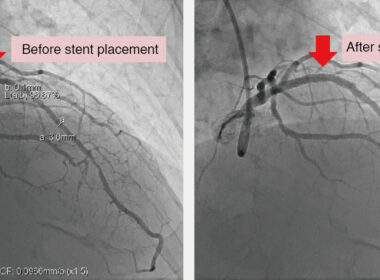
SAN FRANCISCO, April 4, 2024 – Osso VR, the leader in immersive procedural training, announces the release of a co-developed curriculum with the American College of Cardiology (ACC). This collaboration expands Osso VR’s presence in the cardiovascular sector. With the integration of Osso VR’s immersive technology and the expertise of […]
Coronary/Structural Heart
Cardio Diagnostics Holdings, Inc. Enters Into a Nationwide Telehealth Agreement with Navierre
CHICAGO–(BUSINESS WIRE)–Cardio Diagnostics Holdings, Inc. (NASDAQ: CDIO), an AI-driven precision cardiovascular medicine company, announces a strategic partnership with Navierre, an innovative digital health technology platform which revolutionizes how patients access healthcare and serves as a true health companion for every stage from birth to the golden years with a robust care […]
Johnson & Johnson to Showcase its Broad Scientific Leadership and Latest Innovations to Combat Cardiovascular Disease at ACC.24
Clinical and real-world evidence presentations highlight how the Company is transforming care for patients who are fighting some of the most common and devastating cardiovascular diseases NEW BRUNSWICK, N.J., April 4, 2024 /PRNewswire/ — Johnson & Johnson announced today that clinical…
Viz.ai® Announces Three New Studies Supporting Viz HCM™ Module for the Detection and Management of Suspected Hypertrophic Cardiomyopathy
April 03, 2024 08:00 AM Eastern Daylight Time SAN FRANCISCO–(BUSINESS WIRE)–Viz.ai, the leader in AI-powered disease detection and intelligent care coordination, today announced new clinical data supporting advancements in cardiology care. Three studies, presented at the American College of Cardiology’s (ACC) Annual Scientific Session & Expo 2024, have shown positive […]
GE HealthCare Introduces Caption AI on Vscan Air SL Wireless Handheld Ultrasound System to Help More Clinicians Capture Diagnostic-Quality Cardiac Images
April 03, 2024 08:00 AM Eastern Daylight Time CHICAGO–(BUSINESS WIRE)–GE HealthCare (Nasdaq: GEHC) today announced the launch of Caption AI artificial intelligence (AI)-driven software for rapid cardiac assessments at the point of care on Vscan Air SL. Now, with Caption AI technology, clinicians using Vscan Air SL handheld ultrasound will […]
Protembis Announces First Patient Enrolled in the PROTEMBO IDE Pivotal Trial
NEW YORK & AACHEN, Germany–(BUSINESS WIRE)–Protembis GmbH (Protembis), a privately-held emerging cardiovascular medical device company, announced today the enrollment of the first patient in the PROTEMBO Investigational Device Exemption (IDE) Pivotal Trial (NCT05873816). The trial will enroll between 250-500 randomized patients undergoing transcatheter aortic valve replacement (TAVR) in the USA […]
HeartSciences to Present at the LD Micro Invitational XIV Conference
Southlake, TX, April 02, 2024 (GLOBE NEWSWIRE) — Heart Test Laboratories, Inc. d/b/a HeartSciences (Nasdaq: HSCS; HSCSW) (“HeartSciences” or the “Company”), an artificial intelligence (AI)-powered medical technology company focused on transforming ECGs/EKGs to save lives through earlier detection of heart disease, today announced that Chief Executive Officer Andrew Simpson is scheduled to present at the LD Micro Invitational XIV Conference on Tuesday April 9, 2024 at 5:00pm ET at the Sofitel New York. Mr. Simpson will also be hosting one-on-one meetings through the conference.
Innovating “Treadmill Exercise Test AI-Assisted Interpretation System,” CMUH(Taiwan) Timely Saves More Patients with Severe Myocardial Infarction
TAICHUNG, Taiwan, April 2, 2024 /PRNewswire/ — The treadmill exercise test (TET) is an important tool for diagnosing coronary artery disease (CAD), but limited by time spent for the manual interpretation of a dozen charts and the challenges in identifying minor deviations. Traditionally,…
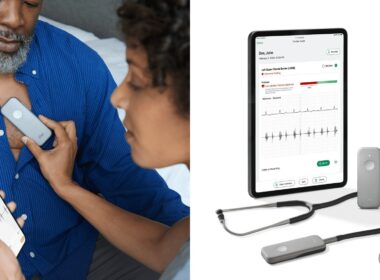
FDA Clears First AI to Aid Heart Failure Detection During Routine Check-ups
Developed with Mayo Clinic, Eko Health’s Low Ejection Fraction (Low EF) AI gives healthcare professionals a powerful tool to more accurately assess possible heart failure in at-risk patients during a standard physical exam SAN FRANCISCO, April 2, 2024 /PRNewswire/ — Eko Health, a pioneer…
Arch Biopartners Adds Three New Clinical Sites into the Phase II Trial for LSALT Peptide Targeting Cardiac Surgery Associated-Acute Kidney Injury
TORONTO, April 02, 2024 (GLOBE NEWSWIRE) — Arch Biopartners Inc., (“Arch” or the “Company”) (TSX Venture: ARCH and OTCQB: ACHFF), announced today that three additional clinical sites in Turkey have joined the Phase II trial for LSALT peptide targeting the prevention and treatment of cardiac surgery-associated acute kidney injury (CS-AKI). LSALT peptide is the Company’s lead drug candidate for preventing and treating inflammation injury in the kidneys, lungs and liver.