SEATTLE, Jan. 23, 2020 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, announced today the commercial launch of the 14-Day version of the Carnation Ambulatory Monitor (“CAM™”), the industry’s only P-wave centric™ ambulatory cardiac patch monitor and arrhythmia detection device, following recent clearance by […]
Author: Ken Dropiewski
Study Finds SureFlex® Steerable Guiding Sheath has Greater Contact Force Stability for Radiofrequency Ablations
TORONTO, Jan. 23, 2020 /PRNewswire/ – A study has found that use of Baylis Medical’s SureFlex Steerable Guiding Sheath results in greater contact force stability for radiofrequency ablations in pulmonary vein isolations. Stable contact force has been linked to more effective ablations, a therapy in which radiofrequency energy is used to create a […]
JanOne Scientific Advisory Board Chair and Leading Pain Expert Featured on ABC-7 WJLA America This Week
Dr. Christopher Kevil, pioneer in nitrite based PAD treatment and Dr. Amol Soin, foremost expert is neuropathic pain, provide scientific insight on the pain relieving qualities of JanOne’s lead drug candidate Interview to be distributed nationally on all 193 Sinclair broadcasting network affiliate stations in 89 markets across the nation […]
Medtronic Receives FDA Approval for Trial Evaluating New Energy Source with Pulsed Electric Fields to Treat Atrial Fibrillation
Investigative Technology Designed to Interrupt Irregular Pathways in the Heart DUBLIN, Jan. 23, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced that it received approval from the U.S. Food and Drug Administration (FDA) to proceed with an investigational device exemption (IDE) trial to evaluate the safety and effectiveness of the PulseSelect™ Pulsed Field […]
CardioFocus® Names Burke T. Barrett As Chief Executive Officer
MARLBOROUGH, Mass., Jan. 22, 2020 /PRNewswire/ — CardioFocus, Inc., a medical device company dedicated to advancing ablation treatment for atrial fibrillation (AFib), announced today that the company’s Board of Directors has promoted Burke T. Barrett to the position of Chief Executive Officer (CEO), effective January 19, 2020. Mr. Barrett previously served as CardioFocus’ President and […]
Teleflex Receives FDA Clearance for Wattson™ Temporary Pacing Guidewire
WAYNE, Pa., Jan. 22, 2020 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE: TFX), a leading global provider of medical technologies for critical care and surgery, today announced that it received 510(k) clearance from the U.S. Food and Drug Administration for the WattsonTM Temporary Pacing Guidewire – the first commercially available bipolar temporary […]
Abbott Reports Fourth-Quarter 2019 Results; Announces Strong Forecast for 2020
– Fourth-quarter sales growth of 7.1 percent; organic sales growth of 8.5 percent – Fourth-quarter GAAP EPS growth from continuing operations of 59.5 percent and adjusted EPS growth from continuing operations of 17.3 percent – Several businesses achieved double-digit sales growth – Forecast reflects continued top-tier sales and earnings growth […]
SEQUANA MEDICAL LAUNCHES EQUITY PLACEMENT
THIS ANNOUNCEMENT IS NOT FOR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN THE UNITED STATES OF AMERICA, AUSTRALIA, CANADA, JAPAN, SOUTH AFRICA OR ANY OTHER JURISDICTION WHERE TO DO SO WOULD BE PROHIBITED BY APPLICABLE LAW PRESS RELEASE REGULATED INFORMATION – INSIDE INFORMATION 22 January 2020, 15:50 CET Ghent, Belgium, 22 January 2020 – Sequana Medical […]
Shockwave Medical to Report Fourth Quarter and Full Year 2019 Financial Results on February 13, 2020
SANTA CLARA, Calif., Jan. 21, 2020 (GLOBE NEWSWIRE) — Shockwave Medical, Inc. (Nasdaq: SWAV), a pioneer in the development and commercialization of Intravascular Lithotripsy (IVL) to treat complex calcified cardiovascular disease, today announced it will report financial results for the fourth quarter and full year 2019 after market close on […]
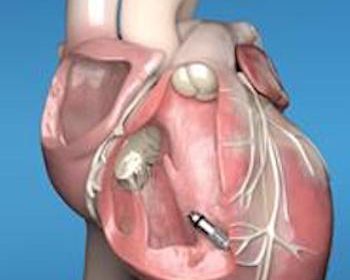
FDA Approves Medtronic Micra™ AV, the World’s Smallest Pacemaker Which Can Now Treat AV Block
With FDA Approval, More Patients in the U.S. Are Now Candidates for a Leadless Pacing Option DUBLIN, Jan. 21, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced it has received U.S. Food and Drug Administration (FDA) approval of Micra™ AV, the world’s smallest pacemaker with atrioventricular (AV) synchrony. Micra AV is […]