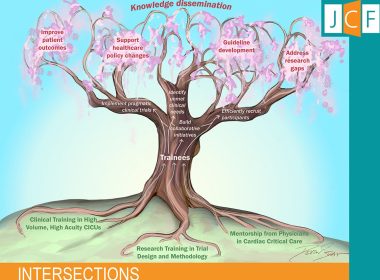
The first issue of the Journal of Cardiac Failure-Intersections, an open-access journal focused on heart failure and its many intersections, is now available. WASHINGTON, Sept. 17, 2025 /PRNewswire/ — Scientific breakthroughs often begin with a single Aha! Moment—an idea that evolves…
Coronary/Structural Heart
Tenax Therapeutics Announces European Patent Office Intention to Grant Patent Covering Use of Levosimendan in PH-HFpEF
Provides Patent Protection in Europe Through at Least December 2040 Further IP Protection for TNX-103 (Oral Levosimendan) and Other Drug Formulations, and its Active Metabolites, in PH-HFpEF Also Provides Patent Protection for Wide Range of Levosimendan Doses, and its Use in Combination with Various Cardiovascular Drugs in PH-HFpEF CHAPEL HILL, N.C., Sept. 16, 2025 (GLOBE NEWSWIRE) — Tenax Therapeutics, Inc. (Nasdaq: TENX) (“Tenax” or “Tenax Therapeutics” or the “Company”), a Phase 3, development-stage pharmaceutical company using clinical insights to develop novel cardiopulmonary therapies, today announced that the European Patent Office (EPO) has notified Tenax of its Intention to Grant a patent that will provide intellectual property (IP) protection for TNX-103 (oral levosimendan), and other formulations of levosimendan, as well as its active metabolites, for use in pulmonary hypertension resulting from heart failure with preserved ejection fraction (PH-HFpEF). Once granted, this patent will provide Tenax with protection in Europe through 2040, and may qualify for an additional European patent term beyond 2040. “This patent will protect our use of levosimendan in PH-HFpEF, including TNX-103, in Europe, where prevalence estimates indicate the number of patients currently suffering from this disease approximates the number estimated in North America. This patent protects an important commercial opportunity for Tenax. It builds on multiple patents already issued in the U.S. and Canada, as we continue to strengthen the global IP portfolio of our novel and potentially first-in-disease drug,” said Chris Giordano, President and Chief Executive Officer of Tenax Therapeutics. “The European patent is particularly timely for the Company as we prepare to initiate our second registrational Phase 3 study, LEVEL-2, which will recruit patients across Europe. We expect to initiate LEVEL-2 this year, an essential step not only in advancing TNX-103 toward regulatory approval in PH-HFpEF, but also to including patients from around the world at the Phase 3 stage of development.” The EPO issued an Intention to Grant notification for Tenax’s patent application titled “LEVOSIMENDAN FOR TREATING PULMONARY HYPERTENSION WITH HEART FAILURE WITH PRESERVED EJECTION FRACTION (PH-HFpEF).” Once granted, this patent will include claims covering the use of levosimendan when delivered through various routes of administration to treat PH-HFpEF, including oral, intravenous, inhaled, transdermal, and subcutaneous use. In addition, this patent will include similar protection for the use of the active metabolites of levosimendan (OR1896 and OR1855) for the treatment of PH-HFpEF. This patent also will expressly provide protection in Europe for a wide range of doses of levosimendan, as well as for use of levosimendan in combination with various cardiovascular drugs, in PH-HFpEF. This new patent, once granted, will have a patent term until December 2040, and it may qualify for European patent supplementary protection certificates (SPC) that would extend the period of patent protection beyond 2040. About Levosimendan (TNX-101, TNX-102, TNX-103) Levosimendan is a novel, first-in-class K-ATP channel activator/calcium sensitizer currently being evaluated to treat pulmonary hypertension (PH) associated with heart failure with preserved ejection fraction (PH-HFpEF). Levosimendan was first developed for intravenous use in hospitalized patients with acutely decompensated heart failure, and it has received market authorization in 60 countries in this indication, although it is not available in the United States or Canada. Tenax’s Phase 2 HELP study, including its open-label extension stage, demonstrated the potential