DURHAM, N.C., Jan. 9, 2018 /PRNewswire/ — Micell Technologies announced the recent online publication of five-year data from the DESSOLVE I and II trials. The report, “Long-term clinical outcomes of a crystalline sirolimus-eluting coronary stent with a fully bioabsorbable polymer coating: five-year outcomes from the DESSOLVE I and II trials” was peer […]
Coronary/Structural Heart
Neovasc Reports Tiara’s Clinical Case Load Accelerating
VANCOUVER, Jan. 8, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company“) (NASDAQ, TSX: NVCN) today provided an update on its clinical program for the Tiara™ (“Tiara”) valve, a self-expanding mitral bioprosthesis for transcatheter implantation in patients with Mitral Regurgitation (“MR”), one of the most prevalent valvular heart diseases in western countries. MR […]
ABS Sets Their Sights on $20M in New Funding Led by New CFO
Published: Jan 05, 2018 Los Angeles, California, November 26, 2017 – Advanced Bifurcation Systems announced that is it seeking $20 million in new funding from outside investors. The new goal was announced in conjunction with the appointment of new CFO Ken Richards, who brings with him a strong background in both debt […]
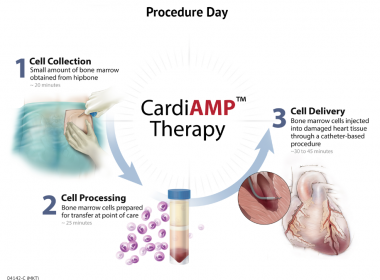
BioCardia Submits Clinical Trial to FDA for New Indication of Chronic Myocardial Ischemia for CardiAMP Cell Therapy
SAN CARLOS, Calif.–(BUSINESS WIRE)-–BioCardia, Inc. [OTC: BCDA], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today reported filing a second Investigational Device Exemption with the FDA for the CardiAMP Chronic Myocardial Ischemia Trial to treat patients with refractory angina. This second potential indication for CardiAMP investigational cell therapy of […]
ReCor Medical Announces Completion of RADIANCE-HTN SOLO Hypertension Study
PALO ALTO, Calif., Jan. 5, 2018 /PRNewswire-USNewswire/ — ReCor Medical announced today randomization of its 146th, and final, subject in the SOLO cohort of its RADIANCE-HTN study. RADIANCE-HTN, ReCor’s study of its Paradise Renal Denervation System in people with hypertension, involves two study cohorts: “SOLO” – whose subjects are removed from anti-hypertensive […]
ABS Release: Major Heart Treatment to Eliminate Thousands of Open Heart Surgeries
Los Angeles, California, November 20, 2017 – Advanced Bifurcation Systems is developing a new procedure that could potentially eliminate the need for open-heart surgery in thousands of patients every year. Their system, which is currently going through trials, could make stenting branched arteries much simpler and less time consuming. It would also […]
New Self-Expanding Valve Addresses the Achilles’ Heel of TAVR
By Ken Dropiewski, Prime-Core Executive Search (ken@prime-core.com) The paravalvular leak (PVL) has long been considered the Achilles’ heel of TAVR. The frequent complication of aortic valve replacement occurs at a much higher rate after the transcatheter approach than conventional surgical replacement. Research has indicated that PVL is linked to late […]
Corindus to Participate With Mayo Clinic in Preclinical Study of Remote Control Robotics for Percutaneous Coronary Interventions Remote Treatment Capabilities
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics [NYSE American: CVRS], a leading developer of precision vascular robotics, announced today that it is working with Mayo Clinic in a preclinical study about use of telestenting. Telestenting, or the remote robotic treatment for percutaneous coronary intervention (PCI), may enable physicians to conduct procedures from virtually any location, […]
PlaqueTec Announces First Peer-Reviewed Publication for its Liquid Biopsy System™ (LBS)
CAMBRIDGE, England, January 3, 2018 /PRNewswire/ — Article in JACC: Basic to Translational Science Validates Feasibility and Unique Potential of LBS to Detect Biomarkers for Coronary Artery Disease PlaqueTec today announced the publication of the first peer-reviewed article demonstrating the feasibility and potential of the PlaqueTec Liquid Biopsy System (TM) (LBS) […]
Tryton Medical Names Former Johnson & Johnson Exec as New CEO
DURHAM, N.C.–(BUSINESS WIRE)– Tryton Medical, Inc., the leading developer of stents designed to definitively treat bifurcation lesions, today announced that the company has appointed Carl J. St. Bernard as president and chief executive officer. St. Bernard has a long track record growing new and established businesses in pharmaceuticals, consumer health products, […]