SANTA CLARA, Calif.–(BUSINESS WIRE)–Shape Memory Medical Inc. announced today it has received CE mark for its TrelliX® Embolic Coil System. The TrelliX Embolic Coil System is intended to obstruct or occlude blood flow in vascular abnormalities of the neurovascular and peripheral vessels. CE-marked indications include: Intracranial aneurysms, other neurovascular abnormalities […]
Neuro
Penumbra Introduces Latest Stroke Thrombectomy Aspiration Technology at SNIS 2019
MIAMI–(BUSINESS WIRE)–Penumbra, Inc. (NYSE: PEN), a global healthcare company focused on innovative therapies, today announced U.S. commercial availability of the Penumbra System’s most advanced technology – the Penumbra JET 7 Reperfusion Catheter with XTRA FLEX technology – at the Society of NeuroInterventional Surgery (SNIS) 16th Annual Meeting in Miami. Penumbra JET […]
Total Brain Joins the American Heart Association’s Center for Health Technology & Innovation to Study Uses of Heart Rate Variability Data to Reduce Stress and Risk of Cardiovascular Incidents
SAN FRANCISCO, June 22, 2019 /PRNewswire/ — Total Brain, a neuroscience-based mental health and fitness platform, is joining the American Heart Association’s Center for Health Technology & Innovation’s (the Center) Innovators Network. The Association’s Center is focused on building and fostering health technologies and relationships in pursuit of innovative and scalable solutions across the healthcare […]
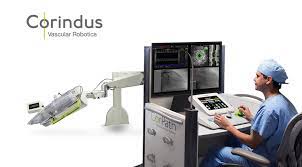
Corindus Announces Regulatory Approval of CorPath GRX for Neurovascular Intervention in Australia and New Zealand
Expanded indication provides access to robotic technology in vascular procedures from head to toe July 17, 2019 06:45 AM Eastern Daylight Time WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. [NYSE American: CVRS], a leading developer of precision vascular robotics, announced today that it received approval from the Australian Therapeutic Goods Administration […]
Q’Apel Medical Announces FDA Clearance for Walrus Balloon Guide Catheter
FREMONT, Calif., July 17, 2019 /PRNewswire/ — Q’Apel Medical announced today that it received U.S. Food and Drug Administration clearance for its novel walrus Balloon Guide Catheter (BGC). In the neurovascular space where speed, reliability and performance are so critical to patient outcomes, physicians and patients demand that innovative technologies are best-in-class. The walrus BGC, […]
RAPID Receives Registration Approval in Japan
MENLO PARK, Calif.–(BUSINESS WIRE)–iSchemaView, the worldwide leader in advanced imaging for stroke, has received registration approval under the Japanese Pharmaceutical Affairs Law through third party review by the Japanese Association for the Advancement of Medical Equipment for its RAPID imaging platform. With this news the technology is available to hospitals […]
BIOXYTRAN’S MDX VIEWER DEMONSTRATES EARLY WARNING SIGNS OF BRAIN HEALTH DURING CARDIOVASCULAR SURGERIES
MDX Viewer use during open heart surgery is an essential vital sign that could mitigate the risk of brain damage caused by prolonged oxygen deprivation during perioperative cardiovascular procedures and validates tissue oxygenation in the brain for stoke patients BOSTON, MASSACHUSETTS, June 06, 2019 (GLOBE NEWSWIRE) — BIOXYTRAN, INC. (OTC: […]
Balt USA, LLC Announces CE Mark Obtained for Titan™ Aspiration Catheter
IRVINE, Calif.–(BUSINESS WIRE)–Balt USA, LLC announces today it has obtained CE mark for the Titan aspiration catheter. The Titan catheter line consists of a highly navigable, large bore (.070”) aspiration catheter and a smaller bore (.036”) catheter for navigation and support. Obtaining the CE mark for Titan* enhances the Balt stroke […]
Rapid Medical Announces FDA Approval of Novel Temporary Aneurysm Embolization Assist Device
YOKNEAM, Israel, May 7, 2019 /PRNewswire/ — Rapid Medical, a company focused on the development of next generation neurovascular devices, today announced that its Comaneci device received FDA clearance as a Temporary Coil Embolization Assist Device. The Comaneci is the first and only device in a new category of temporary coil embolization assist devices. […]
Medtronic Announces U.S. Commercial Launch of Solitaire(TM) X Revascularization Device
DUBLIN – April 30, 2019 – Medtronic plc (NYSE:MDT) today announced the U.S. launch of the Solitaire(TM) X Revascularization Device – and its first use in patients for the treatment of acute ischemic stroke. For ten years, the industry-leading Solitaire(TM) Revascularization Device has enabled physicians in helping patients have a better […]