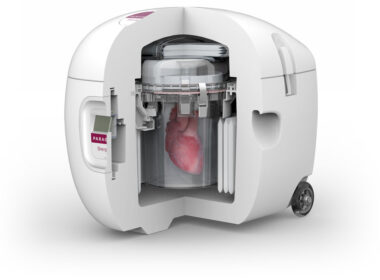
Data from the GUARDIAN Registry, presented at the 45th ISHLT Annual Meeting, shows the Paragonix SherpaPak ® Cardiac Transport System led to statistically significant improvements in short and long-term post-transplant survival and cardiac complications WALTHAM, MA. April 29, 2025 – Paragonix Technologies , a leader in organ transplant technologies and […]
Coronary/Structural Heart
CareDx Announces New Data on AlloSure Lung Presented at 2025 International Society for Heart and Lung Transplantation Meeting
Findings from ALAMO Study Show AlloSure Lung is Associated with Prognosis of Chronic Lung Allograft Dysfunction and Restrictive Allograft Syndrome AlloSure Lung Surveillance Monitoring Identifies the Onset of Acute Cellular Rejection and Treatment Response BRISBANE, Calif.–(BUSINESS WIRE)–CareDx, Inc. (Nasdaq: CDNA) – The Transplant Company™ – a leading precision medicine company […]
Orchestra BioMed Receives FDA Approval of IDE to Initiate U.S. Coronary Pivotal Trial Randomizing First-in-Class Sirolimus-AngioInfusion Balloon, Virtue SAB, Head-to-Head with Paclitaxel-Coated Balloon
Orchestra BioMed’s Virtue® Sirolimus AngioInfusion Balloon™ (“Virtue SAB”) is the only non-coated drug-eluting balloon system under clinical investigation worldwide and has been awarded multiple FDA Breakthrough Device DesignationsThe Virtue Trial will be the first U.S. IDE head-to-head randomized evaluation of a sirolimus-eluting balloon versus a commercially available paclitaxel-coated balloon (AGENT™) for the treatment of coronary in-stent restenosis (“ISR”)Robust non-inferiority trial is designed to provide a clear pathway to regulatory approval as well as potentially showcase clinical advantages of Virtue SABWith the amended IDE approved by the FDA, Orchestra BioMed is currently targeting initiation of the Virtue Trial during the second half of 2025 NEW HOPE, Pa., April 29, 2025 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO, “Orchestra BioMed” or the “Company”), a biomedical company accelerating high-impact technologies to patients through risk-reward sharing partnerships, today announced that the U.S. Food and Drug Administration (“FDA”) has approved its Investigational Device Exemption (“IDE”) amendment to initiate an updated design of the Company’s planned Virtue SAB in the Treatment of Coronary ISR Trial (“Virtue Trial”). The IDE provides FDA regulatory clearance for Orchestra BioMed to initiate a U.S. pivotal clinical trial comparing its highly differentiated, next-generation Sirolimus-AngioInfusion Balloon, Virtue SAB to the Boston Scientific AGENT paclitaxel-coated balloon, currently the only drug-coated balloon (“DCB”) FDA-approved for a coronary indication. Data from the Virtue Trial will be used to support regulatory approval in the U.S. Virtue SAB and SirolimusEFR are investigational technologies owned by Orchestra BioMed, which also controls and is responsible for all regulatory filings, clinical operations, and drug and device supplies for the Virtue Trial. The Differentiation of Virtue SAB, An FDA Breakthrough Designated DeviceVirtue SAB is designed to deliver a proprietary extended-release formulation of sirolimus, SirolimusEFR™, through a non-coated microporous AngioInfusion™ Balloon that protects the drug in transit to consistently deliver a large liquid dose, overcoming certain limitations of DCBs. SirolimusEFR™ enables tissue uptake and extended release of the required therapeutic levels of sirolimus ( > 1ng/mg tissue concentration), the “gold-standard” drug for preventing arterial restenosis, through the critical healing period of approximately 30 days. Virtue SAB has been previously granted FDA Breakthrough Device Designation for the treatment of coronary ISR as well as for coronary small vessel disease and peripheral artery disease below-the-knee. In the multi-center SABRE pilot study, Virtue SAB demonstrated best-in-class clinical results for the treatment of coronary ISR, including 12-month target lesion failure of 2.8% and 6-month late lumen loss of 0.12mm. “Virtue SAB has the potential to be one of the most compelling technologies in interventional cardiology. It’s the only product in development that optimizes both the arterial tissue uptake and retention of sirolimus to achieve pharmacokinetics that match or even exceed those of proven ‘limus-eluting stents,” said Dean J. Kereiakes, M.D., FACC, MSCAI, Chairman of The Christ Hospital Heart & Vascular Institute, Medical Director of The Christ Hospital Research Institute, Professor of Clinical Medicine at The Ohio State University, Professor of Medicine at University of Cincinnati and Co-Principal Investigator on the Virtue Trial. Dr. Kereiakes continued: “Virtue SAB is designed to consistently deliver a large liquid dose of an extended-release formulation of sirolimus to overcome certain