of IV (TNX-101) and oral (TNX-103) levosimendan to bring durable improvements in exercise capacity and quality of life, as well as other clinical assessments, in patients with PH-HFpEF. TNX-103 (oral levosimendan) is currently being evaluated in LEVEL, a Phase 3, double-blind, randomized, placebo-controlled clinical trial in patients with PH-HFpEF. About Tenax Therapeutics Tenax Therapeutics, Inc. is a Phase 3, development-stage pharmaceutical company using clinical insights to develop novel cardiopulmonary therapies. The Company owns global rights to develop and commercialize levosimendan, which it is developing for the treatment of PH-HFpEF, the most prevalent form of pulmonary hypertension globally, for which no product has been approved to date. For more information, visit www.tenaxthera.com. Tenax Therapeutics’ common stock is listed on The Nasdaq Stock Market LLC under the symbol “TENX”. Caution Regarding Forward-Looking Statements Except for historical information, all of the statements, expectations and assumptions contained in this press release are forward-looking statements. These forward-looking statements may include information concerning expected intellectual property developments and possible or projected future business operations. Actual results might differ materially from those explicit or implicit in the forward-looking statements. Important factors that could cause actual results to differ materially include: intellectual property risks; risks of our clinical trials, including, but not limited to, the timing, delays, costs, design, location, initiation, enrollment, and results of such trials; any delays in regulatory review and approval of product candidates in development; risks related to our business strategy, including the prioritization and development of product candidates; our estimates regarding the potential market opportunity for our product candidates; reliance on third parties, including Orion Corporation, our manufacturers and CROs; risks regarding the formulation, production, marketing, customer acceptance and clinical utility of our product candidates; the potential advantages of our product candidates; our competitive position; our ability to maintain our culture and recruit, integrate and retain qualified personnel and advisors, including on our Board of Directors; risks associated with our cash needs; volatility and uncertainty in the global economy and financial markets in light of unexpected changes in tariffs and the possibility of pandemics, global financial and geopolitical uncertainties, including in the Middle East and the Russian invasion of and war against the country of Ukraine; changes in legal, regulatory and legislative environments in the markets in which we operate and the impact of these changes on our ability to obtain regulatory approval for our products; and other risks and uncertainties set forth from time to time in our SEC filings. Tenax Therapeutics assumes no obligation and does not intend to update these forward-looking statements except as required by law. Contact: Investor and Media: Argot Partnerstenax@argotpartners.com
Caristo Congratulates Abcentra for First Patient Dosed Successfully in Phase 2b ‘FORTIFY’ Clinical Trial Evaluating Orticumab in Patients with Cardiovascular Disease
‘FORTIFY’ Selects FAI-Score™ as Primary Outcome Measure, and Caristo as Worldwide Imaging Core Lab for Coronary Inflammation and Plaque Measurement and Monitoring STAMFORD, Conn. and LOS ANGELES, Sept. 16, 2025 /PRNewswire/ — Caristo Diagnostics, on a mission to transform the diagnosis…
Conavi Medical Submits Next Generation Novasight Hybrid Intravascular Imaging System to US FDA for 510(k) Clearance
TORONTO, Sept. 16, 2025 (GLOBE NEWSWIRE) — Conavi Medical Corp. (TSXV: CNVI) (OTCQB: CNVIF) (“Conavi” or the “Company”), a commercial-stage medical device company focused on designing, manufacturing, and marketing imaging technologies to guide minimally invasive cardiovascular procedures, announced that it has submitted its next generation Novasight Hybrid™ IVUS/OCT intravascular imaging system to the U.S. Food and Drug Administration (“FDA”) for 510(k) clearance for coronary applications.
