN 3 Transcatheter Heart Valve Receives Approval In China
IRVINE, Calif., June 8, 2020 /PRNewswire/ — Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced that it has received Chinese regulatory approval for the Edwards SAPIEN 3 transcatheter heart valve for the treatment of patients suffering from severe, symptomatic aortic […]
Surmodics Receives CE Mark for its SurVeil™ Drug Coated Balloon
Next generation device adds new technology to how physicians approach treatment of PAD EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ:SRDX), a leading provider of medical device and in vitro diagnostic technologies to the health care industry, today announced it has received CE Mark Certification in the European Union for its SurVeil™ […]
Medtronic Resolute Onyx™ DES Receives First and Only One-Month DAPT Indication for High Bleeding Risk Patients in Europe
DUBLIN, June 05, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received CE (Conformité Européenne) Mark for a one-month dual antiplatelet therapy (DAPT) indication for high bleeding risk (HBR) patients implanted with the Resolute Onyx™ Drug-Eluting Stent (DES). For HBR patients, whose bleeding risk may be increased by taking longer DAPT […]