– Beckman Coulter to take over distribution to customers of a B-type Natriuretic Peptide (BNP) assay, a marker of congestive heart failure on the company’s immunoassay platforms – This BNP assay for Beckman Coulter immunoassay analyzers, currently sold as TRIAGE® BNP by Quidel, will now be sold by Beckman Coulter […]
Coronary/Structural Heart
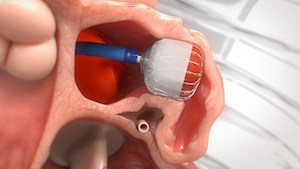
CARMAT Announces the First Human Implant of its Aeson® Artificial Heart in Germany
The implant was performed at Hannover Medical School PARIS–(BUSINESS WIRE)–Regulatory News: CARMAT (FR0010907956, ALCAR) (Paris:ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to provide a therapeutic alternative for people suffering from end-stage biventricular heart failure, today announces the first implant of its Aeson® bioprosthetic […]
Study Data Presented at TVT on the Economics of Minimalist TAVR, PARTNER 3 Bicuspid TAVR
IRVINE, Calif., July 22, 2021 /PRNewswire/ — Edwards Lifesciences (NYSE: EW) today announced that clinical and economic results from the 3M (multidisciplinary, multimodality, but minimalist) Transcatheter Aortic Valve Replacement (TAVR) Economic Study and the PARTNER 3 Bicuspid Registry for SAPIEN 3 TAVR Study were presented at TVT 2021: The Structural Heart Summit. The 3M-TAVR study demonstrated positive […]
PaceMate™ and UNC School of Medicine Advance Cardiac Research with Data Partnership
BRADENTON, Fla.–(BUSINESS WIRE)–PaceMate™, the leading digital healthcare company in compliant cardiac data management, is pleased to announce a new strategic data research partnership with electrophysiologists, cardiologists, and the department research team at the University of North Carolina School of Medicine. The unmatched breadth of cardiac device data collected via PaceMate™’s […]
Late-Breaking Trial Data at TVT Demonstrate Sustained Safety and Performance of WATCHMAN FLX™ Left Atrial Appendage Closure Device
PINNACLE FLX study meets 24-month endpoint, reinforces positive 12-month outcomes with next-generation device for patients with non-valvular atrial fibrillation MARLBOROUGH, Mass., July 21, 2021 /PRNewswire/ — Today, Boston Scientific (NYSE: BSX) announced positive 24-month results from the PINNACLE FLX clinical trial assessing the safety and efficacy of the next-generation WATCHMAN FLX™ Left Atrial […]
BIOMODEX Technology Enables New Milestone in Interventional Cardiology
Physicians at Montreal Heart Institute used LAACS™ solution to simulate left atrial appendage occlusion, validating the company’s efforts to reduce surgical errors and improve outcomes BOSTON, July 21, 2021 /PRNewswire/ — BIOMODEX®, the leader in bio-realistic haptic simulators for patient-specific rehearsal and physician training, today announced that its LAACS™ (Left Atrial Appendage Closure System) solution […]
LEXEO Therapeutics Expands Cardiac Gene Therapy Pipeline with Acquisition of Stelios Therapeutics and its Gene Therapy Programs for Rare Cardiovascular Diseases
LEXEO obtains exclusive rights to three investigational AAV-mediated gene therapy programs for rare cardiac disorders, all of which have no existing disease-modifying treatments available Strategic acquisition expands LEXEO’s rare cardiac gene therapy pipeline making it one of the most extensive in the field NEW YORK, July 21, 2021 (GLOBE NEWSWIRE) […]
VERQUVO® (vericiguat) Approved in the European Union
European Approval Granted to Bayer Marks Another Important Milestone for VERQUVO KENILWORTH, N.J.–(BUSINESS WIRE)–Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced that the European Commission (EC) has granted marketing authorization in the European Union (EU) for soluble guanylate cyclase (sGC) stimulator VERQUVO® (vericiguat). In […]
NIMIUM Therapeutics and Paraza Pharma form partnership to develop new medicines to treat cardiometabolic diseases (CMDs), obesity and type 2 diabetes
Innovative partnership focuses on early-stage development of activators for a glucose detoxification enzyme called glycerol-3-phosphate phosphatase (G3PP) MONTREAL, July 20, 2021 /PRNewswire/ — NIMIUM Therapeutics (www.nimium.ca), a Montreal-based biotech, has formed a partnership with Paraza Pharma (www.parazapharma.com) to develop a new treatment for cardiometabolic diseases (CMDs). The treatment aims to prevent and reverse the adverse […]
CARMAT announces the first commercial implant of its Aeson® artificial heart
The implant, performed at the Azienda Ospedaliera dei Colli hospital in Naples, Italy, represents the first sale for CARMATand paves the way for the Company’s commercial development PARIS–(BUSINESS WIRE)–Regulatory News: CARMAT (Paris:ALCAR) (FR0010907956, ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to provide a […]