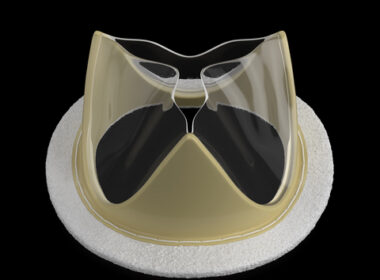
TEL AVIV, Israel, June 9, 2025 /PRNewswire/ — Microtech, a wholly owned subsidiary of Medinol, is happy to announce the first U.S. implantations of its atrial-pressure microsensor. Two surgical implantations were performed on Friday, May 16, 2025, at New York-Presbyterian/Columbia…
Coronary/Structural Heart
Pepin Heart Institute at AdventHealth Tampa becomes first in Tampa Bay to complete 1,000th WATCHMAN™ heart procedure
TAMPA, Fla., June 6, 2025 /PRNewswire/ — The heart care experts at the Pepin Heart Institute, a part of AdventHealth Tampa, are now the first in the Tampa Bay area to complete 1,000 WATCHMAN ™ procedures, a minimally-invasive surgery that helps prevent stroke and cardiovascular death in…
Foldax Secures Approval for TRIA Mitral Heart Valve in India
SALT LAKE CITY–(BUSINESS WIRE)–Foldax® Inc., a leader in heart valve innovation, today announced that the Indian Central Drugs Standard Control Organization (CDSCO) approved its TRIA™ Mitral Valve. Dolphin Life Science India LLP will locally manufacture the TRIA Mitral Valve in India. Foldax’s vision for its novel polymer heart valves is […]
FineHeart Receives ANSM1 Authorization to Deploy Its First-In-Human Clinical Study in France
Patients with advanced heart failure will be treated with FlowMaker®, an innovative, fully implantable device for restoring cardiac output. BORDEAUX, France–(BUSINESS WIRE)–FineHeart S.A., a clinical-stage medical technology company specializing in the development of innovative solutions for cardiology, announces that it has received authorization from the French National Agency for the […]
MemorialCare Heart and Vascular Institute Completes First Successful TriClip Procedure for Tricuspid Valve Repair
LONG BEACH, Calif., June 4, 2025 /PRNewswire/ — The cardiology team at the MemorialCare Heart & Vascular Institute at Long Beach Medical Center, led by Chief of Cardiology David Shavelle, M.D., performed Long Beach Medical Center’s first transcatheter tricuspid valve replacement…
Vasa Therapeutics Announces Successful Completion of First-in-Human Study for Investigational Therapy VS-041, a Small Molecule Drug Candidate for Heart Failure with Preserved Ejection Fracture
VS-041 safe and well tolerated in healthy patients Biomarker trial in patients with HFpEF targeted to initiate in 2025 ENCINITAS, Calif. and WROCLAW, Poland, June 4, 2025 /PRNewswire/ — Vasa Therapeutics (“Vasa”), a clinical stage biopharmaceutical company developing novel therapies for…
FastWave Medical Announces Successful First-in-Human Procedures in Study of Sola™, Its Next-Generation Coronary Laser IVL System
Initial FIH procedures launch FastWave’s multi-center feasibility study evaluating the safety and performance of its laser-based coronary IVL platform. MINNEAPOLIS, June 3, 2025 /PRNewswire/ — FastWave Medical, a pioneering intravascular lithotripsy (IVL) startup, today announced the…
Bitterroot Bio Announces Dosing of First Patient in MATADOR Phase 2a Study of BRB-002, an Immunomodulating Therapy Targeting CD47 in Atherosclerotic Cardiovascular Disease
-Dosing initiated in proof-of-concept MATADOR Phase 2a study- -Initial results expected by year-end 2026- PALO ALTO, Calif. and NEEDHAM, Mass., June 02, 2025 (GLOBE NEWSWIRE) — Bitterroot Bio, a leader in developing innovative medicines in the field of cardio-immunology, today announced the dosing of the first patient in its proof-of-concept MATADOR Phase 2a study in Australia, a randomized, double-blinded, and placebo-controlled trial evaluating BRB-002 in patients with established atherosclerosis. BRB-002 is a novel, investigational, immunomodulating protein therapy that targets CD47 as a potential treatment option for patients with atherosclerotic cardiovascular disease (ASCVD). By targeting CD47, BRB-002 directly acts on atherosclerotic plaque through the immunomodulation of macrophages, an approach distinct from current standards of care. This mechanism holds the potential to reduce vascular inflammation, a key driver of plaque rupture, as well as plaque burden, a significant risk factor for major adverse cardiovascular events (MACE). Dosing initiation follows favorable Phase 1 single ascending dose results, presented at the American College of Cardiology (ACC) Scientific Sessions earlier this year. BRB-002 was safe at all doses tested, with no serious adverse events (SAEs) observed. Notably, there was no clinically significant impact on hematologic parameters, including no observed anemia, thrombocytopenia, or febrile neutropenia. This encouraging safety data was accompanied by robust, dose-dependent target engagement, as measured by receptor occupancy (RO) on circulating blood cells, ranging up to 100% CD47 RO at the highest doses evaluated. The