limitations of traditional DCBs, including lower doses due to surface area and coating integrity constraints, drug loss in transit leading to inconsistent dosing, and the risk of emboli from large coating particulates. As the coronary treatment landscape continues to shift toward more rapid adoption of DCBs, I’m excited about the potential of Virtue SAB to set a new standard of care.” “Drug-coated balloons are emerging as a new standard of care in the treatment of various coronary and peripheral indications, and I believe utilization of this class of technology will continue to grow and evolve over time as the science is expanding. In a field largely reliant on paclitaxel drug-coated balloons, Virtue SAB stands out as the only device with a completely different mechanism of action; namely to provide delivery of a large liquid dose of an extended-release formulation sirolimus,” commented Allen Jeremias, MD, MSc, Associate Director of the Cardiac Catheterization Laboratory at St. Francis Hospital & Heart Center, and Co-Principal Investigator on the Virtue Trial. “Having had the opportunity to work with several DCBs, I anticipate continued momentum for this class, and am eager to see how Virtue SAB, particularly with its anti-restenotic, anti-inflammatory and cytostatic SirolimusEFR formulation, performs in a head-to-head trial against a paclitaxel-coated balloon.” Showcasing Distinctive and Sustainable Advantages of Virtue SAB Through a Head-to-Head Trial “We believe there is a multibillion-dollar U.S. market for coronary drug delivery balloons based on the significant unmet clinical need, market demand, and established reimbursement,” said David Hochman, Chairman and Chief Executive Officer of Orchestra BioMed. “We made a deliberate, strategic decision to pursue a head-to-head trial with the commercially available AGENT paclitaxel-coated balloon, underscoring our confidence in Virtue SAB as a fundamentally differentiated solution for the treatment of atherosclerosis. We believe this approach offers the most direct path to regulatory approval while also providing the best opportunity to demonstrate what we believe are distinctive and sustainable advantages of our proprietary technology.” Mr. Hochman continued, “The superior safety and efficacy of sirolimus over paclitaxel was made clear by the performance of drug-eluting stents. Published meta-analysis involving 76 trials show ‘limus-eluting stents have significantly better clinical performance than paclitaxel-eluting stents in terms of target lesion revascularization and major adverse cardiac events.1 We believe this is due to their ability to maintain sufficient drug tissue concentration through the critical healing period of approximately 30 days, because stents that failed to do this did not perform well clinically.2,3,4 Virtue SAB is the only drug delivery balloon that has demonstrated comparable drug tissue levels to clinically successful drug-eluting stents in large published preclinical studies, without the need to leave a permanent metal implant in the artery. Our pilot clinical results with Virtue SAB also highlight the potential for optimal clinical outcomes with robust sirolimus delivery. We’re excited to showcase the full potential of Virtue SAB in this landmark trial and are proud of our team and grateful to our clinical collaborators for their work in achieving this important milestone.” The Virtue Trial is a prospective, multi-center, randomized trial comparing clinical outcomes of Virtue SAB to AGENT Paclitaxel DCB in the treatment of coronary ISR, a difficult-to-treat and serious complication of coronary stenting. The primary endpoint is a non-inferiority comparison of Target Lesion Failure (TLF) defined as a composite of cardiac death, nonfatal target vessel myocardial infarction and ischemia-driven target lesion revascularization at 12 months. The trial will randomize 740 patients across up to 75 centers in the U.S. With the amended IDE approved by the FDA, Orchestra BioMed is currently targeting initiation of the Virtue Trial during the second half of 2025, bringing the Company one step closer to delivering a next-generation solution for atherosclerotic disease. About Coronary In-Stent Restenosis (ISR)Coronary ISR is a serious complication of coronary stenting, which can increase the risk of life-threatening heart problems. It is characterized by a re-narrowing of a coronary artery segment that was previously treated with a stent. According to the National Cardiovascular Data Registry, coronary ISR occurs in up to 10% of stented patients during the first year and continues at a rate of up to 3% per year thereafter, resulting in an estimated over 325,000 coronary ISR lesions annually worldwide that may require treatment. If left untreated, coronary ISR may lead to stable angina, unstable angina, acute coronary syndrome, acute