DENVER–(BUSINESS WIRE)– Mitralign, Inc.., an innovative developer of transcatheter tricuspid and mitral valve repair solutions, today released 1 year follow up data from the SCOUT I Early Feasibility Study designed to evaluate the performance of its Trialign™ System. The data presented by Rebecca Hahn, M.D., New York-Presbyterian/Columbia University Medical Center, continue […]
Coronary/Structural Heart
Medtronic Evolut(TM) PRO TAVR System with Advanced Sealing Maintains Excellent Outcomes Over Time
DUBLIN and DENVER – November 2, 2017 – Medtronic plc (NYSE:MDT) today presented new data at the Transcatheter Cardiovascular Therapeutics (TCT) Annual Meeting showcasing the excellent clinical performance of the Evolut(TM) Transcatheter Aortic Valve Replacement (TAVR) platform. Six-month data from the newest-generation Evolut PRO System demonstrated continued benefits of its unique valve […]
4C Medical’s Novel Transcatheter Mitral Valve Presented at TCT 2017 Shark Tank Innovation Competition
BROOKLYN PARK, Minn., Nov. 2, 2017 /PRNewswire/ — 4C Medical Technologies, Inc., a developer of minimally invasive technologies for structural heart disease, today announced that its medical device for mitral regurgitation (MR) was presented at the Shark Tank Innovation Competition at the Transcatheter Cardiovascular Therapeutics (TCT) meeting held October 29-November 2, […]
Medtronic Shows Off Early Intrepid TMVR Data
DUBLIN and DENVER – November 1, 2017 – Medtronic (NYSE:MDT) today presented positive data for its self-expanding Intrepid(TM) transcatheter mitral valve replacement (TMVR) system in patients with severe, symptomatic mitral valve regurgitation. Presented as a First Report Investigation at the Transcatheter Cardiovascular Therapeutics (TCT) Annual Meeting and simultaneously published in the Journal of the American […]
Elixir Medical Corporation Unveils Transformational DynamX™ Stent Featuring Adaptive Segments that Uncage the Stented Artery
MILPITAS, Calif.–(BUSINESS WIRE)– Elixir Medical, a leader in the development of breakthrough adaptive remodeling technologies designed to mimic the normal arterial function after cardiovascular and peripheral vascular disease intervention, unveiled for the first time its novel stent technology, DynamXTM, a bioadaptive drug eluting stent (BA-DES) platform at the Transcatheter Cardiovascular Therapeutics […]
TCT 2017: BIOTRONIK Symposium Highlights Differing Roles of Drug-Eluting Stents and Magnesium Scaffolds in Clinical Practice
Denver, United States / Buelach, Switzerland, 31.10.2017 (PresseBox) – Data presented at the BIOTRONIK-sponsored symposium on the Orsiro1 drug-eluting stent (DES) demonstrate why Orsiro is gaining prominence in the crowded DES market. In addition, clinical and preclinical data presented about Magmaris2 reinforce the case that the magnesium-based resorbable technology does not share the same risk of […]
OrbusNeich’s COMBO Dual Therapy Stent Demonstrates Non-Inferiority vs the Market Leading XIENCE™ Everolimus Eluting Stent
DENVER, October 30, 2017 /PRNewswire/ — New results from the HARMONEE Japan/US Registration Trial, reported by OrbusNeich today in the First Report Investigations session at the 29th annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium, demonstrate that the COMBO(TM) Dual Therapy Stent (DTS) compares favorably to the market leading option for treating patients […]
Abbott Shows off Landmark XIENCE Stent Data
DENVER, Oct. 30, 2017 /PRNewswire/ — Abbott (NYSE: ABT) today announced that patients who underwent minimally-invasive implantation with a XIENCE coronary stent for left-main coronary artery disease had the same long-lasting health outcomes at three years but felt better more quickly than patients who underwent open-heart surgery. The data were presented during […]
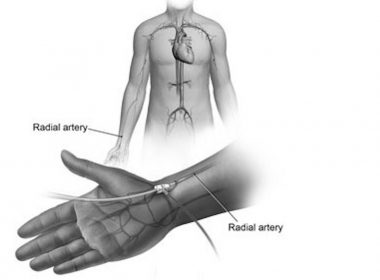
Transradial Interventions can Cause Permanent Vascular Damage, Study Shows
Studies over the past two decades have shown that transradial access for PTCA interventions to have an advantage over transfemoral. Specifically, that access-site and bleeding complications are nearly eliminated. This was an important advantage because although there have been advances in interventional techniques, antithrombotic therapy, and closure devices, bleeding complications […]
Analytics 4 Life to Present New Clinical Data on Novel Cardiac Imaging Technology Using Machine-Learned Algorithms at the Transcatheter Cardiovascular Therapeutics (TCT) 2017 Scientific Symposium
RESEARCH TRIANGLE PARK, N.C. and TORONTO, Oct. 30, 2017 /PRNewswire/ — Analytics 4 Life, a digital health company applying the power of artificial intelligence to develop solutions that improve existing care pathways, today announced it will be presenting new clinical data on the Company’s ongoing Coronary Artery Disease Learning and Algorithm Development (CADLAD) study at […]