ongoing proof-of-concept MATADOR Phase 2a study will evaluate the safety and tolerability of multiple doses of BRB-002 in patients with ASCVD after 13 weeks of treatment. Additional clinical endpoints include pharmacokinetics and pharmacodynamics of BRB-002, as well as the change from baseline in carotid artery plaque inflammation as measured by 18F-FDG-PET/CT imaging. The study will enroll patients with imaging-documented carotid atherosclerosis in one or more dose exploration cohorts followed by a larger expansion cohort. The Company expects to report initial results from the MATADOR Phase 2a study by year-end 2026. “Today marks a significant milestone as we initiate dosing in our proof-of-concept MATADOR Phase 2a study. While current standards of care for ASCVD, particularly lipid-lowering therapies, have improved outcomes, many patients continue to face substantial residual risk, and there is considerable need for new approaches that address different pathological pathways,” said Craig Basson, MD, PhD, Chief Medical Officer of Bitterroot Bio. “This Phase 2a study will provide crucial insights into the safety, tolerability, and biological activity of our innovative approach with BRB-002. We are optimistic that targeting atherosclerotic plaque directly may lower vascular inflammation and residual risk, ultimately improving long-term cardiovascular health for ASCVD patients.” About BRB-002BRB-002 is a novel protein therapy that is being evaluated as an investigational agent in atherosclerotic cardiovascular disease (ASCVD). BRB-002 is an immunomodulatory agent that inhibits the CD47 cell surface receptor (“don’t eat me” signal) to ameliorate the underlying causes of atherosclerosis and reduce inflammatory plaque burden. A Phase 1 clinical study of BRB-002 was completed in Australia by Bitterroot Australia Pty Ltd, a wholly owned subsidiary of Bitterroot Bio, Inc. About Bitterroot BioBitterroot Bio, Inc. is a pioneer in the field of cardio-immunology, which investigates the interplay between the immune system and cardiovascular health. Bitterroot Bio’s research seeks to uncover critical roles that immune modulators play in the progression of cardiovascular disease. By targeting these diseases in this novel way, Bitterroot Bio’s mission is to transform the lives of patients suffering from cardiovascular diseases. For more information, please visit https://www.brbio.com or follow us on Facebook LinkedIn, or X. Media Contact: Pablo Fenton (Bitterroot Bio), media@brbio.com
Simplex Quantum Selected for MedTech Innovator Asia Pacific 2025 Accelerator
AI-Powered Heart Failure Detection Technology Recognized Among Asia-Pacific’s Top 20 MedTech Startups TOKYO, May 29, 2025 /PRNewswire/ — Simplex Quantum, a Tokyo-based medical technology startup specializing in AI-driven heart health solutions, has been selected as one of 20…
HeartSciences Signs First MyoVista Insights™ Platform Customer, Marking Major Commercial Milestone
Southlake, TX, May 29, 2025 (GLOBE NEWSWIRE) — HeartSciences Inc. (Nasdaq: HSCS; HSCSW) (“HeartSciences” or the “Company”), an artificial intelligence (“AI”)-powered medical technology company focused on transforming ECGs/EKGs to enable earlier detection of heart disease, today announced the signing of its first commercial customer for the MyoVista Insights™ platform: Westcliffe Health Innovations, based in the United Kingdom. This milestone follows the recent launch of MyoVista Insights and the initiation of an early adopter program for reference sites earlier this month. Westcliffe Health Innovations provides cardiology services to the U.K. National Health Service (NHS) in the Bradford and Leeds region. As the largest publicly funded healthcare system in the world, the NHS plays a vital role in pioneering access to innovative healthcare technologies. The implementation of MyoVista Insights at Westcliffe is intended to position it as a key reference site for HeartSciences, particularly within the U.K. market. MyoVista Insights is a cloud-native, next-generation ECG management system built to enhance clinical decision-making and improve healthcare IT efficiency. It is being developed as the first vendor- and device-agnostic ECG platform capable of running AI-ECG algorithm models from multiple partners—streamlining ECG workflows and helping reduce operational costs for healthcare providers. Dr. Matthew Fay, Director of Cardiology at Westcliffe, is a leading figure in bringing cardiology services to the frontlines of healthcare across the U.K. He founded a 27-partner GP super-practice, has contributed to the development of both local and national clinical guidelines, and has led efforts to transition cardiovascular services from hospitals into community settings over the past several years. “In