myocardial infarction, or death. About Virtue SABVirtue SAB is designed to deliver a proprietary extended-release formulation of sirolimus, SirolimusEFR™ through a non-coated microporous AngioInfusion™ Balloon that protects the drug in transit to consistently deliver a large liquid dose overcoming certain limitations of drug-coated balloons. SirolimusEFR delivered by Virtue SAB has been shown in published preclinical series involving hundreds of arterial deliveries to achieve sustained tissue levels well above the known required therapeutic tissue concentration for inhibiting restenosis (1 ng/mg tissue) for the entire critical healing period of approximately 30 days. Virtue SAB demonstrated positive three-year clinical data in coronary ISR in the SABRE study, a multi-center prospective, independent core lab-adjudicated clinical study of 50 patients conducted in Europe. Virtue SAB has been granted Breakthrough Device Designation by the FDA for specific indications relating to coronary ISR, coronary small vessel disease and peripheral artery disease below-the-knee. About Orchestra BioMed Orchestra BioMed (Nasdaq: OBIO) is a biomedical innovation company accelerating high-impact technologies to patients through risk-reward sharing partnerships with leading medical device companies. Orchestra BioMed’s partnership-enabled business model focuses on forging strategic collaborations with leading medical device companies to drive successful global commercialization of products it develops. Orchestra BioMed’s lead product candidate is atrioventricular interval modulation (AVIM) therapy for the treatment of hypertension, the leading risk factor for death worldwide. Orchestra BioMed is also developing the Virtue® Sirolimus AngioInfusion™ Balloon (SAB) for the treatment of atherosclerotic artery disease, the leading cause of mortality worldwide. Orchestra BioMed has a strategic collaboration with Medtronic, one of the largest medical device companies in the world, for development and commercialization of AVIM therapy for the treatment of hypertension in pacemaker-indicated patients, and a strategic partnership with Terumo, a global leader in medical technology, for development and commercialization of Virtue SAB for the treatment of artery disease. The company has received four Breakthrough Device Designations from the U.S. FDA across these two core programs, reflecting the significant potential of its technologies to address high unmet needs in cardiovascular care. For further information about Orchestra BioMed, please visit www.orchestrabiomed.com, and follow us on LinkedIn. References to Websites and Social Media Platforms References to information included on, or accessible through, websites and social media platforms do not constitute incorporation by reference of the information contained at or available through such websites or social media platforms, and you should not consider such information to be part of this press release. Forward-Looking StatementsCertain statements included in this press release that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements relating to the timing of the initiation and of the Virtue Trial, the number of patients to be enrolled in the Virtue Trial, and the potential safety and efficacy of the Company’s product candidates, including the ability of Virtue SAB to overcome certain limitations of DCBs. These statements are based on various assumptions, whether or not identified in this press release, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to regulatory approval of the Company’s product candidates; the timing of, and the Company’s ability to achieve, expected regulatory and business milestones; the impact of competitive products and product candidates; and the risk factors discussed under the heading “Item 1A. Risk Factors” in the Company’s annual report on Form 10-K filed with the U.S. Securities and Exchange Commission on March 31, 2025, as updated by any risk factors disclosed under the heading “Item 1A. Risk Factors” in the Company’s subsequently filed quarterly reports on Form 10-Q. The Company operates in a very competitive and rapidly changing environment. New risks emerge from time to time. Given these risks and uncertainties, the Company cautions against placing undue reliance on these forward-looking statements, which only speak as of the date of this press release. The Company does not plan and undertakes no obligation to update any of the forward-looking statements made herein, except as required by law. Investor ContactJeremy FefferLifeSci AdvisorsJfeffer@lifesciadvisors.com Media ContactKelsey Kirk-EllisOrchestra BioMedkkirkellis@orchestrabiomed.com References 1Xinlin Zhang, et. al. PLOS ONE 2014 May 20;9(5):e97934. 2https://slideplayer.com/slide/5787004. 3 Tada, et. Al., Am Heart J. 2013 Jan;165(1):80-6. 4Leon M LBT III, Session 3014 ACC 2011.