EU approval makes Novo Nordisk’s oral semaglutide the first and only oral GLP-1 RA to reduce cardiovascular death, heart attack and stroke
Oral semaglutide (Rybelsus®) is now the first and only oral GLP-1 RA approved for type 2 diabetes, with proven cardiovascular benefits1This approval is based on results from the SOUL clinical trial, where oral semaglutide (Rybelsus®) reduced cardiovascular death, heart attack and stroke by 14% versus placebo, when added to standard of care, in adults with type 2 diabetes at high cardiovascular risk1In addition, new results from SOUL will be presented at one of the largest diabetes conferences (EASD) later this week, showing that oral semaglutide significantly reduced hospitalisations compared with placebo2 Bagsværd, Denmark, 15 September 2025 – Novo Nordisk today announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has approved an update to the Rybelsus® (oral semaglutide) label to reflect the cardiovascular benefits seen in the SOUL trial. SOUL was a phase 3b trial carried out to evaluate the effect of Rybelsus® on cardiovascular outcomes in people with type 2 diabetes and atherosclerotic cardiovascular disease (ASCVD) and/or chronic kidney disease (CKD)1. Rybelsus® is now the first and only oral glucagon-like peptide 1 receptor agonist (GLP-1 RA) – mimicking a natural hormone in your body that helps regulate blood sugar, appetite, and digestion – available in the EU for type 2 diabetes with a proven cardiovascular benefit1. “Heart problems are the leading cause of disability and death for people living with type 2 diabetes. Therefore, treatments that also address heart problems are key to improving not only health outcomes, but also quality of life – and this approval will help do just that,” said Emil Kongshøj Larsen, executive vice president, International Operations at Novo Nordisk. “This milestone makes semaglutide the only oral GLP-1 RA with proven blood glucose and body weight reduction, as well as cardiovascular benefits.” New results from the SOUL trial will be shared later this week at the European Association for the Study of Diabetes (EASD) 2025 Annual Meeting, 15–19 September. These include findings that treatment with oral semaglutide significantly reduced hospitalisations related to serious adverse events compared with placebo2. Additional SOUL results will be presented at the same meeting, which highlight that the cardiovascular benefits of oral semaglutide were consistent regardless of body mass index (BMI) and body weight of participants3. In the US, a decision is expected later this year for a label extension for the cardiovascular indication for Rybelsus®. Novo Nordisk has also submitted an application in the US for a once-daily 25 mg oral formulation of semaglutide (Wegovy® in a pill) in adults living with obesity or overweight and cardiovascular disease. A decision is expected at the turn of this year, and if approved, Wegovy® would become the first oral GLP-1 RA indicated for chronic weight management. Rybelsus® is the first and only oral GLP-1 RA approved for the treatment of type 2 diabetes, following its launch in 2019. It is supported by a strong clinical and real-world evidence base, demonstrating superior blood glucose reduction and body weight reduction versus multiple comparators, as well as an established safety profile in people with type 2 diabetes4-8. About SOUL SOUL was a multicentre, international, randomised, double-blind, parallel-group, placebo-controlled, phase 3 cardiovascular outcomes trial, with 9,650 participants enrolled. It was conducted to assess the effect of oral semaglutide versus placebo, when added to standard of care, on cardiovascular outcomes in people with type 2 diabetes and established cardiovascular disease and/or chronic kidney disease (CKD). The SOUL trial was initiated in 2019. The primary outcome was time-to-first occurrence of major adverse cardiovascular events (MACE; a composite objective consisting of cardiovascular death, heart attack and stroke) 1. The SOUL trial demonstrated a superior reduction in MACE of 14% for people treated with oral semaglutide compared to placebo in people with type 2 diabetes and cardiovascular disease and/or CKD, making Rybelsus® (oral semaglutide) the first and only oral GLP-1 RA with a proven cardiovascular benefit1. About Rybelsus® Rybelsus® (oral semaglutide) is a glucagon-like peptide 1 receptor agonist (GLP-1 RA) indicated for the treatment of adults with insufficiently controlled type 2 diabetes mellitus to improve glycaemic control as an adjunct to diet and exercise7, 9. Rybelsus® is administered once daily and is approved