First clinical cases successfully completed; U.S. launch to be led by Kaneka’s Neurovascular division First clinical cases successfully completed; U.S. launch to be led by Kaneka’s Neurovascular division
Neuro
Route 92 Medical Enrolls First Patient in SUMMIT RISE Study
SUMMIT RISE is expected to enroll up to 500 patients to evaluate the performance of Route 92 Medical’s neurovascular intervention device portfolio WEST JORDAN, Utah, March 9, 2026 – (GLOBE NEWSWIRE) — Route 92 Medical, Inc., a privately held medical technology company dedicated to improving outcomes for patients undergoing […]
Medtronic to acquire Scientia Vascular, marrying access and therapeutic portfolios for neurovascular care
Deal complements Medtronic’s Neurovascular portfolio, delivering simplicity and access when ‘time is brain’ GALWAY, Ireland, March 10, 2026 /PRNewswire/ — Medtronic today announced its entry into a definitive agreement to acquire Scientia Vascular, a company with critical, innovative…
VS3 Medical Announces First Patients Enrolled in First-in-Human Study of Novel Neurovascular Implant to Address Symptomatic Venous Sinus Stenosis
SAN JOSE, Calif.–(BUSINESS WIRE)– #PT–VS3 Medical announced the first patients enrolled in first-in-human study evaluating VS3 Medical™ Stent System for symptomatic venous sinus stenosis.
Harrison.ai Receives FDA Clearance for Acute Infarct Triage on Non-contrast CT Brain
Approval marks Harrison.ai’s second out of three Breakthrough designations to reach marketing authorization, and 9th FDA clearance covering a total of 13 radiological findings. SYDNEY & BOSTON–(BUSINESS WIRE)–Harrison.ai today announced FDA 510(k) clearance for acute infarct triage on non-contrast CT Brain. Acute infarct is one of the most difficult findings to […]
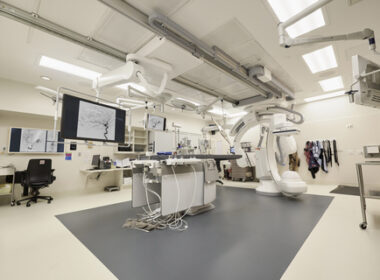
Children’s Minnesota Unveils Region’s First Dedicated Pediatric Neurointerventional Angiography Suite to Treat Stroke Patients
MINNEAPOLIS–(BUSINESS WIRE)–Children’s Minnesota is pleased to announce a $2.5 million, state-of-the-art biplane neuroangiography suite—the region’s only facility dedicated to pediatric neurointerventional procedures. This advanced imaging technology brings new treatment options for children with a variety of cerebrovascular diseases, further expanding Children’s Minnesota’s neurosurgical capabilities. “With this advanced biplane neuroangiography suite, we are raising the bar for children in
Spryte Medical Announces First Participants Enrolled in the INSYTE Trial at Baptist Health Jacksonville
FDA-approved IDE trial to evaluate the safety and effectiveness of the nOCT™ Imaging System to provide quality images for diagnostic assessment during endovascular intracranial aneurysm treatment and follow-up FDA-approved IDE trial to evaluate the safety and effectiveness of the nOCT™ Imaging System to provide quality images for diagnostic assessment during endovascular intracranial aneurysm treatment and follow-up
LRS SECURES IDE TO STUDY ITS THERMOSUIT COOLING DEVICE IN A PIVOTAL TRIAL OF ISCHEMIC STROKE PATIENTS
ALEXANDRIA, La., March 2, 2026 /PRNewswire/ — Life Recovery Systems (LRS) has received an Investigational Device Exemption (IDE) from the US Food and Drug Administration (FDA) for testing of the ThermoSuit System (TSS) in a pivotal trial of ischemic stroke patients. The previous SISCO…
Neuro Tech Forum 2026
By partnering with the Neuro Tech Forum, CardiacVascular News gives its audience a front row seat to where neuro innovation is headed. The event convenes founders, researchers, clinicians, and investors who are actively building and backing the next wave of neurotechnology. It is a strong editorial fit for CardiacVascular News’ […]
RapidAI Highlights Research Depth and Industry Leadership at ISC 2026 With 28 Clinical Abstracts
New clinical data show AI-driven gains in aneurysm growth detection, stroke interpretation accuracy, and radiology workflow efficiency across enterprise imaging environments. SAN MATEO, CA (February 4, 2026) – RapidAI, the pioneer of deep clinical AI and global leader in enterprise imaging, today announced details of 28 scientific abstracts accepted at the International Stroke Conference (ISC) […]