AUCKLAND, New Zealand–(BUSINESS WIRE)–Toku, Inc., a commercial medical device company specializing in imaging technology and AI, announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device designation to its patented CLAiR technology. The CLAiR platform, if cleared by the FDA, will be the first medical device in the US market that can provide affordable, point-of-care and non-invasive evaluation for risk of cardiovascular disease (CVD) using fundus retinal image
Coronary/Structural Heart
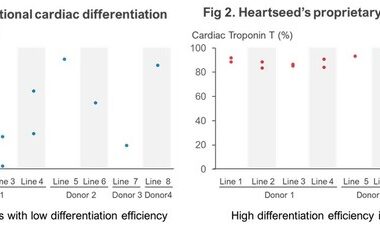
Using iPS Cells from I Peace, Heartseed Succeeds in Stable Production of High Purity Cardiomyocytes, A Major Step Forward in Advancing Autologous Cardiac Regenerative Medicine
PALO ALTO, Calif., Nov. 1, 2023 /PRNewswire/ — Leading GMP cell CDMO I Peace, Inc. (https://www.ipeace.com), specializing in induced pluripotent stem cells (iPSCs) and iPSC-derived cell therapies based in Palo Alto, CA, and Heartseed Inc. (“Heartseed”), a Tokyo-based biotechnology…
Silence Therapeutics Announces Positive Topline Results from Phase 1 Multiple Dose Study of Zerlasiran in Subjects with High Lipoprotein(a) and Stable Atherosclerotic Cardiovascular Disease
LONDON–(BUSINESS WIRE)—- $SLN #SenseofSilence–Silence Therapeutics plc, Nasdaq: SLN (“Silence” or the “Company”), an experienced and innovative biotechnology company committed to transforming people’s lives by silencing diseases through precision engineered medicines, today announced positive topline results from the multiple dose component of the APOLLO phase 1 study of zerlasiran (formerly SLN360) in 36 adults with baseline lipoprotein(a), or Lp(a), levels at or over 150 nmol/L and stable atherosclerotic cardiova
World’s First Patient Enrolled in SELUTION4DeNovo DEB IDE Trial
MIAMI LAKES, Fla., Oct. 31, 2023 /PRNewswire/ — Cordis has announced enrollment of the first patient in its SELUTION4DeNovo IDE trial, the coronary de novo study for SELUTION SLR™ Drug-Eluting Balloon (DEB), the company’s novel sirolimus-eluting balloon. SELUTION4DeNovo is the first DEB…
Toku joins Innovators’ Network at American Heart Association Center for Health Technology & Innovation
SAN DIEGO–(BUSINESS WIRE)–Toku Inc, a provider of an AI-based screening tool for detection of cardiovascular risk through retinal imaging, has joined the American Heart Association Center for Health Technology & Innovation’s (the Center) Innovators’ Network. The Center is focused on building and fostering health technology relationships to develop innovative and scalable solutions. The Innovators’ Network is a health care technology consortium that connects entrepreneurs, providers, resea
4DMT Gains Alignment with FDA on Plan to Lift Clinical Hold on Phase 1/2 INGLAXA Clinical Trial for 4D-310 for Fabry Disease Cardiomyopathy
Initiated single non-human primate (NHP) safety study evaluating intravenous 4D-310 combined with rituximab/sirolimus (R/S) immunosuppressive regimen Amended INGLAXA protocol to minimize risk of atypical hemolytic uremic syndrome (aHUS) associated with intravenous (IV) AAV dosing, including addition of R/S immunosuppressive regimen 4D-310 combines a novel, targeted next generation AAV vector (C102) […]
Peijia Medical Presents Early Clinical Findings of GeminiOne® TEER Device at TCT 2023
SUZHOU, China, Oct. 27, 2023 /PRNewswire/ — Peijia Medical Limited (Peijia, (9996.HK)), a leading Chinese medical device player in transcatheter structural heart device and neurovascular device, presented its GeminiOne® TEER technology, along with the early clinical experiences of the…
egnite, Inc. Unveils Research on Mitral Regurgitation at Transcatheter Cardiovascular Therapeutics Conference 2023
ALISO VIEJO, Calif.–(BUSINESS WIRE)– #digitalhealth–Key Findings: Patients with Moderate-to-Severe MR are 3x Less Likely to be Treated Than Patients with Severe MR Despite Similar Mortality
Boston Scientific AGENT™ Drug-Coated Balloon Demonstrates Superiority to Uncoated Balloon Angioplasty in the AGENT IDE Trial
Data from first coronary DCB study in U.S. meet 12-month primary endpoint, demonstrate low adverse event rates with AGENT DCB MARLBOROUGH, Mass. and SAN FRANCISCO, Oct. 25, 2023 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) today announced positive 12-month results from the…
Magenta Medical Reports Positive Results for US Early Feasibility Study of Elevate™ Heart Pump in Providing Temporary Mechanical Circulatory Support During High-Risk PCI Procedures Results presented at TCT 2023 in San Francisco
KADIMA, Israel and SAN FRANCISCO, Oct. 25, 2023 /PRNewswire/ — The US Early Feasibility Study of Magenta Medical’s Elevate™ System, designed to provide temporary mechanical circulatory support during high-risk percutaneous coronary intervention (HR-PCI) procedures, has successfully met…