for use in the following therapeutic dosages: 1.5 mg, 3 mg, 4 mg, 7 mg, 9 mg, 14 mg, 25 mg and 50 mg4, 5. Rybelsus® offers superior blood glucose lowering versus multiple comparators4, 5, together with consistent weight reduction4, 5, 10, reduction in cardiometabolic risk factors10 and reduction in major adverse cardiovascular events (MACE) 1. Rybelsus® is now the first and only oral GLP-1 RA available in the EU for type 2 diabetes with a proven cardiovascular benefit1. It is currently available in 48 countries11, and more than 2.4 million people with type 2 diabetes are currently being treated with Rybelsus® worldwide12. About Wegovy®Semaglutide 2.4 mg is marketed under the brand name Wegovy®. In the EU, Wegovy® is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management in adults with a BMI of 30 kg/m2 or greater (obesity) or adults with a BMI of 27 kg/m2 or greater (overweight) in the presence of at least one weight-related comorbid condition. In the EU, Wegovy® is also indicated for paediatric patients aged 12 years and older with an initial BMI at the 95th percentile or greater for age and gender (obesity) and body weight above 60 kg. The clinical section of the label also includes data on Wegovy® major adverse cardiovascular events (MACE) risk reduction, improvements in heart failure with preserved ejection fraction (HFpEF)-related symptoms and physical function, as well as pain reduction related to knee osteoarthritis13. In the US, Wegovy® is indicated in combination with a reduced calorie diet and increased physical activity to reduce the risk of MACE in adults with established cardiovascular disease and either obesity or overweight, to reduce excess body weight and maintain weight reduction long term in paediatric patients aged 12 years and older with obesity and in adults with obesity or with overweight in the presence of at least one weight-related comorbid condition, as well as for the treatment of noncirrhotic metabolic dysfunction-associated steatohepatitis (MASH) in adults with moderate to advanced liver scarring (fibrosis), but not with cirrhosis of the liver, in combination with a reduced calorie diet and increased physical activity14. Novo Nordisk is a leading global healthcare company founded in 1923 and headquartered in Denmark. Our purpose is to drive change to defeat serious chronic diseases built upon our heritage in diabetes. We do so by pioneering scientific breakthroughs, expanding access to our medicines, and working to prevent and ultimately cure disease. Novo Nordisk employs about 78,400 people in 80 countries and markets its products in around 170 countries. For more information, visit novonordisk.com, Facebook, Instagram, X, LinkedIn and YouTube. Contacts for further information Media: Ambre James-Brown +45 3079 9289globalmedia@novonordisk.comLiz Skrbkova (US)+1 609 917 0632lzsk@novonordisk.comInvestors: Jacob Martin Wiborg Rode+45 3075 5956jrde@novonordisk.comSina Meyer +45 3079 6656azey@novonordisk.comMax Ung+45 3077 6414 mxun@novonordisk.com Christoffer Sho Togo Tullin+45 3079 1471cftu@novonordisk.comAlex Bruce +45 34 44 26 13axeu@novonordisk.comFrederik Taylor Pitter +1 609 613 0568fptr@novonordisk.com _______________________References1. McGuire DK, et al. N Engl J Med. 2025;392(20):2001-2012.2. Buse JB, et al. Oral presentation presented at the European Association for the Study of Diabetes (EASD) 2025; 15-19 Sep 2025; Vienna, Austria.3. Inzucchi SE, et al. Oral presentation presented at the European Association for the Study of Diabetes (EASD) 2025; 15–19 Sep 2025; Vienna, Austria. 4. Rosenstock J, et al. JAMA. 2019;321(15):1466-1480.5. Rodbard HW, et al. Diabetes Care. 2019;42(12):2272-2281.6. Pratley R, et al. Lancet. 2019;394(10192):39-50.7. Rybelsus® (oral semaglutide): US Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213051s023lbl.pdf. Last accessed: September 2025.8. Aroda VR, et al. Lancet. 2023;402(10403):693-704.9. Rybelsus® (oral semaglutide): Summary of Product Characteristics. Available at: https://www.ema.europa.eu/en/medicines/human/EPAR/rybelsus. Last accessed: September 2025.10. Husain M, et al. N Engl J Med. 2019;381(9):841-851.11. Novo Nordisk Data on File. LEA portal Product Planning, 25th Aug 2025.12. Novo Nordisk Data on File. IQVIA Jun’25 Patients R3M Vol. data. 2025.13. Wegovy® (semaglutide): Summary of Product Characteristics. Available at: https://www.ema.europa.eu/en/documents/product-information/wegovy-epar-product-information_en.pdf. Last accessed: September 2025.14. Wegovy® (semaglutide): US Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215256s015lbl.pdf. Last accessed: September 2025.
