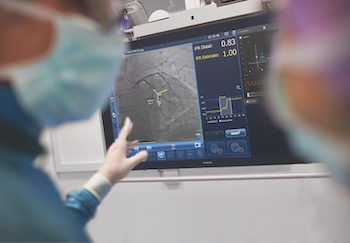
Randomized controlled trial with up to 3,000 participants at 75 sites to compare outcomes between physiological guidance by iFR data co-registered on the angiogram (interventional X-ray image) versus the standard of care use of an angiogram alone Percutaneous coronary intervention (PCI) is one of the main treatment options to open […]
Coronary/Structural Heart
PhaseBio Announces European Regulatory Update for PB2452
PB2452 granted PRIME Designation by the European Medicines Agency PhaseBio receives written scientific advice confirming PB2452 clinical development plan In Phase 1 and Phase 2a clinical trials, PB2452 provided immediate and sustained reversal of the antiplatelet effects of ticagrelor MALVERN, Pa. and SAN DIEGO, Feb. 11, 2020 (GLOBE NEWSWIRE) — PhaseBio […]
CorMatrix® Cardiovascular, Inc. receives FDA approval to expand and enroll 20 additional patients in the adult arm of its early feasibility IDE study of the Cor® TRICUSPID ECM® valve for pediatric and adult patients
ATLANTA, Feb. 7, 2020 /PRNewswire/ — CorMatrix®Cardiovascular, Inc. www.cormatrix.com, a leading developer of regenerative cardiovascular medical devices, today announced receiving FDA approval to expand and enroll 20 additional patients in the adult arm, including up to 4 additional cardiac surgery research and investigative centers, for its early feasibility IDE study of the Cor® TRICUSPID ECM® valve* for […]
Amarin Honored by BioNJ with Innovator Award for VASCEPA® Development and Regulatory Approval
DUBLIN, Ireland and BRIDGEWATER, N.J., Feb. 06, 2020 (GLOBE NEWSWIRE) — Amarin Corporation plc (NASDAQ:AMRN), announced today that BioNJ awarded Amarin with an Innovator Award in recognition of the approval of a new indication for VASCEPA® (icosapent ethyl) by the U.S. Food and Drug Administration (FDA) in December 2019. This […]
BioVentrix Receives 2020 NUB Reimbursement Status 1 Renewal in Germany for the Revivent TC TransCatheter Ventricular Enhancement System
SAN RAMON, Calif.–(BUSINESS WIRE)–BioVentrix, Inc., developer of the first transcatheter device for left ventricular remodeling after a heart attack, today announced it received renewal of the highest priority reimbursement in Germany for the Revivent TC™ TransCatheter Ventricular Enhancement System by achieving the NUB Status 1 designation from InEk, the German […]
Paragonix Technologies, Inc., Announces First Successful Use of Paragonix SherpaPak™ Cardiac Transport System in Pediatric Transplant Recipients
Paragonix SherpaPak™ Cardiac Transport System is the only FDA-cleared and CE-marked device that safeguards hearts between donor and recipient patients CAMBRIDGE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. today announced that the first series of pediatric donor hearts preserved and transported using Paragonix SherpaPak Cardiac Transport System occurred at University of Florida Health (UF […]
Inositec’s INS-3001 Significantly Reduces Cardiovascular Calcification in Proof-of-Concept Studies Published in Nature Communications
ZURICH–(BUSINESS WIRE)–Inositec, a pioneer in the development of life-saving small molecule drugs based on myo-inositol hexaphosphate (IP6), announced today that positive proof-of-concept data on INS-3001, a novel cardiovascular calcification inhibitor, were published in Nature Communications (DOI: 10.1038/s41467-019-14091-4). INS-3001 was found to be a potent and well tolerated inhibitor of pathological soft tissue calcification […]
Abbott’s In-Development Fully Implantable Heart Pump System Earns FDA’s Breakthrough Device Designation
ABBOTT PARK, Ill., Feb. 4, 2020 /PRNewswire/ — Abbott (NYSE: ABT) today announced that the company has received Breakthrough Device designation from the U.S. Food and Drug Administration (FDA) for its in-development Fully Implantable Left Ventricular Assist System (FILVAS). The FDA launched the Breakthrough Devices Program in 2018 to help expedite the development and review […]
Medtronic Begins New Pilot Study as Part of SPYRAL HTN Clinical Program for Renal Denervation in Hypertension Patients
SPYRAL DYSTAL Study to Evaluate the Effects of RDN Using Fewer, Targeted Ablations to Achieve Meaningful Blood Pressure Reductions DUBLIN, Feb. 04, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced it will begin enrollment in a pilot study evaluating the safety and efficacy of the Symplicity™ Spyral renal denervation (RDN) system […]
DILIsym Services Partners with Large Pharmaceutical Company to Begin QSP Model Development for Heart Failure
QSP Modeling Project Targeted at Prevention of Heart Failure RESEARCH TRIANGLE PARK, N.C.–(BUSINESS WIRE)–DILIsym Services Inc. (DSS), a Simulations Plus company (Nasdaq: SLP) and a leading provider of simulation and modeling software for pharmaceutical efficacy and safety, today announced that it is developing a new component for a QSP model […]