CALGARY, Alberta, June 22, 2020 (GLOBE NEWSWIRE) — Resverlogix Corp. (“Resverlogix”) (TSX:RVX) is pleased to announce that the U.S. Food & Drug Administration (FDA) has accepted its BETonMACE2 clinical plan as a registration enabling study with positive implications for Resverlogix and its ongoing partnership discussions. Key written development points from […]
Coronary/Structural Heart
MedAlliance Gains CE Mark Approval for Coronary SELUTION SLR™ Sirolimus Drug Eluting Balloon
MUMBAI, India, June 16, 2020 /PRNewswire/ — MedAlliance has announced the award of its second CE Mark: SELUTION SLR™ 014 PTCA, a novel Sirolimus Drug Eluting Balloon (DEB), for the treatment of coronary arterial disease. This includes indications for both de-novo lesions as well as in-stent restenosis. The approval applies to a broad […]
Abiomed Launches Virtual Physician Education Program, CAMP PCI, to Improve High-Risk PCI Patient Outcomes
DANVERS, Mass.–(BUSINESS WIRE)–Today, Abiomed (NASDAQ: ABMD) launches a cutting-edge digital education platform called CAMP PCI, which stands for Coronary Artery & Myocardial Protected Percutaneous Coronary Intervention, to improve patient outcomes and quality of life with supported high-risk PCI by utilizing best practices, techniques and technologies to enable safer, more effective and complete revascularization. […]
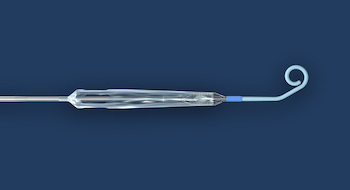
Puzzle Medical Devices Inc. announces the closing of a first financing round; provides update on its minimally invasive transcatheter heart pump
MONTREAL, June 15, 2020 /PRNewswire/ – Puzzle Medical Devices Inc., (www.puzzlemed.com) announces the closure of its first convertible debenture financing round led by Canadian entrepreneur Pascal Labrecque. Other participants include a United States venture capital firm, entrepreneurs and interventional cardiologists practicing in Canada, the United States and Europe. This investment follows the successful completion of Puzzle Medical’s first preclinical studies […]
Heartbeat Health and The American College of Cardiology Join to Revolutionize Cardiology With Breakthrough Virtual Care
Using wearables and devices, physicians can incorporate clinical data into telehealth visits to offer complete remote health management. NEW YORK, June 15, 2020 /PRNewswire/ — The American College of Cardiology (ACC) and Heartbeat Health, the first telemedicine and virtual care platform built by cardiologists for cardiologists, are joining forces to transform telehealth into […]
FDA Approves Abiomed’s First-in-Human Trial of Impella ECP, World’s Smallest Heart Pump
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed (NASDAQ: ABMD) announces the United States Food and Drug Administration (FDA) has approved the company’s investigational device exemption application to start an early feasibility study with a first-in-human trial of the 9 French (Fr) Impella ECP™ heart pump. Impella ECP, which stands for expandable cardiac power, will be […]
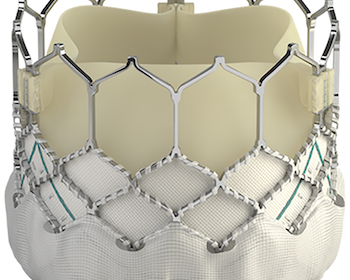
Edwards SAPIEN 3 Transcatheter Heart Valve Receives Approval In China
IRVINE, Calif., June 8, 2020 /PRNewswire/ — Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced that it has received Chinese regulatory approval for the Edwards SAPIEN 3 transcatheter heart valve for the treatment of patients suffering from severe, symptomatic aortic […]
Medtronic Resolute Onyx™ DES Receives First and Only One-Month DAPT Indication for High Bleeding Risk Patients in Europe
DUBLIN, June 05, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received CE (Conformité Européenne) Mark for a one-month dual antiplatelet therapy (DAPT) indication for high bleeding risk (HBR) patients implanted with the Resolute Onyx™ Drug-Eluting Stent (DES). For HBR patients, whose bleeding risk may be increased by taking longer DAPT […]
Mynomx Announces Scientific Validation of Its Cardiometabolic Risk Engines for Assessing Risk of COVID-19 in Populations
PALO ALTO–(BUSINESS WIRE)–Mynomx Inc. (formerly Precision Wellness, Inc.), a Silicon Valley-based company at the intersection of next-generation AI analytics and the latest medical and nutrition science, today announced the scientific validation of their cardio and metabolic predictive models against 2.5M patient population through two sponsored research studies with Stanford University. […]
MedHub AutocathFFR Awarded AMAR Certification, Israel’s Medical Device Regulatory Approval
AutocathFFR, the company’s first commercial product, is a transformative cardiac diagnosis system. TEL AVIV, Israel, June 4, 2020 /PRNewswire/ — MedHub Ltd., a medical technology company focused on developing and commercializing AI powered medical solutions that aid in the diagnosis of cardiac disease, announced that is has gained AMAR approval for its […]