Attachment
PR250915-Rybelsus-label-update-SOUL
Cadrenal Therapeutics Enhances Anticoagulation Pipeline Through Acquisition of eXIthera’s Portfolio of Factor XIa Inhibitors
Acquisition significantly enhances the Company’s pipeline by adding novel assets in acute and chronic anticoagulation settingsCompany is strategically poised to deliver differentiated therapeutics across the spectrum of cardiovascular thrombotic risk PONTE VEDRA, Fla., Sept. 15, 2025 (GLOBE NEWSWIRE) — Cadrenal Therapeutics, Inc. (Nasdaq: CVKD), a biopharmaceutical company developing transformative therapeutics to overcome the gaps in anticoagulation therapy, today announced the acquisition of the assets of eXIthera Pharmaceuticals (“eXIthera”), including its proprietary portfolio of investigational intravenous (IV) and oral Factor XIa inhibitors. The acquisition significantly enhances Cadrenal’s pipeline, adding drug candidates that address large and underserved segments of the current $38 billion global anticoagulation market. eXIthera’s lead asset, frunexian, is a first-in-class, Phase 2-ready intravenous (IV) Factor XIa inhibitor designed for acute care settings where contact activation of coagulation by medical devices plays a significant role, such as cardiopulmonary bypass, catheter thrombosis, and other blood-contacting implanted cardiac devices. The acquisition also includes EP-7327, an oral Factor XIa inhibitor, for the prevention and treatment of major thrombotic conditions. “With this acquisition, Cadrenal is the only company in the world developing a novel vitamin K antagonist (tecarfarin) and Factor XIa inhibitors, a promising new class of anticoagulants,” said Quang X. Pham, Chairman and CEO of Cadrenal Therapeutics. “These newly acquired assets will expand Cadrenal’s capabilities in an effort to address even more critical gaps in current antithrombotic treatment, especially for patients for whom current therapies are unreliable or carry excessive bleeding risk.” Unlike current anticoagulants on the market, which increase the risk of bleeding by broadly impairing coagulation, eXIthera’s compounds are mechanism-based inhibitors of Factor XIa, offering high potency, selectivity, and tunable pharmacokinetics. Factor XIa inhibition is one of the most active and exciting areas of current thrombosis research. “This acquisition reinforces Cadrenal’s long-term vision of becoming a category leader in anticoagulation,” added Pham. “With tecarfarin planning a trial in patients with end-stage kidney disease transitioning to dialysis, our plans for LVAD patients, and the current addition of frunexian and EP-7327, we believe that Cadrenal is strategically positioned to deliver differentiated therapeutics across the entire spectrum of patients with cardiovascular thrombotic risk.” Assets Acquired: Frunexian: Phase 2-ready IV Factor XIa agent for acute anticoagulationEP-7327: IND-ready oral small molecule candidate for chronic indicationsExtensive portfolio of additional novel FXIa small molecules Under a pre-existing license agreement, Sichuan Haisco Pharmaceuticals retains rights to frunexian in China, having completed a successful Phase 1 trial there. Under the terms of the license, Cadrenal will be entitled to receive royalties on future sales of frunexian in China. Deal Terms Overview: Under the terms of the acquisition agreement, eXIthera will receive milestone payments from Cadrenal totaling up to $15 million, contingent upon the realization of certain future clinical and regulatory milestones. Additionally, eXIthera will be entitled to royalties on global sales of the acquired assets upon future commercialization. The structure and terms of the agreement enable Cadrenal to focus capital deployment on advancing the clinical development of tecarfarin and the acquired assets. About Cadrenal Therapeutics, Inc.Cadrenal Therapeutics, Inc. is a biopharmaceutical company developing transformative therapeutics to overcome the gaps in anticoagulation therapy. Cadrenal’s lead investigational product is tecarfarin, a novel oral Vitamin K antagonist anticoagulant that is designed to address unmet needs in anticoagulation therapy. Tecarfarin is a reversible anticoagulant (blood thinner) designed to prevent heart attacks, strokes, and deaths due to blood clots in patients requiring chronic anticoagulation. Although warfarin is widely used, extensive clinical and real-world data have shown it can have significant, serious side effects. With tecarfarin, Cadrenal aims to reduce the clinical complexities of managing Vitamin K antagonists, particularly where direct-acting oral anticoagulants (DOACs) remain inadequate or unproven. Tecarfarin received Orphan Drug Designation (ODD) and fast-track designation for the prevention of systemic thromboembolism (blood clots) of cardiac origin in patients with end-stage kidney disease and atrial fibrillation (ESKD+AFib). The Company also received ODD for the prevention of thromboembolism and