Sensydia Announces First Patient Enrolled in Pivotal Study of Non-Invasive Cardiac Performance System (CPS™)
LOS ANGELES–(BUSINESS WIRE)–Sensydia, a medical technology company pioneering non-invasive cardiac assessment, today announced that the first patient has been enrolled in its multi-center pivotal study to evaluate the accuracy of the Cardiac Performance System (CPS) for hemodynamic assessment as compared to the gold-standard: invasive right heart catheterization. Sensydia’s CPS acquires cardiovascular signals, […]
CorWave Announces Successful Completion of 6-Month Chronic In Vivo Study, Paving the Way for Clinical Trial Initiation
In vivo studies demonstrated the pump’s successful operation for a period of up to six months. Additionally, nine chronic ovine implants were conducted for 60 days with no device failure or sign of thrombosis at explant. These significant milestones mark the final stage of CorWave’s preclinical development, paving the way […]
Elixir Medical Announces First Use of LithiX Hertz Contact (HC) Intravascular Lithotripsy System (IVL) in Asia Pacific at Cardiac Vascular Sentral Kuala Lumpur (CVSKL), Malaysia
First cases completed by Dr. Tamil Selvan Muthusamy, MD, and Dr. Rosli Mohd Ali, MD, and transmitted live at TCTAP Congress in Seoul, Korea. Novel IVL technology designed to deliver calcium fragmentation for treatment of moderate to severely calcified coronary artery lesions without requiring an external energy source. MILPITAS, Calif., April 25, 2025 (GLOBE NEWSWIRE) — Elixir Medical, a developer of disruptive technologies to treat cardiovascular disease, today announced the first clinical use of its LithiX™ Hertz Contact (HC) Intravascular Lithotripsy System (IVL) in the Asia Pacific region, marking a significant milestone in the device’s global introduction. The procedures were successfully performed at Cardiac Vascular Sentral Kuala Lumpur Hospital in Kuala Lumpur by Dr. Tamil Selvan Muthusamy, MD, and Dr. Rosli Mohd Ali, MD. The LithiX™ HC-IVL device represents a novel mechanical approach to calcium modification in calcified coronary lesions, offering the benefits of intravascular lithotripsy using prior technologies without requiring an external energy source. Based on the Hertz Contact Stress principle, the device delivers precise mechanical amplification of force to achieve calcium fractures while minimizing unintended vessel trauma. “The ability to safely perform complex PCI and treat severe calcium with a mechanical IVL system like LithiX™ is a significant advancement for our practice and a big win for patients in our region,” said Dr. Tamil Selvan Muthusamy, MD. “We’re proud to be the first center in Asia to adopt this innovative technology and demonstrate the excellent clinical outcomes via a live case transmission at TCTAP congress.” The LithiX™ HC-IVL System features low-profile metal hemispheres integrated on a semi-compliant balloon. When inflated at low pressure, via the Hertz Contact Stress principle, these hemispheres amplify force at contact points with calcium to produce deep and wide fractures, effectively modifying complex calcified lesions while minimizing injury to healthy vessel tissue. “Prevalence of calcified lesions is very high in Asia Pacific, and the interventional cardiology community has long been waiting for a more optimal IVL solution to treat our patients. With LithiX™, we now have a simple-to-use and versatile lithotripsy solution that demonstrated excellent results in our very complex cases,” said Dr. Rosli Mohd Ali, MD. Motasim Sirhan, CEO of Elixir Medical, stated, “With LithiX™, we’ve developed the first mechanical IVL platform capable of fracturing calcium without requiring an external energy source while avoiding injury to adjacent healthy vessel. The expansion into Malaysia and the greater Asia Pacific region demonstrates global recognition of its unique performance and procedural advantages.” About Elixir Medical Elixir Medical Corporation, a privately held company based in Milpitas, California, develops disruptive platforms to treat coronary and peripheral artery disease. Our transformative technologies have multiple applications across the cardiovascular space capable of delivering improved clinical outcomes for millions of patients. Elixir Medical was named to Fast Company’s prestigious list of the World’s Most Innovative Companies of 2025. Visit us at www.elixirmedical.com and on LinkedIn and X. The LithiX™ HC-IVL system is CE Mark approved. Not available for sale in the United States. CONTACT: Richard Laermer
