– Inclisiran, an investigational medicine, showed durable and potent reduction of low-density lipoprotein cholesterol (LDL-C) in patients with atherosclerotic cardiovascular disease (ASCVD), ASCVD risk equivalents and heterozygous familial hypercholesterolemia (HeFH)[1,2] – Inclisiran reduced LDL-C at 17 months by 52% in patients with ASCVD (ORION-10), 50% for ASCVD and ASCVD risk […]
Coronary/Structural Heart
Tenax Therapeutics Completes Enrollment in Phase 2 Pulmonary Hypertension Trial
HELP study exceeds target enrollment with 37 patients 84% initial open-label lead-in response rate to qualify for randomization Pulmonary capillary wedge pressure (PCWP) was reduced, on average, by 7.5 mmHg during exercise following the initial levosimendan infusion No serious drug-related adverse events have been reported to date Top-line data expected […]
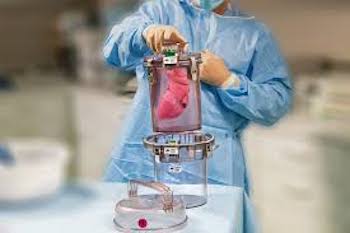
Paragonix Technologies, Inc., Announces AdventHealth’s Adoption of Paragonix SherpaPak™ Cardiac Transport System
Paragonix SherpaPak™ Cardiac Transport System is the only FDA cleared and CE marked device that safeguards hearts between donor and recipient patients CAMBRIDGE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. today announced AdventHealth (Orlando, Florida) has adopted its Paragonix SherpaPak™ Cardiac Transport System (CTS) for the preservation of donor hearts for transplantation. A recent […]
Mentice Expands Coronary Product Portfolio with the Introduction of Coronary Intermediate Software
STOCKHOLM, March 12, 2020 /PRNewswire/ — Mentice AB (STO: MNTC), a world leader in simulation solutions for endovascular therapies, introduces Coronary Intermediate as the latest software addition to the Coronary Product Portfolio. Coronary Intermediate follows the introduction of Coronary Essentials three months earlier at the 20th International Meeting on Simulation in Healthcare […]
VisCardia Announces Completion of Its VisONE Heart Failure Pilot Study
PORTLAND, Ore., March 9, 2020 /PRNewswire/ — VisCardia Inc., a privately held medical device developer, announced today it has completed the company’s First-in-Human study of the implantable VisONE® system for medically refractory heart failure (HF) patients with reduced ejection fraction (HFrEF) and preserved ventricular synchrony. The VisONE Heart Failure pilot was a prospective, […]
SynCardia Systems, LLC. 50cc temporary Total Artificial Heart (TAH-t) Now FDA Approved
TUCSON, AZ, March 09, 2020 (GLOBE NEWSWIRE) — With the FDA approval of the SynCardia Systems, LLC 50cc TAH-t system a greater number of cardiac transplant eligible patients at risk of imminent death from biventricular failure now have access to total artificial heart therapy. The 50cc TAH-t is 37.5% smaller […]
Acasti Pharma Awarded Notice of Allowance for Additional Composition of Matter and Method of Use Patents in the United States and Mexico
Patent upheld in Japan following opposition filings and subsequently allowed Completes TRILOGY 1 audits and accelerates plans to submit FDA meeting request by the end of March LAVAL, Québec, March 11, 2020 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (“Acasti” or the “Company”) (NASDAQ: ACST – TSX-V: ACST), a biopharmaceutical innovator […]
Correvio Partners with Hong Kong Teson Pharma Limited for the Commercialization of Aggrastat® in China
VANCOUVER and HONG KONG, March 10, 2020 /PRNewswire/ – Correvio Pharma Corp. (NASDAQ: CORV) (TSX: CORV), a specialty pharmaceutical company focused on commercializing hospital drugs, and Hong Kong Teson Pharma Limited (Teson), a privately-held pharmaceutical company, today announced that Correvio International Sàrl, a wholly owned subsidiary of Correvio, and Teson have entered into an exclusive agreement […]
FDA Inks Research Collaboration Agreement with AnaBios Focused on Human Primary Cardiomyocytes
SAN DIEGO, March 10, 2020 /PRNewswire/ — Today, AnaBios announced a Research Collaboration Agreement (RCA) with the U.S. Food and Drug Administration (FDA) to study adult human primary cardiomyocytes as a reference for cellular properties and drug-induced effects on cardiac function. Under the RCA, the FDA Center for Drug Evaluation and Research (CDER) […]
Carag First-ever Septal Occluder With Bioresorbable, Metal-free Framework Available to Patients Through New European Registry
First 3-year-old patient successfully implanted and discharged from hospital in Berlin BAAR, Switzerland and BERLIN, March 10, 2020 /PRNewswire/ — CARAG AG, a Swiss privately held medical device development company, announced today the successful implantation of the first-ever transcatheter septal occluder with bioresorbable, metal-free framework, as part of a new international post-market registry. The […]