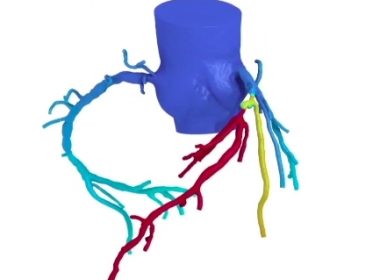
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) has completed enrollment for the pivotal phase of the Gore ASSURED Clinical Study. This investigational device exemption (IDE) trial is researching the new GORE®CARDIOFORM ASD Occluder for the interventional closure of Atrial Septal Defects (ASDs), sized 8 to 35 mm. The […]
Coronary/Structural Heart
SentreHEART Announces 400th Patient Enrollment in the Landmark aMAZE Trial
REDWOOD CITY, Calif.–(BUSINESS WIRE)–SentreHEART, Inc., manufacturer of the LARIAT® Suture Delivery Device (LARIAT) for percutaneous left atrial appendage (LAA) closure announces enrollment of the 400th patient in the prospective, multi-center, randomized controlled Trial known as the aMAZE Trial. The Trial is a superiority design and intends to demonstrate the LARIAT procedure […]
NeoChord Announces Successful Completion of First Beating Heart Repair Procedures in China
ST. LOUIS PARK, Minn., Sept. 04, 2018 (GLOBE NEWSWIRE) — NeoChord, Inc., a privately-held medical technology company leading the advancement of beating heart mitral valve repair in patients suffering from mitral valve regurgitation, today announced that it has successfully treated the first patients in Asia using the NeoChord procedure at Queen […]
Firehawk® Clinical Data From The TARGET AC European Clinical Trial Has Been Accepted For Publication In The Prestigious Medical Journal The Lancet
SHANGHAI, Sept. 3, 2018 /PRNewswire/ — MicroPort Scientific Corporation (“MicroPort®”, HK:0853) announced today that the clinical trial results for its Firehawk® Rapamycin Target Eluting Coronary Stent System (“Firehawk®”) which conducted a multi-center, randomized controlled trial called TARGET AC has been published online in The Lancet, a world leading medical journal currently ranked second […]
Lower Cholesterol Treatment Breakthrough
GORDON, Australia, Aug. 29, 2018 /PRNewswire/ — The American offshoot of Australian biotech Noxopharm, Nyrada, believes it is within reach of achieving a major advance in the treatment of cardiovascular disease. It has made important progress with a drug candidate known as NYX-330 that Nyrada believes may be the next breakthrough medication for […]
FAST-FFR Pivotal Trial Added to TCT 2018 Late-Breaking Clinical Trial Program
KFAR-SABA, Israel–(BUSINESS WIRE)–CathWorks announced that the FAST-FFR pivotal trial has been added to the late-breaking clinical science program during the 2018 TCT (Transcatheter Cardiovascular Therapeutics) annual meeting in San Diego. FAST-FFR trial data will be presented in the Main Arena during the late-breaking science session that begins at 12 noon […]
XBiotech Announces Decisive Results on the Potential Role of Interleukin-1 Alpha in Heart Attack Risk
AUSTIN, Texas, August 27, 2018 (GLOBE NEWSWIRE) – XBiotech Inc. (NASDAQ: XBIT) today announced the release of results indicating that leukocyte-derived Interleukin-1 alpha (IL-1⍺) is the cause clots that can cause a heart attack or stroke. The research was led by a world-renowned cardiovascular researcher, Dr. Peter Libby, Professor Mallinckrodt of […]
European Society of Cardiology incorporates iFR technology into updated guidelines for revascularization
AMSTERDAM and MUNICH, Aug. 28, 2018 /PRNewswire/ — Royal Philips (NYSE : PHG , AEX: PHIA), a global leader in health technology, today announced that the European Society of Cardiology has incorporated instantaneous wave-free ratio (iFR) into its updated revascularization guidelines. The new guidelines have provided the highest recommendation (class I A) for iFR alongside fractional flow reserve (FFR) […]
Janssen Announces Results from MARINER and COMMANDER HF Studies
MUNICH, Aug. 27, 2018 /PRNewswire/ — The Janssen Pharmaceutical Companies of Johnson & Johnson today announced the results of the Phase 3 MARINER and COMMANDER HF (heart failure) studies, which were presented this week at the European Society of Cardiology (ESC) Congress 2018 and simultaneously published in The New England Journal of Medicine. In both studies, there was no […]
HeartFlow Analysis Enables Physicians to Confidently and Safely Differentiate Patients in Need of Invasive Treatment and Patients Who Can be Managed Medically for Coronary Artery Disease
MUNICH–(BUSINESS WIRE)–Late-breaking data published and presented today confirm that the non-invasive HeartFlow®FFRct Analysis enables physicians to effectively differentiate patients in need of coronary stenting or bypass surgery from those who can be managed with medications alone. The ADVANCE Registry was published in the European Heart Journal (EHJ) and the Aarhus University Hospital […]