RLM PR
elixir@rlmpr.com
(212) 741-5106 X 216
Autonomix Medical, Inc. Technology and Early Proof-of-Concept Study Results to be Featured in Poster Presentation at the World-Leading Course in Interventional Cardiovascular Medicine, EuroPCR
THE WOODLANDS, TX, April 24, 2025 (GLOBE NEWSWIRE) — Autonomix Medical, Inc. (NASDAQ: AMIX) (“Autonomix” or the “Company”), a medical device company focused on advancing precision nerve-targeted treatments, today announced its technology and early proof-of-concept study results will be featured in a poster presentation as part of PCR Innovators Day at the world-leading course in interventional cardiovascular medicine, EuroPCR, being held May 20-23, 2025 in Paris, France.
Renibus Therapeutics Announces Completion of Enrollment in PROTECT, a Pivotal Phase 3 Clinical Trial of RBT-1 to Reduce the Risk of Post-Operative Complications After Cardiac Surgery
– Topline results expected in Q3 2025 – SOUTHLAKE, Texas, April 24, 2025 /PRNewswire/ — Renibus Therapeutics® (“Renibus”), a clinical late-stage biopharmaceutical company focusing on the prevention and treatment of cardiac, renal, and metabolic diseases, today announced the completion of…
Mineralys Therapeutics Announces Publication of Pivotal Phase 2 Advance-HTN Results in the New England Journal of Medicine (NEJM)
– Significant blood pressure reductions among patients with uncontrolled or resistant hypertension treated with lorundrostat reinforce key role of dysregulated aldosterone in disease onset and progression – – Detailed results from the second pivotal Phase 3 Launch-HTN trial to be presented at an upcoming medical conference and published in a peer-reviewed publication – RADNOR, Pa., April 23, 2025 (GLOBE NEWSWIRE) — Mineralys Therapeutics, Inc. (Nasdaq: MLYS), a clinical-stage biopharmaceutical company focused on developing medicines to target hypertension, chronic kidney disease (CKD), obstructive sleep apnea (OSA) and other diseases driven by dysregulated aldosterone, today announced that the New England Journal of Medicine (NEJM) published the detailed results from the Company’s pivotal Phase 2 Advance-HTN trial, the first of two pivotal trials evaluating lorundrostat in patients with uncontrolled hypertension (uHTN) or resistant hypertension (rHTN). The full manuscript is titled, “Lorundrostat Efficacy and Safety in Patients with Uncontrolled Hypertension,” and is featured in the April 23, 2025 issue of NEJM. The key data from the publication showed that lorundrostat 50 mg demonstrated a 15.4 mmHg absolute reduction and a 7.9 mmHg placebo-adjusted reduction (p=0.001), in 24-hour ambulatory blood pressure at week 12. Lorundrostat worked equally well in those taking two baseline medications and those taking three or more, and in both men and women as well as in white and black patients. Lorundrostat demonstrated a favorable safety and tolerability profile, with modest changes in potassium, sodium and eGFR. “The publication of our Advance-HTN trial results in the New England Journal of Medicine is a significant milestone that underscores both the strength of our clinical data and the potentially transformative nature of this new class of medicines that could help address dysregulated aldosterone, an unaddressed, key driver of hypertension,” stated Jon Congleton, Chief Executive Officer of Mineralys Therapeutics. “Prior studies have shown that even modest reductions in systolic blood pressure can lead to a substantial decrease in the incidence of major cardiovascular events. The blood pressure reductions with lorundrostat observed in the Advance-HTN trial are particularly meaningful given the well-established correlation between elevated blood pressure, dysregulated aldosterone production and cardiovascular risk.” “The significant blood pressure lowering with lorundrostat 50 mg in the Advance-HTN trial was seen in patients treated by specialists who were taking an optimized standardized antihypertensive regimen – those patients