Pharmaceutical industry leader to join as the Company prepares for the Phase II clinical trial with merimepodib, an orally administered broad-spectrum anti-viral for COVID-19 Westport, CT, May 28, 2020 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM) (“BioSig” or the “Company”) today appointed Mr. Michael R. Dougherty to the Board […]
Author: Ken Dropiewski
Philips Foundation publishes its 2019 Annual Report
May 28, 2020 Support provided by the Philips Foundation helped 5 million more people to access one or more healthcare services in 2019 Almost 900,000 lives were improved as a result of the foundation’s 2018-2019 employee volunteering program to fight childhood pneumonia Additional 38 projects launched in 2019 to explore […]
Caladrius Biosciences Closes $4.3 Million Registered Direct Offering Priced At-The-Market under Nasdaq Rules
BASKING RIDGE, N.J., May 28, 2020 (GLOBE NEWSWIRE) — Caladrius Biosciences, Inc. (Nasdaq: CLBS) (“Caladrius” or the “Company”), a clinical-stage biopharmaceutical company dedicated to the development of cellular therapies designed to reverse, not manage, disease, today announced that it has closed on its previously announced sale of an aggregate of […]
Premier Inc. ProvideGx™ Program Partners with Hikma Pharmaceuticals to Secure the Supply of Two Drugs Essential for COVID-19 Patient Care
CHARLOTTE, N.C.–(BUSINESS WIRE)–Premier Inc. (NASDAQ: PINC), through its ProvideGx™ program, has partnered with Hikma Pharmaceuticals PLC (Hikma, Group) to supply amiodarone and sterile water to healthcare providers, helping to stabilize the long-term supply of two medications vital for COVID-19 patient care. Amiodarone is used to manage late onset cardiac arrest and […]
PhaseBio Launches Clinical Trial to Evaluate PB1046 as a Treatment for Hospitalized COVID-19 Patients
PhaseBio receives clearance of IND application from U.S. FDA under Coronavirus Treatment Acceleration Program (CTAP) “VANGARD” trial will assess the efficacy and safety of PB1046 in hospitalized COVID-19 patients at high risk for rapid clinical deterioration and acute respiratory distress syndrome Based on FDA feedback, PhaseBio believes that positive, clearly […]
AngioSoma Focusing on Development of Coating for Metal Stents
Houston, TX, May 27, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — AngioSoma, Inc. (OTC: SOAN) (“AngioSoma” or the “Company”) is pleased to announce that the Company is focusing on development and commercialization of our coating for metal stents. A stent is an expandable “scaffold-like” device, usually constructed of a metallic material, that […]
Alucent Biomedical Announces First Patient Enrolled in Phase 1 Natural Vascular Scaffolding Clinical Trial Multicenter ACTIVATE I Study to Enroll up to 15 Patients in t
SALT LAKE CITY–(BUSINESS WIRE)–Alucent Biomedical Inc. announced that it has enrolled the first two patients in ACTIVATE I, a Phase 1 clinical trial to evaluate the safety and efficacy of its revolutionary Natural Vascular Scaffolding (NVS) technology. The therapy is designed to open vessels and keep them open without the use […]
Merit Medical Announces Agreement With Starboard
Nominates Three Medical Device Industry Veterans – Lonny Carpenter, David Floyd and James Hogan – for Election to the Board of Directors Announces Formation of Operating Committee to Review Margin Improvement Initiatives and Establish Targets SOUTH JORDAN, Utah, May 26, 2020 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ: MMSI), a […]
Neovasc Announces Offering of Convertible Debt and Warrants for Gross Proceeds of up to US$5.0 Million, Warrant Exchange Transaction and Repayment of 2017 Convertible Notes and Related Settlement
VANCOUVER and MINNEAPOLIS, May 26, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies, and minimally invasive devices for the treatment of refractory angina, announced today 1) an offering (“Offering”) of convertible notes […]
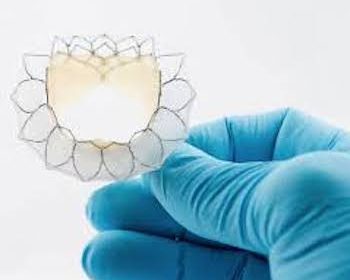
Neovasc Has Filed for CE Mark for Tiara TA Transapical Mitral Valve Replacement System
Less invasive approach provides hope for the millions of patients that suffer from leaky mitral valves VANCOUVER and MINNEAPOLIS, MN, May 27, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies, and minimally invasive […]