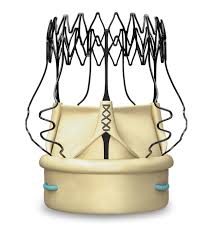
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN) (“LivaNova” or the “Company”), a market-leading medical technology company, today announced the publication of the analysis, “The Perceval® Sutureless Aortic Valve: Review of Outcomes, Complications and Future Direction,” by Powell, Pelletier, Chu, Bouchard, Melvin and Adams in the latest issue of Innovations1. The literature review, which included […]
Coronary/Structural Heart
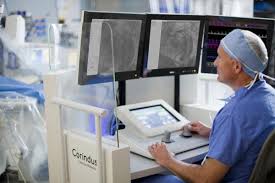
Corindus Announces First Patient Enrolled in PRECISION GRX Registry
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. [NYSE American: CVRS], a leading developer of precision vascular robotics, today announced first patient enrollment in the PRECISION GRX Registry, a post-market study to continue market surveillance of the Company’s second generation CorPath GRX System. The PRECISION GRX Registry will include up to 25 sites […]
Abbott is stopping the sale of Absorb first generation BVS in all markets
Time to throw in the towel for Abbott on this generation of their Absorb BVS because of slow sales. However, there could still be a platform for this technology… Abbott halts global sales of 1st-gen Absorb stents SEPTEMBER 8, 2017 BY FINK DENSFORD , MassDevice Abbott (NYSE:ABT) said today it will halt global sales […]
Micro Interventional Devices Completes First Clinical Tricuspid Bicuspidization Procedure Utilizing MIA Technology
NEWTOWN, Pa., Sept. 7, 2017 /PRNewswire/ — Micro Interventional Devices, Inc.(MID) has successfully completed the first in human tricuspid valve bicuspidization procedure utilizing its MIA (Minimally Invasive Annuloplasty) technology. The successful bicuspidization resulted in a 34.5% reduction in valve area, reducing the patient’s tricuspid regurgitation from severe/moderate to trace. The patient was the […]
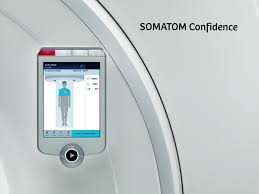
Siemens Healthineers introduces Somatom Confidence RT Pro CT Scanner
Erlangen, 2016-Sep-27 Somatom Confidence RT Pro is a dedicated CT for radiation therapy DirectDensity algorithm delivers electron density images independent of kV settings, ensuring a smooth simple dose calculation workflow New version of syngo.via RT Image Suite software offers advanced treatment of moving tumors Siemens Healthineers introduced its new Somatom […]
BioCardia Receives U.S. Patent Covering A Method Of Preparing And Characterizing Mesenchymal Cells, Providing Further Protection To Cardiallo Cell Therapy Program
SAN CARLOS, Calif.–(BUSINESS WIRE)–BioCardia®, Inc. [OTC:BCDA], a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, with clinical programs in heart failure and sub-acute infarction, today announced the issuance of United States Patent No. 9,301,975 relating to a method of producing mesenchymal stem cells from bone marrow cells in […]
Cardiac Insight, Inc. Continues Steady Beat of Progress with New $4.5 Million Investment and Attracts Prestigious Healthcare M&A Executive, Clifford J. Stocks, to Its Board of Directors
KIRKLAND, Wash.–(BUSINESS WIRE)–Cardiac Insight, Inc., a U.S. developer of wearable medical devices and diagnostic software, announced today that the company continues to experience a steady beat of market traction and industry endorsement with a recent infusion of $4.5 million into their new C-1 round of funding, currently capped at $10 […]
NaviGate Cardiac Structures, Inc. (“NCSI”) reports world’s first successful transcatheter implantation of 52-mm tricuspid valve into a transplanted-heart patient
LAKE FOREST, Calif., Aug. 30, 2017 – NaviGate Cardiac Structures Inc. (“NCSI”) announced today that its GATE™ catheter-guided tricuspid atrioventricular valved stent (A VS) was implanted through the jugular vein six weeks ago into a patient’s transplanted heart that was failing due to severe tricuspid valve insufficiency. The successful implantation […]
Data Published in The Lancet Shows Positive Results at Primary Endpoint in the Treatment of Resistant Hypertension with MobiusHD Device
MOUNTAIN VIEW, Calif., Sept. 01, 2017 (GLOBE NEWSWIRE) — Vascular Dynamics, Inc. (VDI), a privately held medical device company developing novel solutions for the treatment of hypertension, today announced European results of the company’s first-in-human trial of its MobiusHD®implant published in The Lancet. The publication reported an average reduction of 24-hour […]
Neovasc’s Reducer System Reviewed in European Heart Journal
VANCOUVER, Sept. 5, 2017 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company“) (NASDAQ, TSX: NVCN) today announced the European Heart Journal published a clinical review of its Neovasc Reducer™ (“Reducer”) device for the treatment of refractory/persistent angina pectoris, a common and disabling medical condition, and a major public health problem that affects […]