with true uncontrolled or resistant hypertension that desperately need new options to lower their blood pressure,” stated Luke Laffin, M.D., co-director of the Center for Blood Pressure Disorders in the Heart, Vascular & Thoracic Institute at Cleveland Clinic and the study’s lead author. “Currently available therapies to treat hypertension do not decrease aldosterone production in the body, and we know aldosterone dysregulation is a driving factor in the blood pressure elevation of many of our patients. The findings reinforce the critical role of aldosterone in the pathogenesis of hypertension and the potential of lorundrostat to address unmet medical needs facing patients with uncontrolled or treatment-resistant disease.” The NEJM publication of the detailed Advance-HTN results follows a late-breaking presentation of the data at the American College of Cardiology’s Annual Scientific Session & Expo (ACC.25) in Chicago on March 29, 2025, and the announcement of positive topline results from both Advance-HTN and Launch-HTN earlier in March. Mineralys plans to provide additional data from the pivotal Phase 3 Launch-HTN at an upcoming medical conference and in a peer-reviewed publication. Additionally, the ongoing Transform-HTN open-label extension trial allows subjects to continue to receive lorundrostat and obtain additional safety and efficacy data. About Hypertension Having sustained, elevated blood pressure (or hypertension) increases the risk of heart disease, heart attack and stroke, which are leading causes of death in the U.S. In 2020, more than 670,000 deaths in the U.S. included hypertension as a primary or contributing cause. Hypertension and related health issues resulted in an average annual economic burden of about $219 billion in the U.S. in 2019. Less than 50% of hypertension patients achieve their blood pressure goal with currently available medications. Dysregulated aldosterone levels are a key factor in driving hypertension in approximately 30% of all hypertensive patients. About Lorundrostat Lorundrostat is a proprietary, orally administered, highly selective aldosterone synthase inhibitor being developed for the treatment of uHTN or rHTN, as well as CKD and OSA. Lorundrostat was designed to reduce aldosterone levels by inhibiting CYP11B2, the enzyme responsible for its production. Lorundrostat has 374-fold selectivity for aldosterone-synthase inhibition versus cortisol-synthase inhibition in vitro, an observed half-life of 10-12 hours and demonstrated approximately a 70% reduction in plasma aldosterone concentration in hypertensive subjects. In a Phase 2, proof-of-concept trial (Target-HTN) in uncontrolled or resistant hypertensive subjects, once-daily lorundrostat demonstrated statistically significant and clinically meaningful blood pressure reduction in both automated office blood pressure measurement and 24-hour ambulatory blood pressure monitoring. Adverse events observed were a modest increase in serum potassium, decrease in estimated glomerular filtration rate, urinary tract infection and hypertension with one serious adverse event possibly related to study drug being hyponatremia. About Advance-HTN The Advance-HTN trial (NCT05769608) was a randomized, double-blind, placebo-controlled Phase 2 clinical trial that evaluated the efficacy and safety of lorundrostat for the treatment of uHTN or rHTN, when used as an add-on therapy to a standardized background treatment of two or three antihypertensive medications in adult subjects. Subjects who met screening criteria had their existing hypertension medications discontinued and started on a standard regimen of an angiotensin II receptor blocker (ARB) and a diuretic, if previously on two medications, or a standard regimen of ARB, diuretic and calcium channel blocker if previously on three to five medications. Subjects who remained hypertensive despite the standardized regimen were then randomized into three cohorts and treated for twelve weeks: lorundrostat 50 mg once-daily (QD), lorundrostat 50 mg QD and an option to titrate to 100 mg QD at week four based on defined criteria, or placebo. The trial’s primary endpoint was the change in 24-hour ambulatory systolic blood pressure at week twelve from baseline for active cohorts versus