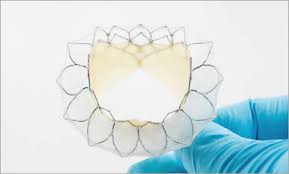
VANCOUVER, March 4, 2019 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN)(TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today announced that its Tiara™ (“Tiara”) transcatheter mitral valve replacement device […]
Coronary/Structural Heart
VGS Completes Enrollment of VEST U.S. Pivotal Trial for the Evaluation of Its Venous External Support Technology for Coronary Bypass Surgery
TEL AVIV, Israel, March 5, 2019 /PRNewswire/ — Vascular Graft Solutions Ltd. (VGS) announces the completion of patient enrollment in the company’s US Investigational Device Exemption (IDE) randomized, controlled pivotal study, evaluating the efficacy of the VEST external support for saphenous vein grafts in coronary artery bypass (CABG) surgery. The study enrolled 224 […]
Innovative Cardiovascular Systems Announces First Use of its Next-Generation Emblok™ Embolic Protection System in European Feasibility Study
KALAMAZOO, Mich., March 5, 2019 /PRNewswire/ — Innovative Cardiovascular Solutions, LLC (ICS), a privately held medical device company, today announced the first clinical use of its next-generation Emblok™ Embolic Protection System in patients undergoing transcatheter aortic valve replacement (TAVR) procedures in its European feasibility study. The cases were successfully performed by primary investigator […]
Microbot Medical Announces Notice of Intention to Grant of European Patent for Adjustable Guidewire Technology
HINGHAM, Mass., March 04, 2019 (GLOBE NEWSWIRE) — Further expanding its global IP portfolio, Microbot Medical Inc. (Nasdaq CM: MBOT) received an official communication from the European Patent Office regarding its intention to grant European Patent Application No. 13 746 563, which covers an adjustable guidewire for use in percutaneous […]
MyoKardia Announces Positive Six-Month Safety and Efficacy Data from PIONEER-OLE Study of Mavacamten for the Treatment of Symptomatic, Obstructive HCM
SOUTH SAN FRANCISCO, Calif., March 04, 2019 (GLOBE NEWSWIRE) — MyoKardia, Inc. (Nasdaq: MYOK), a clinical-stage biopharmaceutical company pioneering a precision medicine approach for the treatment of serious cardiovascular diseases, today announced positive 12- and 24-week safety, efficacy and biomarker data for its investigational drug, mavacamten, from the company’s PIONEER […]
National Institutes of Health awards Geisinger over $3.1 million for research of sudden cardiac death
DANVILLE, Pa., March 4, 2019 /PRNewswire/ — The National Institutes of Health has awarded researchers at Geisinger $3.1 million over the next five years to explore innovative approaches for early, genomics-based detection of arrhythmogenic right ventricular cardiomyopathy (ARVC). ARVC is a degenerative heart disease responsible for approximately 20 percent of sudden cardiac death in individuals […]
BioVentrix Achieves 2019 NUB Reimbursement Status 1 in Germany for Revivent TC™ TransCatheter Ventricular Enhancement System
SAN RAMON, Calif., Feb. 28, 2019 /PRNewswire/ — BioVentrix, Inc., a pioneer of technologies and procedures for less invasive treatment of heart failure (HF), today announced a significant reimbursement achievement. InEk, the German Institute for Hospital Remuneration, has reconfirmed the award of NUB Status 1 for the Revivent TC™ TransCatheter Ventricular Enhancement System. […]
Blackstone Life Sciences and Novartis Launch Anthos Therapeutics to Develop Innovative Medicines for Cardiovascular Disease
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Blackstone Life Sciences today announced the launch of Anthos Therapeutics Inc. (“Anthos”), a new biopharmaceutical company focused on advancing next-generation targeted therapies for high-risk cardiovascular patients. As part of this launch, Novartis has licensed to Anthos MAA868, an antibody directed at Factor XI and XIa, key components of […]
Medtronic Resolute(TM) Drug-Eluting Stent (DES) Platform Receives Expanded Indication for Treatment of Chronic Total Occlusion (CTO)
DUBLIN – February 26, 2019 – Medtronic plc (NYSE:MDT) today announced the U.S. Food and Drug Administration (FDA) approval of its Resolute Drug-Eluting Stent (DES) platform (including the Resolute Onyx(TM) and Resolute Integrity(TM) DES) for the treatment of patients with coronary artery disease who have de novo chronic total occlusion (CTO), a complex […]
LivaNova Announces Availability of Safety and Technical Information for Valve-in-Valve Procedures on Perceval Platform
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN), a market-leading medical technology company, announced today that given the increasing viability of Valve-in-Valve (ViV) procedures, it has added new safety and technical information to the Perceval U.S. Instructions For Use (IFUs). This addition was made following completion of the Company’s application to the U.S. Food […]