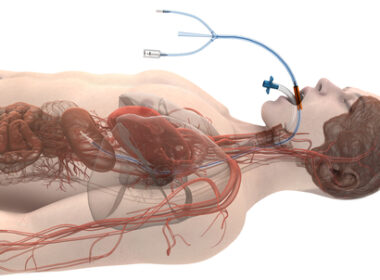
IRVINE, Calif., Sept. 25, 2023 (GLOBE NEWSWIRE) — Eledon Pharmaceuticals, Inc. (“Eledon”) (NASDAQ: ELDN) today announced that tegoprubart, the company’s investigational anti-CD40 antibody, was used as a cornerstone component of the chronic immunosuppressive regimen administered following the second-ever transplant of a genetically modified heart from a pig to a human. The procedure […]
Coronary/Structural Heart
Pi-Cardia Successfully Completes Enrollment in Pivotal Study for ShortCut™
REHOVOT, Israel–(BUSINESS WIRE)–Pi-Cardia Ltd., a global leader in the development of non-implant, catheter-based, leaflet modification solutions for treating heart valves, announced today the successful completion of its ShortCut™ Pivotal Study enrollment. ShortCut™ is the world’s first dedicated device designed to split the leaflets of pre-existing valves to enable safe Transcatheter […]
HeartSciences Signs Definitive Agreements with the Icahn School of Medicine at Mount Sinai to Commercialize Artificial Intelligence Cardiovascular Algorithms
Southlake, TX, Sept. 21, 2023 (GLOBE NEWSWIRE) — Heart Test Laboratories, Inc. d/b/a HeartSciences (Nasdaq: HSCS; HSCSW) (“HeartSciences” or the “Company”), an AI-powered medical technology company focused on transforming ECGs/EKGs to save lives through earlier detection of heart disease, today announced it has executed definitive agreements with the Icahn School of Medicine […]
Saranas Successfully Completes Enrollment of Clinical Trial Assessing the Utility of Early Bleed Detection in Patients Undergoing Mechanical Circulatory Support
HOUSTON–(BUSINESS WIRE)–Saranas, Inc. announced today that it has completed enrollment in SAFE-MCS, a multi-center, single arm, open-label clinical trial evaluating the safety of complex high-risk percutaneous coronary intervention (PCI) using mechanical circulatory support (MCS) and surveillance with the Early Bird® Bleed Monitoring System. The Early Bird Bleed Monitoring System is the […]
Us2.ai Partners with Leading Academic Medical Center to Collaborate on AI-Enabled Echocardiography Software
SINGAPORE–(BUSINESS WIRE)–Us2.ai, a Singapore-based MedTech firm, has partnered with Duke University to co-develop and commercialize AI tools for echocardiography, to enable earlier detection, improved diagnosis and more efficient management of heart disease. “This advances Duke University’s efforts in digitalising clinical trials” Tweet this In clinical practice, echocardiography is now firmly established as […]
Edgewise Therapeutics Begins Dosing First-in-Human Phase 1 Trial of EDG-7500, its Lead Clinical Candidate for Hypertrophic Cardiomyopathy (HCM) and Other Serious Diseases of Cardiac Diastolic Dysfunction
BOULDER, Colo.–(BUSINESS WIRE)–Edgewise Therapeutics, Inc., (Nasdaq: EWTX), a leading muscle disease biopharmaceutical company, today announced initial dosing in a Phase 1 trial of EDG-7500. EDG-7500 is a first-in-class oral, selective, cardiac sarcomere modulator, specifically designed to slow early contraction velocity and address impaired cardiac relaxation associated with HCM and other […]
Attune Medical’s ensoETM™ Granted FDA De Novo Marketing Authorization to Reduce the Likelihood of Ablation-related Esophageal Injury Resulting from Radiofrequency Cardiac Ablation Procedures
CHICAGO–(BUSINESS WIRE)–Attune Medical has been granted De Novo marketing authorization from the US Food and Drug Administration (FDA) for its ensoETM device to reduce the likelihood of ablation-related esophageal injury resulting from radiofrequency cardiac ablation procedures. “Our lab has published studies showing a 30 percent reduction in procedure time and a 14 percent […]
MedAlliance Announces Enrollment of over 1,660 Patients in Landmark SELUTION DeNovo Study
GENEVA, Sept. 14, 2023 /PRNewswire/ — MedAlliance has announced enrollment of over 1,660 patients in its ground-breaking SELUTION DeNovo coronary randomized study. Recruitment is now half way towards a planned 3,326 patients. SELUTION DeNovo compares the treatment strategy using a novel sirolimus drug-eluting balloon (SELUTION SLR), versus any limus drug-eluting stent (DES). […]
CorVista Health Announces FDA Clearance of the Novel CorVista System to Detect the Presence of Cardiac Disease
WASHINGTON–(BUSINESS WIRE)–CorVista Health, Inc., a leading digital health company dedicated to improving cardiovascular disease diagnosis, is pleased to announce the FDA clearance of the CorVista System® with CAD Add-On to evaluate the presence of significant coronary artery disease as an aid in diagnosis. “CorVista Health’s innovative solution has the potential […]
New Study Confirms HeartFlow FFRCT Leads to Improved Patient Outcomes
CLEVELAND – A diagnostic test, first offered in the United States at University Hospitals (UH) Harrington Heart & Vascular Institute, has now shown through a clinical study to significantly decrease cardiovascular mortality, reduce additional non-invasive heart testing, and increase cath lab efficiency. These results show a promising, new approach that […]