placebo. About Mineralys Mineralys Therapeutics is a clinical-stage biopharmaceutical company focused on developing medicines to target hypertension, CKD, OSA and other diseases driven by dysregulated aldosterone. Its initial product candidate, lorundrostat, is a proprietary, orally administered, highly selective aldosterone synthase inhibitor that Mineralys Therapeutics is developing for the treatment of cardiorenal conditions affected by dysregulated aldosterone, including hypertension, CKD and OSA. Mineralys is based in Radnor, Pennsylvania, and was founded by Catalys Pacific. For more information, please visit https://mineralystx.com. Follow Mineralys on LinkedIn and Twitter. Forward Looking Statements Mineralys Therapeutics cautions you that statements contained in this press release regarding matters that are not historical facts are forward-looking statements. The forward-looking statements are based on our current beliefs and expectations and include, but are not limited to, statements regarding: the potential therapeutic benefits of lorundrostat; the Company’s expectation that Advance-HTN and Launch-HTN may serve as pivotal trials in any submission of a new drug application (NDA) to the United States Food and Drug Administration (FDA); the Company’s ability to evaluate lorundrostat as a potential treatment for CKD, OSA, uHTN or rHTN; and the planned future clinical development of lorundrostat and the timing thereof. Actual results may differ from those set forth in this press release due to the risks and uncertainties inherent in our business, including, without limitation: topline results that we report are based on a preliminary analysis of key efficacy and safety data, and such data may change following a more comprehensive review of the data related to the clinical trial and such topline data may not accurately reflect the complete results of a clinical trial; our future performance is dependent entirely on the success of lorundrostat; potential delays in the commencement, enrollment and completion of clinical trials and nonclinical studies; later developments with the FDA may be inconsistent with the feedback from the completed end of Phase 2 meeting, including whether the proposed pivotal program will support registration of lorundrostat which is a review issue with the FDA upon submission of an NDA; the results of our clinical trials, including the Advance-HTN and Launch-HTN trials, may not be deemed sufficient by the FDA to serve as the basis for an NDA submission or regulatory approval of lorundrostat; our dependence on third parties in connection with manufacturing, research and clinical and nonclinical testing; unexpected adverse side effects or inadequate efficacy of lorundrostat that may limit its development, regulatory approval and/or commercialization; unfavorable results from clinical trials and nonclinical studies; results of prior clinical trials and studies of lorundrostat are not necessarily predictive of future results; regulatory developments in the United States and foreign countries; our reliance on our exclusive license with Mitsubishi Tanabe Pharma to provide us with intellectual property rights to develop and commercialize lorundrostat; and other risks described in our filings with the Securities and Exchange Commission (SEC), including under the heading “Risk Factors” in our annual report on Form 10-K, and any subsequent filings with the SEC. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, and we undertake no obligation to update such statements to reflect events that occur or circumstances that exist after the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement, which is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Contact:Investor Relationsinvestorrelations@mineralystx.com Media RelationsTom WeibleElixir Health Public RelationsPhone: (1) 515-707-9678Email: tweible@elixirhealthpr.com
PrepMD OMNI: The Most Comprehensive CIED Software Solution, Now Listed in the Epic Showroom
BRAINTREE, Mass., April 23, 2025 /PRNewswire/ — PrepMD OMNI’s inclusion in the Epic Showroom establishes it as a trusted, integrated solution within one of the nation’s leading EHR ecosystems. This visibility gives hospitals and cardiac care centers across the country easier access to…