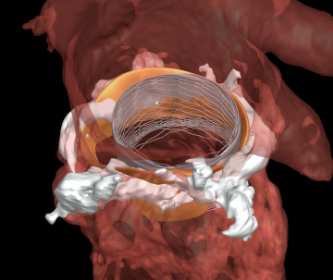
GENT, Belgium–(BUSINESS WIRE)–FEops, a leader in personalized predictive planning for structural heart interventions, is proud to announce that FEops HEARTguideTM was the winning entry at CSI Frankfurt 2019 voted on by physician attendees for the Highlight of the year award in the category interventional imaging. This award celebrates promising technologies that will have the […]
Coronary/Structural Heart
Eko Unveils Algorithm to Assist in the Detection of Aortic Stenosis, a Valvular Heart Disease Affecting Over 3 Million People in the U.S.
Berkeley, June 28, 2019 (GLOBE NEWSWIRE) — Eko, a Berkeley-based digital health company applying artificial intelligence and machine learning in the fight against heart disease, unveiled an investigational Aortic Stenosis (AS) detection algorithm at the American Society of Echocardiography (ASE) 2019 Scientific Sessions aimed to assist healthcare providers in the early detection of […]
Cardiac Dimensions Reaches 1,000 Implants Of Carillon® System, A Minimally Invasive Treatment For Functional Mitral Regurgitation
KIRKLAND, Wash., July 1, 2019 /PRNewswire/ — Cardiac Dimensions®, a leader in the development of innovative, minimally invasive treatments for functional mitral regurgitation (FMR) in patients with heart failure, today announced the Carillon Mitral Contour System® has been implanted in 1,000 patients in the United States, Europe, Australia, Turkeyand the Middle East. The Carillon System is a right-heart transcatheter mitral […]
Acarix CADScor®System included in NICE guidelines (UK)
Malmö June 27, 2019 Acarix CADScor®System included in NICE guidelines (UK) Acarix AB (publ) (ACARIX: FN Stockholm) today announced that the CADScor®System has been included as a first line investigation for ruling out suspected stable Coronary Artery Disease by the British National Institute for Health and Care Excellence (NICE). The […]
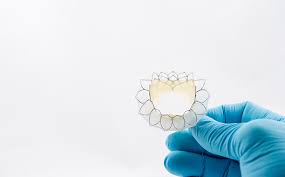
Neovasc Announces Presentations on Tiara™ for Treatment of Mitral Regurgitation and Reducer™ for Treatment of Refractory Angina at the CSI Frankfurt 2019 Conference
VANCOUVER, June 27, 2019 /PRNewswire/ – Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ:NVCN)(TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today announced that its Tiara™ (“Tiara”) transcatheter mitral valve replacement device […]
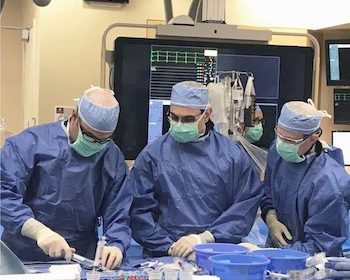
First TAVR Successfully Deployed at Henry Ford Allegiance Health
JACKSON, Mich., June 26, 2019 /PRNewswire/ — Doctors performed the first catheter-based replacement of an aortic heart valve today at Henry Ford Allegiance Health, part of the nationally recognized transcatheter aortic valve replacement (TAVR) program of Henry Ford Health System. TAVR is a minimally invasive alternative to open-heart valve replacement surgery. The first Henry […]
Amgen And MBC BioLabs Announce Winners Of The Amgen Golden Ticket
SAN FRANCISCO, June 25, 2019 /PRNewswire/ — Amgen (NASDAQ:AMGN) and MBC BioLabs (formerly QB3@953) today announced that Regencor and Nitrome Biosciences have each been awarded an Amgen Golden Ticket to MBC BioLabs. The companies receive priority admission or renewal for one year of lab bench space and access to core facilities at the MBC BioLabs […]
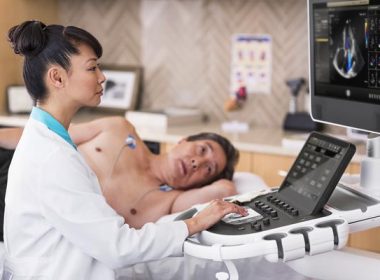
Philips extends advanced automation capabilities on its EPIQ CVx cardiology ultrasound platform, making accurate exams faster and easier to conduct
June 20, 2019 With new release, clinicians can reduce the number of touches of the system by 21% per exam, equivalent to more than 400 exams each year Platform provides advanced automation for right ventricle volumes and ejection fraction measurements in as little as 15 seconds, speeding exam times Amsterdam, […]
FFR-SEARCH Shows Clear Trend Towards Major Adverse Cardiac Events with Lower FFR
EDEN PRAIRIE, Minn., June 20, 2019 /PRNewswire/ — Dr. Roberto Diletti from The Erasmus Center in Rotterdam, Netherlands, presented the results of the two-year follow-up analysis of the FFR-SEARCH observational registry during the EuroPCR conference in Paris. The analysis evaluated the outcomes of patients and prognostic value of post-percutaneous coronary intervention (post-PCI) FFR assessment, indicating patients with a […]
Study Links Use of the Baylis Medical RF Transseptal Needle to Procedural Cost-Savings
TORONTO, June 19, 2019 /PRNewswire/ – A cost-effectiveness analysis completed by the University of California, San Francisco (UCSF) and Baylis Medical has shown that use of Baylis’ RF transseptal needle results in cost savings per procedure, compared to use of a mechanical transseptal needle. The study, entitled A Cost-Effectiveness Analysis Comparing the Baylis RF Needle to […]