an area with socio-economic challenges and higher-than-average rates of cardiovascular disease, ensuring easy access to ECGs, the gateway cardiac investigation, is imperative. While healthcare professionals recognize the importance of ECGs, the ability to interpret them varies among generalists. This partnership facilitates easy access to ECGs and provides expert interpretation right in the centre of the community, with results stored in the shared patient record for seamless access.” — Dr. Matthew Fay, Clinical Director of Cardiology, Westcliffe Health Innovations, U.K. “We are thrilled to partner with Dr. Matthew Fay and Westcliffe Health Innovations as the first commercial adopter of MyoVista Insights,” said Andrew Simpson, CEO of HeartSciences. “Dr. Fay and his team have been trailblazers in advancing cardiovascular care across the NHS, and we share a common vision of transforming frontline screening through innovative technology. This agreement represents a pivotal step forward in HeartSciences’ commercial journey and validates our belief in the future of AI-driven ECG management.” For more information about MyoVista Insights https://www.heartsciences.com or X: @HeartSciences and for those interested in exploring collaboration or early adoption opportunities, please contact HeartSciences info@heartsciences.com About HeartSciences HeartSciences is a medical technology company focused on applying innovative AI-based technology to an ECG (also known as an EKG) to expand and improve an ECG’s clinical utility. Millions of ECGs are performed every week and the Company’s objective is to improve healthcare by making it a far more valuable cardiac screening tool, particularly in frontline or point-of-care clinical settings. HeartSciences has one of the largest libraries of AI-ECG algorithms and intends to provide these AI-ECG algorithms on a device agnostic cloud-based solution as well as a low-cost ECG hardware platform. Working with clinical experts, HeartSciences ensures that all solutions are designed to work within existing clinical care pathways, making it easier for clinicians to use AI-ECG technology to improve their patient’s care and lead to better outcomes. HeartSciences’ first product candidate for FDA clearance, the MyoVista® wavECG™, or the MyoVista®, is a resting 12-lead ECG that is also designed to provide diagnostic information related to cardiac dysfunction which has traditionally only been available through the use of cardiac imaging. The MyoVista® also provides conventional ECG information in the same test. For more information, please visit: https://www.heartsciences.com. X: @HeartSciences Safe Harbor Statement This announcement contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements are made under the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995 and are relating to the Company’s future financial and operating performance. All statements, other than statements of historical facts, included herein are “forward-looking statements” including, among other things, statements about HeartSciences’ beliefs and expectations. These statements are based on current expectations, assumptions and uncertainties involving judgments about, among other things, future economic, competitive and market conditions and future business decisions, all of which are difficult or impossible to predict accurately and many of which are beyond the Company’s control. The expectations reflected in these forward-looking statements involve significant assumptions, risks and uncertainties, and these expectations may prove to be incorrect. Investors should not place undue reliance on these forward-looking statements, which speak only as of the date of this press release. Potential risks and uncertainties include, but are not limited to, risks discussed in HeartSciences’ Annual Report on Form 10-K for the fiscal year ended April 30, 2024, filed with the U.S. Securities and Exchange Commission (the “SEC”) on July 29, 2024, HeartSciences’ Quarterly Report on Form 10-Q for the fiscal quarter ended July 31, 2024, filed with the SEC on September 12, 2024, HeartSciences’ Quarterly Report on Form 10-Q for the fiscal quarter ended October 31, 2024, filed with the SEC on December 16, 2024, HeartSciences’ Quarterly Report on Form 10-Q for the fiscal quarter ended January 31, 2025, filed with the SEC on March 13, 2024 and in HeartSciences’ other filings with the SEC at www.sec.gov. Other than as required under the securities laws, the Company does not assume a duty to update these forward-looking statements. Investor Relations: Integrous CommunicationsMark KomonoskiPartnerPhone: 877-255-8483Email: mkomonoski@integcom.us Media Contact: HeartSciences Gene Gephart +1-682-244-2578 Ext. 2024 info@heartsciences.com