thrombosis in patients with implanted mechanical circulatory support devices, including Left Ventricular Assist Devices (LVADs). For more information, visit https://www.cadrenal.com/ and connect with the Company on LinkedIn. Safe Harbor Any statements in this press release about future expectations, plans, and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements.” The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potentially,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These statements include statements regarding Cadrenal’s ability to deliver differentiated therapeutics across the entire spectrum of cardiovascular thrombotic risk and overcome the gaps in anticoagulation therapy; the acquisition significantly enhancing Cadrenal’s pipeline and addressing large and underserved segments of the global anticoagulation market; the size of the global anticoagulation market; the potential of EP-7327 for the prevention and treatment of major thrombotic conditions; Cadrenal’s ability to address even more critical gaps in current antithrombotic treatment with the acquisition; Cadrenal becoming a leader in anticoagulation; commencement of a trial in patients with end-stage kidney disease transitioning to dialysis; Cadrenal’s receipt of royalties on future sales of frunexian in China; the payment to eXIthera of milestone payments by the Company totaling up to $15 million contingent upon the realization of certain future clinical and regulatory milestones as well as global sales of the acquired assets upon future commercialization; Cadrenal’s ability to focus capital deployment on advancing the clinical development of tecarfarin and the acquired assets; and tecarfarin addressing the unmet needs in anticoagulation therapy; tecarfarin reducing the clinical complexities of managing Vitamin K antagonists. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including the Cadrenal’s ability to deliver differentiated therapeutics across the entire spectrum of cardiovascular thrombotic risk and overcome the gaps in anticoagulation therapy; the potential of EP-7327 for the prevention and treatment of major thrombotic conditions; Cadrenal’s ability to address even more critical gaps in current antithrombotic treatment; the payment of milestone payments and royalties; Cadrenal successfully advancing tecarfarin and the acquired assets into clinical practice; the commencement of a trial in patients with end-stage kidney disease transitioning to dialysis; tecarfarin addressing the unmet needs in anticoagulation therapy; tecarfarin reducing the clinical complexities of managing Vitamin K antagonists and the other risk factors described in the Company’s Annual Report on Form 10-K for the year ended December 31, 2024, and the Company’s subsequent filings with the Securities and Exchange Commission, including subsequent periodic reports on Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. Any forward-looking statements contained in this press release speak only as of the date hereof and, except as required by federal securities laws, the Company specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events, or otherwise. For more information, please contact: Cadrenal Therapeutics:Matthew Szot, CFOpress@cadrenal.com Investors:Lytham Partners, LLCRobert Blum, Managing Partner602-889-9700CVKD@lythampartners.com
Medtronic initiates global pivotal study of cardiac pacing in a new patient population
Study to evaluate whether a new approach to pacing the heart can improve the lives of patients with heart failure with preserved ejection fraction (HFpEF) who have limited treatment options today GALWAY, Ireland, Sept. 15, 2025 /PRNewswire/ — Medtronic plc (NYSE: MDT), a global leader in…
Pulnovo Medical Receives two IDE approvals for PADN system’s clinical trials with CMS coverage
SHANGHAI, Sept. 12, 2025 /PRNewswire/ — Pulnovo Medical, a globally recognized leader in medical devices for pulmonary hypertension (PH) and heart failure (HF), is proud to announce that its PADN catheter and generator has received two Investigational Device Exemption (IDE) approvals—HDE…
AI-Enabled Eko Health Stethoscope TRICORDER Trial Marks Historic Leap in Early Heart Disease Detection
Landmark TRICORDER study finds major gains in detection of heart failure, atrial fibrillation, and valvular disease with Eko Health’s AI-enabled stethoscopes in routine exams. SAN FRANCISCO, Sept. 11, 2025 /PRNewswire/ — Eko Health, a leader in AI-powered cardiac and pulmonary disease…
European Society of Cardiology (ESC) Congress 2025 Late Breaking Results Confirm Caristo FAI-Score™ Technology’s Superior and Complementary Predictive Value for Cardiac Death Beyond hsCRP
FAI-Score has stronger predictive value than hsCRP for 10-year cardiac death, even among those with zero calcium score and no visible coronary plaque In patients with low hsCRP, high FAI-Score predicts 7-fold higher risk of cardiac death OXFORD, England, Sept. 11, 2025 /PRNewswire/ –…